Editorial
Asymptomatic Pancreatic Masses: An Overview
Marco Chiarelli2, Matilde De Simone1, Sabina Terragn2 and Ugo Cioffi1*
1Department of Surgery, University of Milan, Italy
2Department of Surgery, Ospedale Alessandro Manzoni, Italy
*Corresponding author: Ugo Cioffi, Department of Surgery, University of Milan, Via F. Sforza 35, 20122 Milano, Italy
Published: 29 Mar, 2018
Cite this article as: Chiarelli M, De Simone M, Terragn
S, Cioffi U. Asymptomatic Pancreatic
Masses: An Overview. Clin Surg. 2018;
3: 1947.
Keywords
Pancreatic masses; Surgery; Diagnosis
Editorial
Nowadays pancreatic masses are more and more frequently detected. In the last decades the
advances in radiological techniques and the wide diffusion of cross-sectional diagnostic imaging
led to an increasingly number of asymptomatic pancreatic masses detection. In few years the use
of Computed Tomography (CT) imaging has increased noticeably. A significant proportion of this
boost is related to its role in the diagnosis, particularly in acute care environment: the use of CT in
the emergency department has increase by 4 fold in the last decade [1].
Currently intravenous contrast-enhanced multi detector CT is considered the fundamental
technique for the detection and the staging of pancreatic tumor allowing the definition of size,
localization, texture and vascularization of the mass. For pancreatic imaging several authors suggest
a high-resolution biphasic CT scan with a late arterial phase (performed 20-40 seconds after contrast
injection) and a portal venous phase. Indeed the late arterial phase, named also pancreatic phase,
seems to allow a better definition of the pancreatic gland [2].
Unlike CT scan, there is a wide variability among Magnetic Resonance Imaging (MRI) protocols
for pancreatic lesions. A complete pancreatic MRI study includes pre- and post-Gadolinium T1
weighted sequences, T2-weightened sequences with fat suppression, and Magnetic Resonance
Cholagiopancreatography (MRCP). The advantages of this cross-sectional technique are optimal
soft tissue-contrast resolution, accurate assessment of the internal structure of cystic lesions, and
detailed study of pancreatic ducts [3].
The use of these imaging techniques allows an excellent characterization of the asymptomatic
masses, the so called “pancreatic incidentalomas”, and an ever more frequent discovery of unusual
pancreatic tumors [4,5].
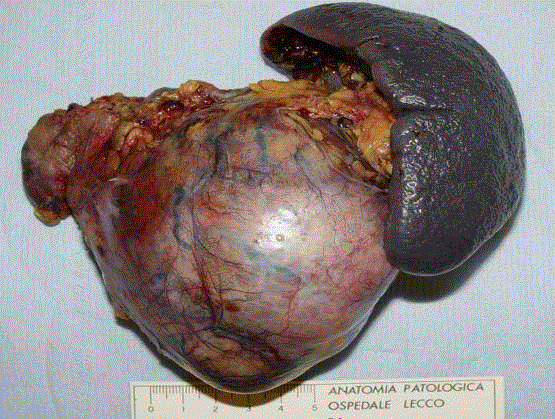
Asymptomatic pancreatic cystic masses frequently present a benign or premalignant nature
while solid masses are usually considered malignant tumors (Figure 1).
In the preoperative work-up of a cystic lesion is essential to differentiate a cystic neoplasm from
an inflammatory pseudocyst. A history of acute pancreatitis, upper abdominal pain or blunt trauma
can help in the differential diagnosis. Endoscopic Ultrasonography (EUS) with cyst fluid analysis
may support the diagnosis of pseudocysts [6].
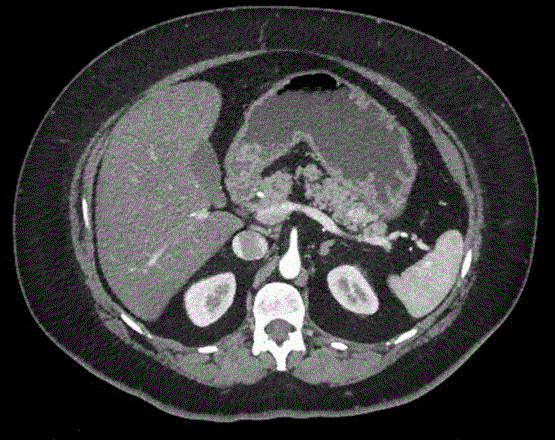
Serous cystic neoplasm (SNC) appears as a cystic lesion consisting on several thin-walled spaces
filled by serous fluid, frequently arranged around a central dense tissue. SNC is usually considered a
benign lesion due to a very low rate of malignant progression (about 3%). Serous cystadenoma may
occur as oligocystic variant in 7% to 20% of patients and this form is difficult to differentiate from
mucinous neoplasms [7]. Indeed the microcystic variant of SNC can mimic a solid mass at imaging
for the presence of numerous inner fibrous septa (Figure 2). A conservative approach is appropriate
for these pancreatic lesions; surgery should be considered in case of large tumors (size >4 cm) or
when preoperative exams are not conclusive [8].
Mucin-producing cystic pancreatic tumors are frequently incidental. Mucinous Cystic
Neoplasms (MCN) are thick-walled septated macrocystic lesions more frequently located in
body-tail region of the pancreas and almost exclusively affect middle-aged women [9]. MCNs are
characterized by the absence of communication with the ductal system and the presence of columnar
mucin-producing epithelium supported by ovarian-type stroma [10]. Intraductal Papillary
Mucinous Neoplasms (IPMN) is intraductal tumors characterized by papillary proliferation of the epithelium and mucin hypersecretion that causes a cystic dilatation of the ductal system. IPMNs often occur in the head of the pancreas,
they are more frequent in the seventh decade of life showing an equal
gender distribution [9]. The communication between the cystic
tumor and the pancreatic ducts is a specific feature of this type of
neoplasm. IPMNs are subdivided in main duct type and branch
duct type according to the site of origin. Mucin-producing cystic
pancreatic tumors are considered premalignant lesions because they
can degenerate in invasive carcinoma [9]. Peripheral calcifications,
irregular septa, intramural nodules or dilatation of the main duct
are findings frequently associated with malignant histology. Surgical
resection is mandatory for MCN and main duct type IPMN; small
branch duct type IPMN (< 3 cm) can be observed with a radiologic
follow-up [10].
Solid Pseudopapillary Neoplasm (SPPN) is a rare pancreatic
tumor affecting predominantly young women. This neoplasm is
solitary large mass (8 cm to 10 cm) consisting of a solid component
alternating cystic spaces originated by intratumoral hemorrhage.
SPPN is a slow growing neoplasm with low malignant potential and
surgical resection is considered resolutive [11]. The cystic variant
of Neuroendocrine Tumor (NET) is very rare and represent 2%
to 20% of all endocrine pancreatic neoplasms. Cystic NETs are
usually non-functioning small lesions. CT scan shows homogenous
hypoenhancing pancreatic mass, frequently surrounded by a
hyperenhancing rim. Conservative resection is recommended for
these lesions [12].
In the preoperative work-up of a solid pancreatic mass it is
essential to exclude focal pancreatitis and intraparenchymal spleen,
two benign lesions that can simulate solid neoplasm of the body and
tail [13].
Pancreatic Ductal Adenocarcinoma (PDAC) and pancreatic
NET are the most frequent histotypes detected in solid pancreatic
incidentalomas. NETs represent 2% of all pancreatic malignancies
and their incidence has increased 2 to 3 fold in the past decades
[14]. The majority of endocrine neoplasms are non-functional with
a more frequent location in the tail of the gland [15]. They typically
appear as hyperintense pancreatic masses at CT scan. EUS can be
useful in the detection and characterization of pancreatic NETs.
Preoperative dosage of serum chromogranine A and somatostatin
receptor scintigraphy can support NET diagnosis [14]. Recently
a non-operative management has been proposed for incidentally
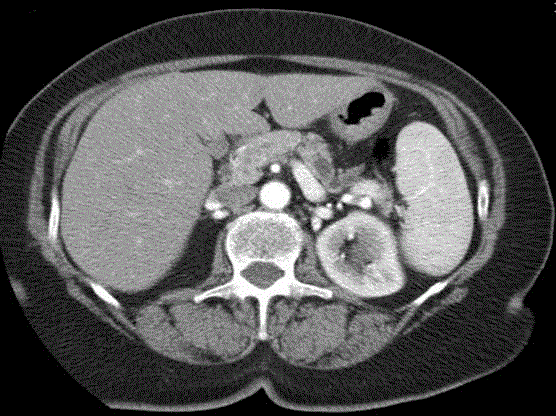
discovered small non-functioning pancreatic NETs [16]. However
large series demonstrated that even endocrine tumors with size < 2
cm may develop local or distant disease progression and the surgical
resection is associated with a better survival (Figure 3). Consequently
the majority of the authors suggest that these pancreatic lesions
should be resected [15].
Acinar Cell Carcinoma (ACC) is a rare pancreatic malignancy
(2% of all pancreatic tumors) affecting predominantly patients in the
sixth decade of life. ACC presents as a bulky mass or multinodular
lesion. The tumor is composed by uniform malignant cells arranged
in small glandular units secreting low quantities of exocrine enzymes.
Prognosis of ACC seems to be slightly better than PDAC although
5-year survival rate is low [17].
A relevant proportion of incidentally discovered solid pancreatic
lesions are PDACs. In large series of pancreatic incidentalomas
malignant lesions were very frequent (30% to 35%) [18,19]. Even
if PDAC is overall more frequent in the head of the gland (about
80%), in asymptomatic patients it seems to prefer the distal pancreas
[13,20]. PDACs discovered incidentally are detected at earlier stages
than symptomatic lesions; it is debated whether early diagnosis means
a better prognosis [18].
Surgical resection is considered the standard treatment of
asymptomatic pancreatic solid lesions. Conversely, the management
of cystic masses is complex due to the high rate of benign lesions: size
>3 cm, main duct dilatation >10 mm, presence of mural nodules and
cytology suspicious for malignancy are indications for surgery [13].
Furthermore the detection of an asymptomatic complex mass with
solid and cystic components imposes a surgical resection.
Figure 1
Figure 2
Figure 3
Conclusion
In conclusion the wide diffusion of cross-sectional radiologic techniques in the last decades has allowed the diagnosis of a large variety of pancreatic asymptomatic lesions. In many cases these incidental masses are rare tumors with more favorable prognosis than PDAC. Nevertheless, the suspicion of malignant lesions must be high and surgical exploration still remains necessary in the majority of cases.
References
- Kocher KE, Meuer WJ, Fazel R, Scott PA, Krumholz HM, Nallamothu BK. National trends in use of computed tomography in the emergency department. Ann Emerg Med. 2011;58(5):452-62.
- Low G, Panu A, Millo N, Leen E. Multimodality imaging of neoplastic and non neoplastic solid lesions of the pancreas. Radiographics. 2011;31(4):993-1015.
- Raman SP, Chen Y, Fishman EK. Cross-sectional imaging and the role of positron emission tomography in pancreatic cancer evaluation. Semin Oncol. 2015;42(1):40-58.
- Chiarelli M, Gerosa M, Tagliabue F,Fumagalli L, Guttadauro A, Gabrielli F, et al. Left-sided pancreatic incidentalomas treated with laparoscopic approach: a report of 20 cases. World J Surg Oncol. 2016;14(1):204.
- Sheehan M, Latona C, Aranha G, Pickleman J. The incriseasing problem of unusual pancreatic tumors. Arch Surg. 2000;135:644-8.
- Buerke B, Domagk D, Heindel W, Wessling J. Diagnostic and radiological management of cystic pancreatic lesions: important features for radiologists. Clin Radiol. 2012;67(8):727-37.
- Tseng JF, Warshaw AL, Sahani D, Lauwers GY, Rattner DW, Fernandez-del Castillo C. Serous cystoadenoma of the pancreas. Tumor growth rates and recommendations for treatment. Ann surg. 2005;242(3):413-9.
- Sakorafas GH, Smyrniotis V, Reid-Lombardo KM, Sarr MG. Primary pancreatic cystic neoplasms revisited. Part I: serous cystic neoplasms. Surg Oncol. 2011;20(2):e84-92.
- Adsay NV. Cystic lesions of the pancreas. Mod Pathol. 2007;20:71-93.
- Tanaka M, Fernandez-del Castillo C, Adsay V, Chari S, Falconi M, Jang JY, et al. International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology. 2012;12(3):183-97.
- Martin RC, Klimstra DS, Brennana MF, Conlon KC. Solid-pseudopapillary tumor of the pancreas: a surgical enigma ? Ann Surg Oncol. 2002;9(1):35-40.
- Baker MS, Knuth JL, deWitt J, LeBlanc J, Cramer H, Howard TJ, et al. Pancreatic cystic neuroendocrine tumors: preoperative diagnosis with endoscopic ultrasound and fine-needle immunocytology. J Gastrointest Surg 2008;12(3):450-6.
- Sachs T, Pratt WB, Callery MP, Vollmer CM. The incidental asymptomatic pancreatic lesion: nuisance or threat ?. J Gastrointest Surg. 2009;13(3):405-15.
- Metz DC, Jensen RT. Gastrointestinal neuroendocrine tumors: pancreatic endocrine tumors. Gastroenterology. 2008;135(5):1469-92.
- Haynes AB, Deshpande V, Ingkakul T, Vagefi PA, Szymonifka J, Thayer SP, et al. Implications of incidentally discovered, nonfunctioning pancreatic endocrine tumors. Short-term and long-term patient outcomes. Arch Surg. 2011;146(5):534-8.
- Lee LC, Grant CS, Salomao DR, Fletcher JG, Takahashi N, Fidler JL, et al. Small, nonfunctioning, asymptomatic pancreatic neuroendocrine tumors (PNETs): role for nonoperative management. Surgery. 2012;152(6):965-74.
- Holen KD, Klimstra DS, Hummer A, Gonen M, Conlon K, Brennan M, et al. Clinical characteristics and outcomes from an institutional series of acinar cell carcinoma of the pancreas and related tumors. J Clin Oncol. 2002;20(4):4673-8.
- Lahat G, Ben Haim M, Nachmany I, Sever R, Blachar A, Nakache R, et al. Pancreatic incidentalomas: high rate of potentially malignant tumors. J Am Coll Surg. 2009;209(3):313-9.
- Bruzoni M, Johnston E, Sasson AR. Pancreatic incidentalomas: clinical and pathologic spectrum. Am J Surg. 2008;195(3):329-32.
- Sharma C, Eltawil KM, Renfrew PD, Walsh MJ, Molinari M. Advances in diagnosis, treatment and palliation of pancreatic carcinoma: 1990-2010. World J Gastroenterol. 2011;17(7):867-97.