Research Article
Preoperative Platelet to Lymphocyte Ratio Predicts Early Postoperative Relapses in Patients with Stage II Colon Cancer
Takashi Okuyama1*, Shunya Miyazaki1, Shinichi Sameshima1, Emiko Takeshita1, Takashi Mitsui1, Yuko Ono2, Tamaki Noie1 and Masatoshi Oya1
1Department of Surgery, Dokkyo Medical University, Japan
2Department of Pathology, Dokkyo Medical University, Japan
*Corresponding author: Takashi Okuyama, Department of Surgery, Dokkyo Medical University, Saitama Medical Center, Japan
Published: 11 Oct, 2018
Cite this article as: Okuyama T, Miyazaki S, Sameshima
S, Takeshita E, Mitsui T, Ono Y, et al.
Preoperative Platelet to Lymphocyte
Ratio Predicts Early Postoperative
Relapses in Patients with Stage II Colon
Cancer. Clin Surg. 2018; 3: 2152.
Abstract
Background: Its predictive utility in patients with stage II colon cancer undergoing curative resection
is yet to be fully evaluated. The purpose of this study was to evaluate its utility as a predictive maker
of preoperative PLR on postoperative relapse in patients with stage II colon cancer.
Methods: A total 154 patients with stage II colon cancer who had undergone curative resection
between 2007 to 2016 were eligible for this study. The present study retrospectively analyzed the data
of these patients from a single hospital. Receiver Operator Characteristic (ROC) curve was applied
to determine the most significant cutoff vale of PLR. Kaplan-Meier curve and Cox proportional
models were used to compare high and low PLR groups and to identify the risk factor for relapse.
Results: The median PLR was significantly higher in patients who had developed tumor recurrence
than in those who did not (p=0.04). Using receiver-operator characteristics curve analysis, the
optimal cutoff value of PLR for the discrimination between patients who had relapse and those who
did not was 198. Tumor relapses occurred significantly more frequent in patients with high PLR
than compared in those with low PLR (p=0.001). Patients with low PLR were significantly better
RFS than those with high PLR (95% CI 74.9-107.5, p<0.001). High PLR was an independent risk
factor for RFS in multivariate analysis (HR=4.32, 95% CI=1.58-11.79, p=0.004).
Conclusion: In this study, PLR was suggested as a useful predictive marker for postoperative relapse
in patients with stage II colon cancer.
Introduction
Surgical resection with curative intent is the standard treatment for patients with localized
colorectal cancer. About 20% of patients with stage II colon cancer defined by negative lymph
Nodemetastasis (N0) develop tumor relapse, although the presence of lymph node metastasis is
the most reliable risk factor for a poor prognosis [1]. Current international guidelines propose that
adjuvant chemotherapy be considered for patients with high-risk stage II colon cancer, because
postoperative relapses may result from cancer cells remaining somewhere in the patient’s body
after surgical resection [2,3]. In contrast, only 36% of high-risk patients with stage II colon cancer
received adjuvant chemotherapy, because a significant survival benefit has not been demonstrated
in such patients [4].
Previous studies have shown that the interactions between tumor and host-derived
microenvironments, including inflammation, coagulation state, or immune response, contribute to
the development and progression of cancer [5,6]. Recently, to predict the survival of patients with
colorectal cancer, blood-based inflammatory parameters such as the Neutrophil to Lymphocyte
Ratio (NLR), the Platelet to Lymphocyte Ratio (PLR), the Lymphocyte to Monocyte Ratio (LMR),
C-reactive protein, and the modified Glasgow prognostic score, have been proposed as inexpensive
and widely available biomarkers that are routinely measured in daily clinical practice [7-11].
Among these inflammatory markers, platelets have been shown to play an important role in several
processes, such as carcinogenesis, angiogenesis, thrombosis, and metastasis in tumor growth
[12,13]. This study aimed to investigate the clinical impact of the PLR on postoperative relapse in
patients with stage II colon cancer after curative resection.
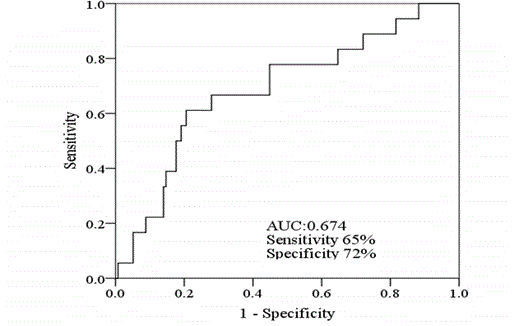
Figure 1
Material and Methods
Patients
Between April 2007 and March 2016, the medical records of
154 patients with stage II colon adenocarcinoma at Saitama Medical
Center, Dokkyo Medical University were retrospectively reviewed.
Thirty-six patients who had received adjuvant chemotherapy and 3
patients who had received neoadjuvant chemotherapy were excluded.
Nine patients with synchronous or metachronous, multiple advanced
cancers, 1 patient with ulcerative colitis, 1 patient with liver cirrhosis,
and 1 patient with malignant lymphoma were also excluded. The
other exclusion criterion was that the medical records did not include
the complete blood cell count, positive surgical resection margin,
or history of familial CRC. This study was approved by the Ethics
Committee of Saitama Medical Center, Dokkyo Medical University
No.1809.
Surgical procedures and follow-up
All patients underwent curative resection including the primary
tumor and enbloc dissection of regional lymph nodes up to the
root of the main feeding artery (total mesocolic excision). The
pathological findings were recorded by pathologists of Saitama
Medical Center, Dokkyo Medical University. Cancer staging was
according to the American Joint Committee on Cancer (AJCC)
staging manual [14]. Postoperative surveillance including medical
examinations and laboratory tests was performed every 3 months.
Computed Tomography (CT) of the chest, abdomen, and pelvis was
performed every 6 months. Colonoscopy for luminal surveillance
was performed within 1 year after surgery and annually thereafter
until no abnormality was recorded. If no abnormality was detected
on surveillance colonoscopy, the subsequent colonoscopy was
performed within 3 years.
Assessment of the PLR and statistical analysis
In this study, the primary end point was Relapse-Free Survival
(RFS) calculated from the date of surgery to the date of the first
observation of tumor relapse. The correlations between the PLR and
clinicopathological characteristics were also explored. Peripheral
blood obtained within 3 weeks prior to the surgery was used. The
PLR was calculated by dividing the absolute platelet count by the
absolute lymphocyte count. The median PLR value of each group
with or without relapse was analyzed by the Mann-Whitney U test.
The optimal cut-off value of the PLR for relapse was calculated using
receiver operator characteristic (ROC) curve analysis. Furthermore,
after patients were divided into a high-PLR group and a low-PLR group
by the cut-off value, differences in clinicopathological characteristics
between the two groups were examined by the χ2 test or Fisher’s
exact test. The RFS curves were compared using the Kaplan-Meier
method, and differences were evaluated by the log-rank test. Uniand
multivariate analyses to identify significant factors for RFS were
performed using Cox proportional hazard regression models. All
analyses were performed using the SPSS statistical software package,
version 24 (IBM Japan Ltd., Tokyo, Japan). A 2-sided P value of <0.05
was considered significant.
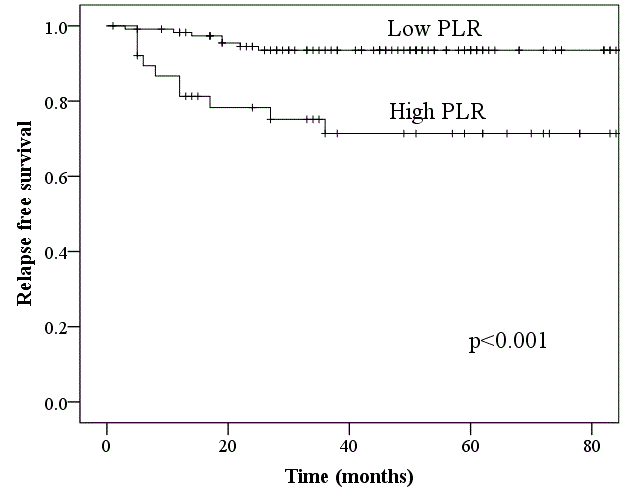
Figure 2
Table 1
Table 2
Table 3
Results
The patients’ median age at the time of surgical resection was 72
years (range 29 - 89 years). The median follow-up period for RFS was
50 months (range 3 - 123 months). The baseline clinicopathological
characteristics of the 154 patients are shown in Table 1. Eighteen
(12%) of 154 patients developed tumor relapses, including lung (4
patients), liver (6 patients), peritoneal dissemination (3 patients),
local recurrence (2 patients), and metastasis to the para-aortic lymph
nodes (3 patients). Fifteen patients (15/154, 9.7%) died of any causes
within the follow-up period, and ten of these patients (10/15, 67%)
died of causes other than primary colon cancer.
The median PLR was 146 (range 32 - 1830). A significant
difference in PLR was found between patients who developed relapse
(median 208.6, range 85.0-483.3) and those who did not develop
relapse (median 141.8, range 32.3-1830.2) (p=0.04). Using ROC
analysis, the optimal cut-off value of the PLR for predicting relapse
was 198. The largest area under the curve (AUC) for RFS was 0.688
(95% CI 0.555-0.821, p=0.01, Figure 1). When the cut-off was set to
198, the sensitivity and specificity for relapse were 65% and 72%,
respectively. The tumor recurred in 11 (28%) of 39 patients with a
high PLR and in 7 (6.1%) of 115 patients with a low PLR (p=0.001).
On univariate proportional hazard analysis performed for PLR cutoff
values of 50, 100, 200, 250, and 300, only the cut-off value of
200 showed a significant difference in RFS between patients with a
high PLR and those with a low PLR (HR=5.08, 95% CI=1.93-13.36,
p=0.001, Table 2).
Univariate and multivariate analyses to identify factors
significantly related to RFS were performed using Cox proportional
hazard models (Table 3). On univariate analyses, a serum CEA level
higher than 5 ng/ml and a high PLR were associated with shorter RFS
(HR=2.25, 95% CI=1.38-3.64, p=0.004 and HR=2.67, 95% CI=1.028-
6.917, p=0.04, respectively). However, on multivariate analysis, a high
PLR was the only factor significantly associated with shorter RFS
(HR=4.32, 95% CI=1.58-11.79, p=0.004). Patients with PLR greater
than 198 had a significantly increased risk of relapse compared to
those with a lower PLR. The 4-year RFS was 88% for the entire study
population. Figure 2 shows the Kaplan-Meier survival curves for RFS
in patients according to the PLR value. Patients with a low PLR had
a significantly longer RFS than those with a high PLR (95% CI 74.9-
107.5, p<0.001, Figure 2). The 4-year RFS rates were 94% and 71% for
patients with a low PLR and those with a high PLR, respectively. The
relationships between the PLR and clinicopathological characteristics
are shown in Table 4. Patients with a higher PLR had a higher serum
CEA level, larger tumor size, lower hemoglobin value, and more
frequent venous invasion than those with a lower PLR (p=0.005,
p=0.03, p=0.001, and p=0.04, respectively) (Table 5). None of the
other clinicopathological characteristics was associated with a high
PLR.
Discussion
The aim of the present study was to explore its usefulness of PLR
for the prediction of relapse in postoperative patients with stage
II colon cancer. The median PLR value of patients who developed
relapse was significantly higher than that of those who did not.
Among clinicopathological characteristics, only high PLR showed
a significant correlation with postoperative relapse. The Kaplan-
Meier analysis revealed that patients with high PLR had significantly
shorter RFS in comparison to those with low PLR. Multivariate
analysis demonstrated that high PLR was independently predictive
indicator of postoperative relapse in patients with stage II colon
cancer. In addition, higher PLR was significantly associated with
clinicopathological parameters suggesting malignant potential of
colon cancer, such as higher serum CEA value, larger tumor size,
and more frequent venous invasion [15,16]. Since previous trials
have failed to show the benefit of adjuvant chemotherapy on patients
with stage II colon cancer, most of them do not receive adjuvant
chemotherapy [1,4,17,18]. However, the current international
guidelines suggest that adjuvant chemotherapy should be considered
for high-risk stage II colon cancer patients. As risk factor in stage II
colon cancers, poorly differentiated histology, lymphovascular or
perineural invasion, bowel obstruction, localized perforation, positive
or incompetent resection margin, insufficient searched lymph node
(<12 lymph nodes) are included [3,4]. Recent approaches using
genetic and molecular markers including Microsatellite Instability
(MSI), may provide better information for relapse or prognosis of
cancers [19,20]. Sargent et al. [21] have reported that patients with
MSI-H have a better prognosis, while fluorouracil-based adjuvant
chemotherapy is not beneficial in patients with stage II colon cancer
[21]. PLR is an inexpensive and easily available parameter compared
to molecular and genetic markers. It has already been indicated that
high PLR is associated with the prognosis of several solid cancers
including ovarian, pancreatic, gastric, esophageal, and colorectal
cancers [8,22,25]. In the previous reports, however, there are few
studies limited to stage II colon cancer. Ozawa et al. reported that
in 234 patients with stage II colorectal cancer, patients with low PLR
had significantly better prognosis regarding DFS and cancer specific
survival than in those with high PLR [26]. They also reported that the
PLR was an independent prognostic marker in multivariate analysis.
Kim et al. reported that PLR was a prognostic indicator of both OS
and DFS in patients with stage III and IV colorectal cancer [27]. In
their study, however, PLR was not correlated with either OS or DFS
in patients with stage I or II colorectal cancer. The difference in these
results may be influence by differences in the selection criteria of the
subject and whether the OS and DFS were calculated using each the
cutoff value. Gu et al. [28] have indicated that the effect of platelets
on cancer is different between rectal cancer and colon cancer [28].
In the carcinogenesis of rectal cancer, p53 mutation involves more
commonly than in that of colon cancer, and the activation of plateletderived
growth factor receptor alpha pathway is accompanied by the
suppression of p53 [13,29,30]. It has therefore been considered that
the elevated platelet number may act as tumor suppressor rather than
promotor. Therefore, we selected only patients with stage II colon
cancer for investigating a homogeneous group of patients in the
present study.
Chronic inflammation is strongly involved in carcinogenesis.
This fact has been established by the observations in patients with
inflammatory bowel disease. Recent experimental and clinical studies
suggest several potential explanations for the association between
the inflammatory and cancer [5]. Platelets promote cancer growth
by increasing angiogenesis through the production of vascular
endothelial growth factor, which can activate the invasiveness of tumor
cell by enhancing the formation of tumor stroma and supporting
the stable adhesion of tumor cells to the endothelium [31]. Labelle
et al. proved that platelet-derived signals promote the formation of
early metastatic niches through chemokines including CXCR5 and
CXCR7 [32]. On the contrary, lymphocytes induce cytotoxic cell
death of the host and are indispensable as antitumor agents which
promote tumor cell apoptosis and suppress tumor metastasis [33,34].
Hence, the combination of these blood parameters appears the state
of equilibrium between tumor-promoting and host-immune activity.
Previous studies have proposed various cut-off values of PLR for
OS, RFS, DFS, or time to recurrence (TTR) [8,26,27,35]. There is no
agreement on the optimal cut-off value, because the selection criteria
of patients in each study differ from each other. Diseases such as
coronary artery diseases, kidney diseases, liver diseases, inflammatory
disease and infectious diseases, antiplatelet agents, medication for
hyperlipidemia, chemotherapy and radiotherapy may affect PLR [36].
Since these factors may influence the prognostic significance of PLR,
we excluded patients complicated with ulcerative colitis, cirrhosis of
the liver, malignant lymphoma, and patients who received neo- or
adjuvant chemotherapy.
Several previous studies have explored OS as an endpoint.
However, recent advance in the treatment for metastatic and recurrent
colorectal cancer appears to strongly affect the outcome. In the
present study, the patients underwent surgical resection between 2007
and 2016. In the last ten years, triplet chemotherapy combined with
molecular targeted therapy, late-line chemotherapy, and salvage or
conversion surgery have prolonged survival of patients with recurrent
colorectal cancer. In the present study, postoperative follow-up with
an interval of 3 months has been carried out in majority of patients.
We therefore selected RFS rather than OS or cancer specific survival
as the endpoint of the present study. Actually, we also tried to obtain
the cutoff value of OS using the same method as to obtain cutoff
value for RFS, but only marginally significant result was obtained
(p=0.053, data not shown). This study has several limitations such its
retrospective design and small sample size. In addition, neither MSI
nor MMR, which are recommended for predicting the prognosis of
patients and the effect of adjuvant chemotherapy by NCCN guideline,
have been not examined in the present study. On the contrast, the
strength of this study is that the patients were relatively homogenous
compared to those in the previous studies. In conclusion, the present
study shows that preoperative high-PLR is a useful as a predictive
indicator of relapse in patients with stage II colon cancer who
underwent curative surgery. Although PLR is a convenient and
inexpensive marker, it may reflect malignant potential of colon
cancer, not evaluable from other clinicopathological findings. PLR
therefore may be one of useful clinical indicator for the application of
adjuvant chemotherapy for stage II colon cancer.
Table 4
Table 5
References
- Gray R, Barnwell J, McConkey C, Hills RK, Williams NS, Kerr DJ. Adjuvant chemotherapy versus observation in patients with colorectal cancer: a randomized study. Lancet. 2007;370(9604):2020-9.
- National Comprehensive Cancer Network: NCCN Clinical Practice Guidelines in Oncology, Colon Cancer v3. 2018.
- Van Cutsem E, Oliveria J; ESMO Guidelines Working Group. Primary colon cancer: ESMO clinical recommendations for diagnosis, adjuvant treatment and follow-up. Ann Oncol. 2009;20(suppl 4):49-50.
- Chaqpar R, Xing Y, Chiang YJ, Feiq BW, Chianq GJ, You YN, et al. Adherence to stage-specific treatment guidelines for patients with colon cancer. J Clin Oncol. 2012;30(9):972-9.
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436-44.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646-74.
- Malietzis G, Giacometti M, Askari A, Nachiappan S, Kennedy RH, Faiz OD, et al. A preoperative neutrophil to lymphocyte ratio of 3 predicts disease-free survival after curative elective colorectal cancer surgery. Ann Surg. 2014;260(2):287-92.
- You J, Zhu GQ, Xie L, liu XW, Shi L, Wng OC, et al. Preoperative platelet to lymphocyte ratio is a valuable prognostic biomarker in patients with colorectal cancer. Oncotarget. 2016;7(18):25516-27.
- Shibutani M, Maeda K, Nagahara H, Iseki Y, Ikeya T, Hirakawa K. Prognostic significance of the preoperative lymphocyte-to-monocyte ratio in patients with colorectal cancer. Oncol Lett. 2017;13(2):1000-1006.
- Pathak S, Nunes QM, Daniels IR, Smart NJ. C-reactive protein useful in prognostication for colorectal cancer? A systematic review. Colorectal Dis. 2014;16(10):769-76.
- Guthrie GJ, Roxburgh CS, Farhan-Alanie OM, Horgan PG, McMillan DC. Comparison of the prognostic value of longitudinal measurements of systemic inflammation in patients undergoing curative resection of colorectal cancer. Br J Cancer. 2013;109(3):24-8.
- Neiswandt B, Aktas B, Moers A, Sachs UJ. Platelets in athero-thrombosis: Lessons from mouse models. J Thromb Haemost. 2005;3(8):1725-36.
- Lei H, Velez, Kazlauskas A. Pathological signaling via platelet-derived growth factor receptor α involves chronic activation of Akt and suppression on p53. Mol Cell Biol. 2011;31(9):1788-99.
- Edge SB, Byrd DR, Compton CC. American Joint Committee on Cancer (AJCC) Cancer Staging Manual. 7th ed. 2010.
- Armi R, Bordeianou LG, Sylla P, Berger DL. Preoperative carcinoembryonic antigen as an outcome predictor in colon cancer. J Surg Oncol. 2013;108(1):14-18.
- Saha S, Shaik M, Johnston G, Saha SK, Berbiqlia L, Hicks M, et al. Tumor size predicts long-term survival in colon cancer: an analysis of the National Cancer Data Base. Am J Surg. 2015;209(3):570-4.
- Gill S, Loprinzi CL, Sargent DJ, Thome SD, Alberts SR, Haller DG, et al. Polled analysis of Fluorouracil-based adjuvant therapy for stage II and III colon cancer: who benefits and by how much? J Clin Oncol. 2004;22(10):1797-806.
- Wilkinson NW, Yothers G, Lopa S, Costantino JP, Petrelli NJ, Wolmark N. Long-term survival results of surgery plus 5-fluorouracil and leucovorin for stage II and stage III colon cancer: pooled analysis of NSABP C-01 through C-05. A baseline from which to compare modern adjuvant trials. Ann Surg Oncol. 2010;17(4):959-66.
- Watanabe T, Wu TT, Catalano PJ, Ueki T, Satriano R, Haller DG, et al. Molecular predictors of survival after adjuvant chemotherapy for colon cancer. N Engl J Med. 2001;344(16):1196-206.
- Des Guetz G, Schischmanoff O, Nicolas P, Perret GY, Morere JF, Uzzan B. Does microsatellite instability predict the efficacy of adjuvant chemotherapy in colorectal cancer? A systematic review with meta-analysis. Eur J Cancer. 2009;45(10):1890-6.
- Sargent DJ, Marsoni S, Monges G, Thibodeau SN, Labianca R, Hamilton SR, et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J Clin Oncol. 2010;28(20):3219-26.
- Asher V, Lee J, Innamaa A, Bali A. Preoperative platelet to lymphocyte ratio as an independent prognostic marker in ovarian cancer. Clin Transl Oncol. 2011;13(7):499-503.
- Shirai Y, Shiba H, Sakamoto T, Horiuchi T, Haruki K, Fujiwara Y, et al. Preoperative platelet to lymphocyte ration predicts outcome of patients with pancreatic ductal adenocarcinoma after pancreatic resection. Surgery. 2015;158(2):360-5.
- Chen XL, Xue L, Wang W, Chen HN, Zhang WH, Liu K, et al. Prognostic significance of the combination of preoperative hemoglobin, albumin, lymphocyte and platelet in patients with gastric carcinoma: a retrospective cohort study. Oncotarget. 2015;6(38):41370-82.
- Feng JF, Huang Y, Zhao Q, Chen QX. Clinical significance of preoperative neutrophil lymphocyte ratio versus platelet lymphocyte ratio in patients with small cell carcinoma of the esophagus. Scientific World Journal. 2013;2013:504365.
- Ozawa T, Ishihara S, Nishikawa T, Tanaka T, Tanaka J, Kiyomatsu T, et al. The preoperative platelet to lymphocyte ratio is a prognostic marker in patients with stage II colorectal cancer. Int J Colorectal Dis. 2015;30(9):1165-71.
- Kim JH, Lee JY, Kim HK, Lee JW, Jung JW, Jung K, et al. Prognostic significance of the neutrophil-to-lymphocyte ratio and platelet-lymphocyte ratio in patients with stage III and IV colorectal cancer. World J Gastroenterol. 2017;23(3):505-15.
- Gu X, Gao XS, Qin S, Li X, Qi X, Ma M, et al. Elevated platelet to lymphocyte ratio is associated with poor survival outcomes in patients with colorectal cancer. PLoS One. 2016;11(9):e0163523.
- Kapiteijn E, Liefers GJ, Los LC, Kranenbarg EK, Hermans J, Tollenaar RA, et al. Mechanisms of oncogenesis in colon versus rectal cancer. J Pathol. 2001;195(2):171-8.
- Ko HM, Jung HH, Seo KH, Kang YR, Kang YR, Kim HA, et al. Platelet-activating factor-induced NF-kappaB activation enhances VEGF expression through a decrease in p53 activity. FEBS Letters. 2006;580(13):3006-12.
- Weisner T, Bugl S, Mayer F, Hartmann JT, Kopp HG. Differential changes in platelet VEGF, Tsp, CXCL12, and CXCR4 in patients with metastatic cancer. Clin Exp Metastasis. 2010;27(3):141-9.
- Labelle M, Begum S, Hynes RO. Platelet guides the formation of early metastatic niches. Proc Natl Acad Sci USA. 2014;111(30):E3053-3061.
- Niederhuber JE. Cancer vaccines: the molecular basis for T cell killing of tumor cells. Oncogist. 1997;2(5):280-3.
- Dunn GP, Old LJ, Schreiber RD. The immunobiology of cancer immunosurveillance and immunoediting. Immunity. 2004;21(2):137-48.
- Szkandera J, Pichler M, Absenger G, Stotz M, Arminger F, Weissmueller M, et al. The elevated preoperative platelet to lymphocyte ratio predicts decreased time to recurrence in colon cancer patients. Am J Surg. 2014;208(2):210-4.
- Yodying H, Matsuda A, Miyashita M, Matsumoto S, Sakazawa N, Yamada M, et al. Prognostic significance of neutrophil-to-lymphocyte ratio and platelet-lymphocyte ratio in oncologic outcomes of esophageal cancer: a systematic review and meta-analysis. Ann Surg Oncol. 2016;23(2):646-54.