Research Article
Application of Octacalcium Phosphate Collagen Composite to Bone Defects in Humans: A Long-Term Observational Study
Tadashi Kawai1*, Tetsu Takahashi1, Keiko Matsui1, Yushi Ezoe1, Osamu Suzuki2 and Shinji Kamakura3
1Tohoku University Graduate School of Dentistry, Japan
2Division of Craniofacial Function Engineering, Tohoku University Graduate School of Dentistry, Japan
3Division of Bone Regenerative Engineering, Tohoku University Graduate School of Biomedical Engineering, Japan
*Corresponding author: Tadashi Kawai, Department of Oral Medicine and Surgery, Tohoku University Graduate School of Dentistry, Japan
Published: 01 Oct, 2018
Cite this article as: Kawai T, Takahashi T, Matsui K, Ezoe
Y, Suzuki O, Kamakura S. Application
of Octacalcium Phosphate Collagen
Composite to Bone Defects in Humans:
A Long-Term Observational Study. Clin
Surg. 2018; 3: 2130.
Abstract
Clinical research on octacalcium phosphate collagen (OCP/Col) application in bone defect has
demonstrated its high bone regeneration ability. Herein, we conducted a long-term study involving
the clinical evaluation of OCP/Col-applied cases. A successful outcome was confirmed in two
patients followed up for 7 years and 6.5 years. Both cases showed stable radiographic findings of the
bone tissue without any abnormal findings of the oral cavity. To our knowledge, this is the first report
to confirm the long-term stability and safety of newly formed bone after OCP/Colimplantation.
Keywords: Bone regeneration; Bone substitute material; Long-term observation
Introduction
Certain calcium phosphate cement materials have been used as bone substitute materials for
bone augmentation [1-3]. However, the use of these materials was found to lead to conditions, such
as infection [4,5] or lower osteo conductivity, compared with the use of the autologous bone [6].
Therefore, a study of the long-term stability of substitute materials is imperative for reconstructive
surgery. Octacalcium Phosphate (OCP) has been recognized as a bone substitute material with high
osteo conductivity both in vitro and in vivo [7,8] and has been recommended as a precursor of
biological apatite in bones and teeth [9]. In addition, direct evidence of the presence of OCP in
the central part of human dentine crystals has been demonstrated, and apatite has been detected
in the outermost layers of the same crystals [10] as well as in the porcine enamel [11] and sutures
of the mouse calvaria during intramembranous osteogenesis [12]. The osteogenic potential of
OCP was confirmed for the first time in 1991 by implantation under the periosteum of the mouse
calvarium [7]. Subsequently, several studies were conducted on the bone regeneration ability of
OCP, and repair of bone defect using OCP was confirmed in rats [13] and rabbits [14]. Moreover,
the bone regeneration ability of OCP has been confirmed to be superior to that of hydroxyapatite
and beta-tricalcium phosphate both in vitro and in vivo [15-17]. Since OCP is a granular material
of inferior operability, a combination of OCP and atelocollagen (OCP/Col) was used for improving
its operability [18]. Compared with OCP alone, OCP/Col yielded improved operability as well as
bone regeneration ability [18]. On the basis of translational research for clinical application, some
experiments have been performed on the bone regeneration ability of OCP/Col in various dog
models of bone defects, such as tooth extraction hole [19], critical-sized calvarial bone defect [20],
artificial alveolar cleft [21], and mandibular bone defect [22]. Successful bone repair was confirmed
using OCP/Col in each experiment.
OCP/Col was first clinically applied in 10 cases between April 2011 and September 2013 as
part of clinical research approved by the Research Ethics Committee of the Graduate School of
Dentistry, Tohoku University. OCP/Col was used in five cases of tooth extraction socket and five
cases of cystectomy cavity, and the effective bone healing of these bone defects was observed without
infection or allergic reaction. The results of the first application of OCP/Col and the efficacy of OCP/
Col have been previously reported [23-25]. Another group has also reported the clinical use of OCP
granules for the defect after resection of fibrous dysplasia at the mandible and generation of space by
sinus floor elevation [26]. However, these reports have demonstrated
the short-term progress after treatments using OCP, and no longterm
progress has been reported thus far. In the present study, we
investigated the long-term progress of two cases treated using OCP/
Col for bone defects.
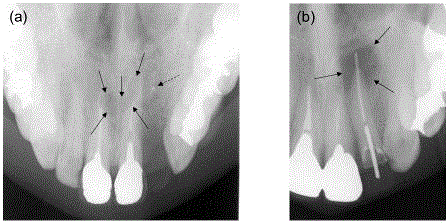
Figure 1
Figure 1
X-ray images of two cases before OCP/Col implantation. (a) Case
1. Arrows indicate a heart-shaped transmission region as a nasopalatine duct
cyst in this X-ray image. Dotted arrow indicates punctiform foreign matter. (b)
Case 2. Arrows indicate a radicular cyst.
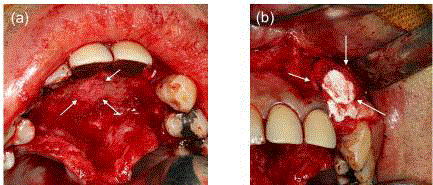
Figure 2
Figure 2
Images of OCP/Col discs implantation in case 1. (a) After the
extraction of the nasopalatine duct cyst, OCP/Col discs were implanted. (b)
After curettage of the tooth extraction cavity at the left upper lateral incisor
region, the OCP/Col discs were implanted. Arrows indicate OCP/Col discs
implanted in a bone defect.
Materials and Methods
Preparation of OCP/Col
OCP was prepared by mixing calcium and phosphate solutions
as previously described [7]. Sieved OCP granules (particle size range:
300-500 μm) obtained from dried OCP were sterilized by heating at
120°C for 2 hr. Our previous study showed that such heating does
not affect the physical properties of OCP granules, such as crystalline
structure or specific surface area [27] although it has been reported
that increasing the temperature to >100°C can induce the collapse of
OCP structure because of dehydration [28,29]. Collagen was prepared
from NMP collagen PS (Nippon Meat Packers, Tsukuba, Ibaraki,
Japan), a lyophilized powder of pepsin-digested atelocollagen isolated
from porcine dermis. NMP collagen PS was dissolved in distilled
water and adjusted to a final concentration of 3% with a pH of 7.4.
OCP/Col was prepared from NMP collagen PS and OCP granules.
OCP was added to concentrated collagen and mixed well. The weight
percentage of OCP in OCP/Col was 77%. This OCP/Col mixture was
then lyophilized and the discs were moulded (9-mm diameter, 1-mm
thickness). The moulded OCP/Col was subjected to de hydro thermal
treatment (150°C, 24 hr) in the Vacuum Drying Oven DP32 (Yamato
Scientific, Tokyo, Japan) and then sterilized using 5-kGy electron
beam irradiation.
Cases
The study trial protocol was approved by the Ethics Committee of
Tohoku University Graduate School of Dentistry (reference numbers
20-27 and 24-31). In this study, case1 was a 45-year-old man who was
tentatively diagnosed with a nasopalatine duct cyst of approximately
10-mm diameter using X-ray examination (Figure 1a). Additionally,
the failure of tooth extraction cavity healing because of the presence
of a punctiform foreign matter in the upper left lateral incisor region
was confirmed (Figure 3a, 4a). OCP/Col was implanted into each
defect after cystectomy at the nasopalatine duct region and curettage
of tooth extraction cavity at the left upper lateral incisor region
under general anaesthesia (Figure 2a, 2b). Histological examination
revealed a nasopalatine duct cyst. At 1 year of OCP/Col implantation,
a titanium dental implant (Brånemark System® Mk III Groovy; Nobel
Biocare Japan K.K., Tokyo, Japan) was inserted into the new bone
region at the left upper lateral incisor region. The final prosthesis
was set and the occlusion was reconstructed at 2 years after OCP/Col
implantation. The patient is presently under observational follow-up
at ongoing visit to our hospital for his dental implant. Case 2 was
a 37-year-old man diagnosed with a radicular cyst of approximately
8-mm diameter at the left upper lateral incisor region based on
X-ray examination (Figure 1b). OCP/Col was implanted into the
defect after cystectomy and apicoectomy under local anaesthesia.
Histological examination revealed a radicular cyst. After the study,
this patient visited the hospital for dental check-up because of injury
to the maxillary region, where OCP/Col was implanted.
Radiographic and clinical examination
Laboratory and radiographic examinations were performed
before cystectomy and at 1 and 7 days and 1, 3, 6 and 12 months
after OCP/Col implantation. Next, radiographic examination was
performed whenever deemed necessary. Computed Tomography
(CT) was performed before cystectomy and at 3 or 6 months after
OCP/Col implantation. The Regions of Interest (ROIs) were defined
at the centre of the augmented region. ROI was circular, with 5-mm
diameter. CT values of ROIs were measured using software (We View
Open-Pacs series, Hitachi Medical Corp., Tokyo, Japan); all values
were reported as mean ± standard deviation. In intraoral findings,
the presence or absence of abnormalities, such as inflammation and
infection was confirmed during each visit.
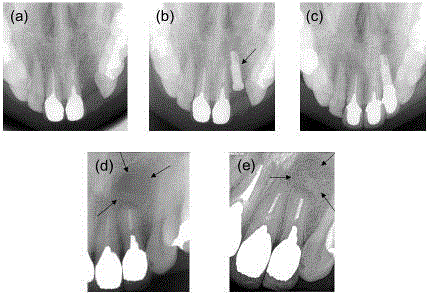
Figure 3
Figure 3
X-ray images of two cases after OCP/Col implantation. (a) Case 1.
Heart-shaped transmission region disappeared at 6 months. (b) Case 1 at 1
year and 3 months. Arrow indicates the dental implant. (c) Case 1 at 7 years.
Showing no significant change. (d) Case 2 at 1 year. Arrows indicates the
cystectomy region. (e) Case 2 at 6 years and 6 months. Bone-like tissue was
confirmed at the cystectomy region (arrows).
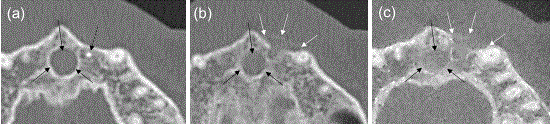
Figure 4
Figure 4
Horizontal CT scans of case 1. (a) Before OCP/Col implantation.
(b) At 3 months after OCP/Col implantation. (c) At 6 months after OCP/Col
implantation. Radiopacity increased at the OCP/Col implantation region.
Black arrows indicate the nasopalatine duct cyst region. Dotted black arrow
indicates foreign body. White arrows indicate the newly formed bone using
OCP/Col at the curettage region.
Figure 5
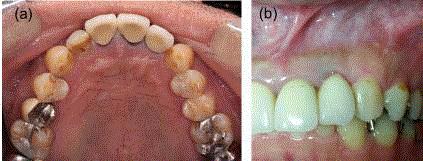
Figure 5
Images of the treated region in each case. (a) Case 1 at 7 years.
(b) Case 2 at 6 years and 6 months. No abnormal findings observed.
Result
Radiographic examination
Although OCP/Col has low radiopacity under normal x-ray
conditions, radiopacity at the OCP/Col implantation region was
noted to increase with time for up to 6 months after cystectomy
in each case. In case 1, a heart-shaped transmission region was
observed before OCP/Col implantation at the nasopalatine duct
region and alveolar bone absorption was detected at the left upper
lateral incisor region on X-ray (Figure 1a). However, these findings
disappeared at 6 months after OCP/Col implantation (Figure 3a). At
1 year and 3 months, a dental implant body was confirmed at the
site repaired with newly formed bone using OCP/Col (Figure 3b).
CT images indicated hard tissue formation with radiopacity at 3 and
6 months after OCP/Col implantation; CT findings also confirmed
that the foreign matter was removed and that the tooth extraction
cavity was healed with the bone-like tissue (Figure 4b, 4c). CT values
of the cystectomy and curettage regions were respectively 300.12 ±
52.05 and 248.63 ± 89.99 at 3 months after OCP/Col implantation
(preoperative values were respectively 121.37 ± 74.03 and 157.27 ±
90.23). The corresponding values of OCP or OCP/Col were 130-140
HU. At 6 months, these values increased to 300.12 ± 52.05 and 378.31
± 56.35, respectively. Even though the surgical sites were confirmed
by X-ray examination at every 6 months, no abnormal findings were
noted, and the dental implant was stable until 7 years after OCP/Col
implantation (Figure 3c). Moreover, in case 2, the X-ray transmission
image became unclear at 1year after implantation (Figure 3d), which
is in concordance with the results, obtained in a previous study [23].
Additionally, the radiopacity further increased, and the boundary
with the surrounding bone became unclear at 6 years and 6 months
after OCP/Col implantation (Figure 3e).
Clinical examination
No abnormal healing, infection and allergic reaction were observed
in any of the treated regions until the last clinical examination (Figure
5a, 5b). Laboratory examination revealed no abnormal findings, and
no infection occurred in any case. A slight increase in C-reactive
protein level was observed only after cystectomy. No swelling or pain
was recorded in the treated regions until the last clinical examination.
Moreover, no abnormal findings, such as scar tissues or loss of
surrounding teeth, were observed in any case.
Discussion
We have previously applied OCP/Col in humans for the first time
[23]. Subsequently, another study group has reported the clinical
application of OCP granules [26]. Both these studies have confirmed
bone regeneration using OCP materials. In previous in vivo studies, the
use of OCP resulted in improved bone regeneration through complex
formation with collagen [18]; therefore, OCP/Col was selected as a
bone substitute material in this study. OCP/Col was implanted in the
bone defects of 10 patients and no abnormal findings were observed
until 1 year after implantation [25]. According to the protocols
followed in this clinical research, the follow-up was performed only
up to 1 year, and observation after this period was performed in only
one case of dental implantation. Owing to the regular follow-up of
the dental implant status, continuous confirmation of the OCP/Col
implantation region was possible for case 1. During the 7-year followup
period, no infection at the treated region or formation of neoplastic
lesions was observed and no adverse effects on the surrounding teeth,
such as mobility or loss, were confirmed; thus, it can be considered that
OCP/Col was safely absorbed and replaced in the body. Furthermore,
the dental implant inserted into the OCP/Col implantation region
were stable, and the newly formed bone using OCP/Col demonstrated
an affinity to the dental implant. A recent study has demonstrated
similar stability with the simultaneous implantation of OCP/Col and
dental implant body in vivo [30]. In case 2, long-term progress could
be confirmed by examination because the patient visited the hospital
due to an injury at the site treated in this clinical research. This patient
was not continuously followed up thereafter. However, no abnormal
findings were observed in X-ray and intraoral examination at 6 years
and 6 months after OCP/Col implantation, and stable results were
confirmed in the region treated using OCP/Col. Moreover, the newly
formed bone using OCP/Col did not mutate and remained stable for
a long time in this patient.
Reportedly, other materials, such as hydroxyapatite, are
not completely absorbed in the body and cause infection [4,5].
Several studies have been performed or improving absorption
and osteo conductivity by combining with other materials [31,32],
incorporating other factors [33,34] or using mesenchymal stem cells
[35,36]. However, OCP can irreversibly convert to bone-like apatite
in vitro [8], and OCP/Col can form a new bone without remaining
the phase of OCP because complete conversion from OCP to bonelike
apatite has been confirmed directly using a micro-beam X-ray
diffraction analysis in situ for the corresponding area of the implanted
OCP in both bone and subcutaneous tissues [7,27]. In this study, the
boundary between newly formed bone and the surrounding bone
remained unclear until the last X-ray examination. Because OCP/Col
does not remain as a foreign matter, the newly formed bone using
OCP/Col may be stable.
Bone regeneration using OCP/Col or OCP granules has been
reported in the clinical setting, but the long-term progress remains
unknown. Even though we analyzed only two cases in this study,
we could confirm, for the first time, the long-term treatment course
using OCP/Col and indicate its safety and stability. Recently, a
clinical trial of OCP/Col has been performed as a prospective, multicentre,
single-arm study. In this clinical trial, OCP/Col has been used
in patients undergoing dental implantation and in those with cleft
palate. In this study, however, the data have been collected over a
short period until the confirmation of bone regeneration although
these cases generally require long-term management. Therefore, in
the future, long-term courses of several cases need to be observed for
validating of the stability of OCP/Col.
Conclusion
This present study confirmed for the first time the long-term stability and safety of newly formed bone using octacalcium phosphate collagen composite in two cases.
References
- LeGeros RZ. Properties of osteoconductive biomaterials: calcium phosphates. Clin Orthop Relat Res. 2002(395):81-98.
- Jo SH, Kim YK, Choi YH. Histological Evaluation of the Healing Process of Various Bone Graft Materials after Engraftment into the Human Body. Materials (Basel). 2018;11(5).
- Okada T, Kanai T, Tachikawa N, Munakata M, Kasugai S. Histological and Histomorphometrical Determination of the Biogradation of beta-Tricalcium Phosphate Granules in Maxillary Sinus Floor Augmentation: A Prospective Observational Study. Implant Dent. 2017;26(2):275-83.
- Sato S, Yoshinuma N, Kishida O, Fujisaki Y, Ito K. Removal of infected non-resorbable hydroxyapatite graft material in recurrent periodontitis: a report of two cases. J oral sci. 2009;51(4):659-63.
- Dold A, Perretta D, Youm T. Osteomyelitis after Calcium Phosphate Subchondroplasty A Case Report. Bull Hosp Jt Dis (2013). 2017;75(4):282-5.
- Lichte P, Pape HC, Pufe T, Kobbe P, Fischer H. Scaffolds for bone healing: concepts, materials and evidence. Injury. 2011;42(6):569-73.
- Suzuki O, Nakamura M, Miyasaka Y, Kagayama M, Sakurai M. Bone formation on synthetic precursors of hydroxyapatite. Tohoku J Exp Med. 1991;164(1):37-50.
- Suzuki O, Kamakura S, Katagiri T, Nakamura M, Zhao B, Honda Y, et al. Bone formation enhanced by implanted octacalcium phosphate involving conversion into Ca-deficient hydroxyapatite. Biomaterials. 2006;27(13):2671-81.
- Brown W SJ, Lehr J, Frazier A. Crystallographic and chemical relations between octacalcium phosphate and hydroxyapatite. Nature. 1962;196:1050-5.
- Bodier-Houlle P, Steuer P, Voegel JC, Cuisinier FJ. First experimental evidence for human dentine crystal formation involving conversion of octacalcium phosphate to hydroxyapatite. Acta Crystallogr D Biol Crystallogr. 1998;54(Pt 6 Pt 2):1377-81.
- Tohda H, Yamada M, Yamaguchi Y, Yanagisawa T. High-resolution electron microscopical observations of initial enamel crystals. Journal of Electron Microscopy. 1997;46(1):97-101.
- Crane NJ, Popescu V, Morris MD, Steenhuis P, Ignelzi MA Jr. Raman spectroscopic evidence for octacalcium phosphate and other transient mineral species deposited during intramembranous mineralization. Bone. 2006;39(3):434-42.
- Kamakura S, Sasano Y, Homma H, Suzuki O, Kagayama M, Motegi K. Implantation of octacalcium phosphate (OCP) in rat skull defects enhances bone repair. J Dent Res. 1999;78(11):1682-7.
- Imaizumi H, Sakurai M, Kashimoto O, Kikawa T, Suzuki O. Comparative study on osteo conductivity by synthetic octacalcium phosphate and sintered hydroxyapatite in rabbit bone marrow. Calcif Tissue Int. 2006;78(1):45-54.
- Kamakura S, Sasano Y, Shimizu T, Hatori K, Suzuki O, Kagayama M, et al. Implanted octacalcium phosphate is more resorbable than beta-tricalcium phosphate and hydroxyapatite. J Biomed Mater Res. 2002;59(1):29-34.
- Anada T, Kumagai T, Honda Y, Masuda T, Kamijo R, Kamakura S, et al. Dose-dependent osteogenic effect of octacalcium phosphate on mouse bone marrow stromal cells. Tissue engineering Part A. 2008;14(6):965-78.
- Kawai T, Anada T, Honda Y, Kamakura S, Matsui A, Matsui K, et al. Analysis of the osteoblastic cell differentiation by synthetic octacalcium phosphate (OCP) compared with commercially available beta-TCP ceramic. Jpn J Oral Maxillofac Surg. 2010;56(1):2-8.
- Kamakura S, Sasaki K, Honda Y, Anada T, Suzuki O. Octacalcium phosphate combined with collagen orthotopically enhances bone regeneration. J Biomed Mater Res B Appl Biomater. 2006;79(2):210-7.
- Iibuchi S, Matsui K, Kawai T, Sasaki K, Suzuki O, Kamakura S, et al. Octacalcium phosphate (OCP) collagen composites enhance bone healing in a dog tooth extraction socket model. Int J Oral Maxillofac Surg. 2010;39(2):161-8.
- Kawai T, Matsui K, Iibuchi S, Anada T, Honda Y, Sasaki K, et al. Reconstruction of critical-sized bone defect in dog skull by octacalcium phosphate combined with collagen. Clin Implant Dent Relat Res. 2011;13(2):112-23.
- Matsui K, Matsui A, Handa T, Kawai T, Suzuki O, Kamakura S, et al. Bone regeneration by octacalcium phosphate collagen composites in a dog alveolar cleft model. Int J Oral Maxillofac Surg. 2010;39(12):1218-25.
- Miura K, Matsui K, Kawai T, Kato Y, Matsui A, Suzuki O, et al. Octacalcium phosphate collagen composites with titanium mesh facilitate alveolar augmentation in canine mandibular bone defects. Int J Oral Maxillofac Surg. 2012;41(9):1161-9.
- Kawai T, Echigo S, Matsui K, Tanuma Y, Takahashi T, Suzuki O, et al. First clinical application of octacalcium phosphate collagen composite in human bone defect. Tissue Eng Part A. 2014;20(7-8):1336-41.
- Kawai T, Suzuki O, Matsui K, Tanuma Y, Takahashi T, Kamakura S. Octacalcium phosphate collagen composite facilitates bone regeneration of large mandibular bone defect in humans. J Tissue Eng Regen Med. 2017; 11(5):1641-7.
- Kawai T, Tanuma Y, Matsui K, Suzuki O, Takahashi T, Kamakura S. Clinical safety and efficacy of implantation of octacalcium phosphate collagen composites in tooth extraction sockets and cyst holes. J Tissue Eng. 2016;7:2041731416670770.
- Komlev VS, Barinov SM, Bozo II, Deev RV, Eremin II, Fedotov AY, et al. Bioceramics composed of octacalcium phosphate demonstrate enhanced biological behavior. ACS Appl Mater Interfaces. 2014;6(19):16610-20.
- Suzuki O, Nakamura M, Miyasaka Y, Kagayama M, Sakurai M. Maclura pomifera agglutinin-binding glycoconjugates on converted apatite from synthetic octacalcium phosphate implanted into subperiosteal region of mouse calvaria. Bone Miner. 1993;20(2):151-66.
- Fowler BO, Moreno EC, Brown WE. Infra-red spectra of hydroxyapatite, octacalcium phosphate and pyrolysed octacalcium phosphate. Arch Oral Biol. 1966;11(5):477-92.
- Nelson DG, McLean JD. High-resolution electron microscopy of octacalcium phosphate and its hydrolysis products. Calcified tissue international. 1984;36(2):219-32.
- Kawai T, Matsui K, Ezoe Y, Kajii F, Suzuki O, Takahashi T, et al. Efficacy of Octacalcium Phosphate Collagen Composite for Titanium Dental Implants in Dogs. Materials. 2018;11(2):229.
- Castilho M, Moseke C, Ewald A, Gbureck U, Groll J, Pires I, et al. Direct 3D powder printing of biphasic calcium phosphate scaffolds for substitution of complex bone defects. Biofabrication. 2014;6(1):015006.
- Rh Owen G, Dard M, Larjava H. Hydoxyapatite/beta-tricalcium phosphate biphasic ceramics as regenerative material for the repair of complex bone defects. J Biomed Mater Res B Appl Biomater. 2018; 106(6):2493-2512.
- Lee JH, Ryu MY, Baek HR, Lee HK, Seo JH, Lee KM, et al. The effects of recombinant human bone morphogenetic protein-2-loaded tricalcium phosphate microsphere-hydrogel composite on the osseointegration of dental implants in minipigs. Artif Organs. 2014;38(2):149-58.
- Silva L, Porto GG, Andrade ESS, Laureano Filho JR. Demineralized bone matrix and calcium-phosphate cement in bone regeneration in rats. Acta cirurgica brasileira. 2018;33(4):354-61.
- Zou D, Guo L, Lu J, Zhang X, Wei J, Liu C, et al. Engineering of bone using porous calcium phosphate cement and bone marrow stromal cells for maxillary sinus augmentation with simultaneous implant placement in goats. Tissue Eng Part A. 2012;18(13-14):1464-78.
- Wang P, Zhao L, Chen W, Liu X, Weir MD, Xu HH. Stem Cells and Calcium Phosphate Cement Scaffolds for Bone Regeneration. J Dent Res. 2014;93(7):618-25.