Research Article
Unenhanced Chest CT Assisted SLNB could be a Highly Reliable Option for only Methylene Blue available Breast Cancer Patients
Haiyan Wei1*, Minya Yao1, Peifen Fu1 and Xiaoqun Ba2
1Department of Breast, Zhejiang University School of Medicine, China
2Department of Pathology, Zhejiang University School of Medicine, China
*Corresponding author: Haiyan Wei, Department of Breast, Breast Center, the First Affiliated Hospital, Zhejiang University School of Medicine, No. 79 Qingchun Road, Hangzhou, Zhejiang Province, 310003, China
Published: 05 Oct, 2018
Cite this article as: Wei H, Yao M, Fu P, Ba X. Unenhanced
Chest CT Assisted SLNB could be
a Highly Reliable Option for only
Methylene Blue available Breast Cancer
Patients. Clin Surg. 2018; 3: 2139.
Abstract
Background: Blue dye and Radioisotope (RI) combined SLNB is the “best” protocol. However, RI is
not available worldwide. This study is to evaluate the feasibility of unenhanced chest Computerized
Tomography (CT) with three criteria for suspicious nodal metastasis assisted SLNB using only
Methylene Blue (MB) as an alternative reliable option.
Patients and Methods: A total of 1771 consecutive patients with clinically node negative breast
cancer were enrolled. For limiting FNR and surgical complications, the number of removed Sentinel
Lymph Nodes (SLNs) was suggested to be 3~5. Unenhanced chest CT is mandatory to locate
SLNs, which were graded from 1 to 3 according to suspicious criteria for metastasis. Three doctors
adopted three SLNB methods. Periareolar sub-dermal injection using 4 ml of 0.5% MB, alone or in
combination with a radioisotope was individually employed by two doctors. The third doctor used
2 ml of 1% MB to inject into parenchyma to map SLNs.
Results: Both FNR and outcomes of patients showed no difference between three methods. We also
confirmed that Lymphovascular Invasion (LVI) and CT grade were both significantly correlated
with SLN status (coefficients were 0.68 and 0.25, p<0.001). Although there was no difference found
in complications, but parenchymal injection did never cause skin necrosis.
Conclusion: Unenhanced chest CT could be are liable assistance to improve SLNB using only MB
via parenchymal injection. This technique might be the safest and convenient option for SLNB in
the study and RI could be safely spared.
Keywords: Breast cancer; Sentinel lymph node biopsy; Methylene blue; Computerized tomography
Introduction
Chinese incidence of breast cancer has increased remarkably with the socioeconomic development, particularly in eastern coastal areas, and it has been expected to approach more than 100 cases per 100000 women aged 55~69 years by 2021 [1,2]. Whereas, benefitting from systemic therapy, the invasiveness of surgical treatment has been largely controlled. Clinical trials have revolutionized the pattern of clinical practice. Mastectomy and Axillary Lymph Node Dissection (ALND) were both profoundly proven not to improve survival and Quality of Life (QOL) when comparing with Breast-Conserving Therapy (BCT) and Sentinel Lymph Node Biopsy (SLNB) [3,4]. National Surgical Adjuvant Breast and Bowel Project (NSABP) B-32 and other trials identified that SLNB could not only reduce the surgical morbidities, but could also be reliable for axillary staging of clinically lymph node negative (cN0) breast cancer [5-6]. SNB has become the standard of care in patients with cN0 disease. Although blue dye and Radioisotope (RI) combined lymphatic mapping was well established as a standard procedure to limit FNR [7-9], but the optimal protocol for SLNB is still under investigation. In many Japanese hospitals, due to the lack of RI, contrast enhanced CT Lymphography (CT-LG) has been routinely used to stage the axillary accurately with dye-only SLNB [10]. It was concluded that using the only size criterion by CT-LG or MRI alone to diagnose SLNs was not sufficient. No matter what contrast regimen used, the cost and procedure of the technique they used were not practical in China. Based on previous studies on ultrasonography and CT-LG, metastatic nodes have several abnormal signs [11,12]. In our daily wok, US-guided Core Needle Biopsy (CNB) and unenhanced chest CT have routinely been preoperatively adopted to evaluate node status and triage the patients. Because of unavailable RI and limited financial support in our country, most of patients, especially who live in remote rural areas, must receive more unnecessary ALND. In this context, we conduct this research to evaluate an unenhanced chest CT assisted method for SLNB and provide an alternative option for the given population.
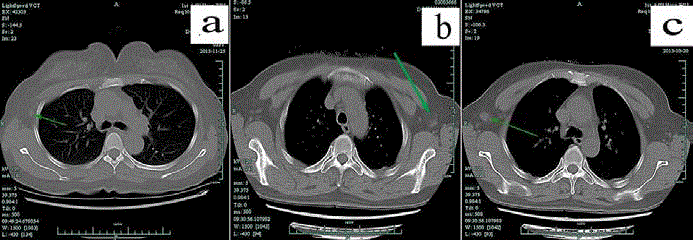
Figure 1
Figure 1
In unenhanced chest CT, axillary lymph nodes were graded according to their imaging features.
Green arrow marked representative lymph nodes. "Normal" node was shown in panel "a" and means nodes were thought without metastasis and it was featured
with diameter less than 2 cm and clear margin and clear corticomedullary demarcations. "Suspicious" node was shown in panel "b" and means metastasis could not
be excluded and their characteristics were clear margin and unclear corticomedullary demarcations or a diameter more than 2cm. As pointed out by green arrow,
the lymph node was displayed in consecutive 8 layers and pathologically identified as a false negative metastatic node. "Typical" node means metastasis was the
first consideration and displayed in panel "c". It was featured with unclear margin and corticomedullary demarcations and a diameter more than 1cm. "Suspicious"
and "typical" nodes were applied to core needle biopsy (CNB) under ultrasound guidance if they were found in ultrasound.
Patients and Methods
Patients' characteristics
From February 2007 to October 2015, a total of 1771cases of
breast cancer patients were involved in this study. They ranged in
age from 25 to 93 (median, 59). Their tumor size was ranged from
0.5 cm to 10 cm. The 10 cm tumor was identified as pure mucinous
carcinoma in the left breast of an 83-year old lady. All patients were
diagnosed by Fine Needle Aspiration (FNA) or Core Needle Biopsy
(CNB) before surgery. Patients who were arranged to neoadjuvant
chemotherapy including those diagnosed with inflammatory breast
cancer and pregnant patients were excluded. When axillary lymph
node was highly suspicious for metastasis, Ultrasound (US)-
guided CNB was introduced. When metastasis was not considered
in the node on palpation or US, cN0 was identified and SLNB was
applied. At the initial stage of SLNB, 190 patients consented to SLNB
followed by level I/II ALND. Three SLNB techniques were employed
individually by three surgeons. Thereafter, 1581 patients consented
to SLNB followed by ALND only if any metastasis was found in
Sentinel Nodes (SNs) except for Isolated Tumor Cells (ITC). Level
III clearance is performed unless metastasis occurred in nodes at
level II [13]. Lymphovascular Invasion (LVI) was widely found to be
significantly correlated with node status, therefore it was evaluated as
well and classified as three levels [14,15]. The first level was defined
as absence of LVI and the second and third levels were defined as
focal and diffused LVI, respectively. This retrospective research was
approved by the Ethics Committee of the First Affiliated Hospital,
Zhejiang University School of Medicine according to the revised
version of Declaration of Helsinki and its amendments in 1983, 1989,
and 1996 [16]. Patients have signed informed consent for publication
of their clinical data.
SLNB protocols
After cN0 breast cancer was confirmed, unenhanced chest CT
with 3 mm slice thickness was routinely performed and assessed by
two imaging experts. According to their experience and previous
study, three abnormal signs for suspicious nodes on CT were
identified [11]. Firstly, it is cortical thickening and diminished or
absent hilum. Secondly, a height/width ratio is close to 1 on the twodimensional
slice of a coronal scan across the hilum of the node.
Thirdly, the maximum diameter of the node is no less than 1 cm [12].
When the node meets one or more criteria, it was supposed to be
suspicious or typical metastatic SLNs and marked on the skin (Figure
1 and 2a). During operation, SLNs could easily be localized by the
size, numbers and the predicted anatomical position [17].
There is no consensus regarding the volume and location of the
injectate. In our institution, there are three experienced surgeons
who used to perform SLNB with their own fashion. Subcutaneous
injection of 4 ml of 0.5% MB at the periaerolar region (Method A)
was used by two surgeons and one of them used MB in combination
with radioisotope (Method B). MB was injected after the onset
of anesthesia. The radioactive tracer (99mTc) was injected and
lymphoscintigraphy was performed before the day of surgery. A
handheld gamma probe was employed for method B to detect hot
spots and all radioactive lymph nodes with highest signal comparing
with residual surgical field were removed. The third doctor use 2 ml
of 1% MB to inject into the parenchyma at the projection point of
tumor around the areola (Method C, Figure 2). SLNs did not include
parasternal lymph nodes.
The injection site was massaged for 2~3 min and operation started
10~15 min later. When no blue or radioactive node was found, ALND
was performed. It was suggested to harvest no less than 3 and no more
than 5 SNs as possible as it could be done. If there were only one or
two blue and/or hot nodes, palpable or visible nodes directed by CT
during operation were harvested as SLNs and this was suggested to
decrease FNR to less than 5% [18,19]. All patients were reexamined
unenhanced chest CT to evaluate the axillary 6 months after surgery.
Pathologic evaluation
All SLNs were bisected longitudinally at the level of the hilum
and half of it was taken for immediate frozen-section examination
during operation. The remainder was fixed in 10% formalin and
paraffin embedded and stained with Hematoxilin and eosin or further
histological examination. SLNs were all sliced at 2~3 mm intervals.
The definitions of micro metastasis and Isolated Tumor Cells
(ITC) were illustrated in National Comprehensive Cancer Network
(NCCN) breast cancer guideline [20].
Statistical analysis
Statistical analysis was performed with SPSS 22.0. IR and FNR
of three techniques for SLNB were estimated and compared by Chisquare
test. One-way ANOVA was used to compare numbers of
removed lymph nodes by three doctors. Disease Free Survival (DFS)
was defined as time (months) from the date of surgery to the date
of any advent of the first recurrence, and Overall Survival (OS) was
defined as time (months) from the date of surgery to the date of death
caused by breast cancer. DFS and OS were established and estimates
by Kaplan-Meier. Log-Rank (Mantel-Cox) test was used to compare
survival outcomes. Cox proportional-hazards regression model
was employed to estimate Hazard Ratio (HR) and 95% Confidence
Intervals (CI). Pearson's correlate coefficience and Multiple Linear
Regression (MLR) were used to assess correlation between SLNs
status and clinical parameters. A two-sided P value less than 0.05
were considered significant.
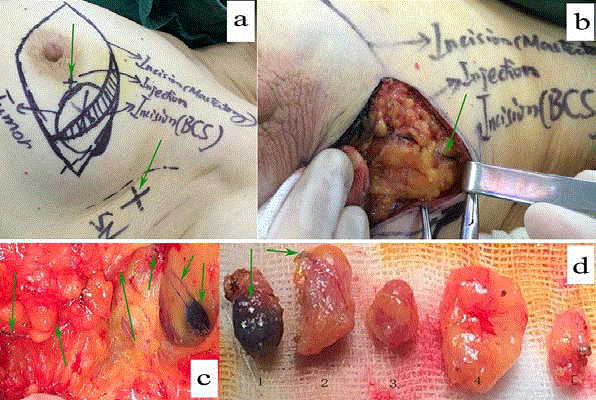
Figure 2
Figure 2
Lymphatic mapping using method C for a 36-year-old patient.
In panel "a", all possible incisions were designed and green arrows pointed out the only injection location with method C (2 ml of 1% MB was used to inject into the
parenchyma at the projection point of tumor around the areola) and the surface landmark of "suspicious" sentinel node guided by unenhanced chest CT. Panel "b"
and "c" and "d" showed blue lymphatic vessel and sentinel node dyed by methylene blue. In panel "d", the first and the fourth lymph nodes were proved metastasis
with breast carcinoma and the fourth node was found with palpation guided by CT.
Results
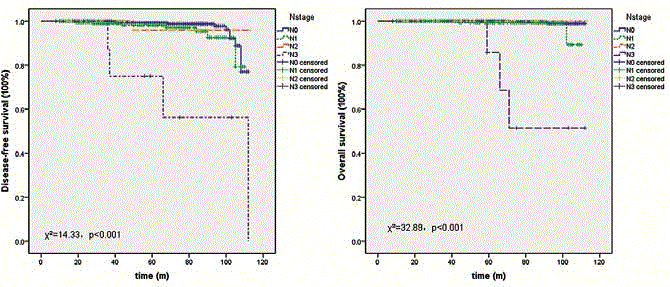
Of all enrolled patients with cN0 primary disease, 190 accepted SLNB followed by ALND. The IRs of Method A, B and C were 94.8% (55/58), 95% (57/60) and 94.4% (68/72) (χ²=0.0006, p=0.9997). FNRs were 4.35% (2/46), 6.12% (3/49) and 5.26% (3/57) (χ²=0.15, p=0.93). No significant difference was found between three groups of patients in N0~3 distributions (χ²=2.97, p=0.56). Skin necrosis happened in one case with Method A and no other complication was found. Afterward, ALND was applied only for patients with micro or macro metastasis in SLNs. By October 2015, additional 1581 patients adopted SLNB for their lymph node staging and the characteristics of successful patients were summarized in Table 1. The IRs of three methods were 97.5% (497/510), 96.8% (507/524) and 96.5% (528/547) (χ²=0.01, p=0.99). Because ALND was not mandatory, FNRs were inferred from limited results and they were discriminately 4.9% (18/365), 4.4% (16/367) and 4.6% (18/390). No significant different was found between three groups (χ²=0.14, p=0.93). Also, we evaluated FNR of chest CT for SLN and found significant difference between three grades (FNRs of grade 1 to 3 were 5.66%, 2.78% and 0; p<0.0001). Further, no difference was found in N0~3 distributions (χ²=2.67, p=0.85) and in complications, such as skin necrosis (χ²=3.72, p=0.16), sensory defects (χ²=0.91, p=0.63) and edema (χ²=2.02, p=0.36). Additionally, LVI displayed strong correlation with SLN status and axillary staging and chest CT. Pearson's coefficience were 0.59 and 0.73 and 0.449 (p<0.001). Besides, it showed weak correlation with tumor size and histopathology (0.08 and 0.10, p=0.01) and no correlation with cancer subtype (-0.03, p=0.44). In this study, MLR identified that LVI was a determining factor for lymph node status (β=0.68, 95% CI 0.599~0.76, p<0.001). Grade of lymph nodes in images of unenhanced chest CT and tumor grade were both revealed a weak effect on lymph node metastasis and their β values were 0.25 (95% CI 0.18~0.32, p<0.001) and 0.06 (95% CI 0.001~0.12, p=0.046). During follow-up, there are 16 patients found enlarged axillary lymph nodes when compared with before and these nodes were identified chronic inflammation by core needle biopsy. In the present dataset, survival outcomes of patients treated with SLNB and median follow-up of 42 months (range 8 to 113 months) were analyzed to compare three methods. There was no difference either in their DFS (χ²=0.44, p=0.51) or OS (χ²=0.004, p=0.95). Most of clinical characteristics, including tumor stage, LVI and unenhanced chest CT, showed no influence on survival of these patients, while lymph node stage always significantly influenced their DFS and OS (χ²=14.33, p<0.001; χ²=32.89, p<0.001; Figure 3). Multivariate analysis showed similar results in line with the above results. Cox proportional-hazards regression found that only lymph node status significantly influence DFS and OS and their HR were 2.06 (95% CI 1.39~3.06, p<0.001) and 4.37 (95% CI 2.28~8.38, p<0.001).
Discussion
In 1994, Sentinel Lymph Node Biopsy (SLNB) was reported by Giuliano and colleagues that it was firstly considered to be a potential method to replace ALND [21]. Now, SLNB has undoubtedly recognized as the standard procedure for cN0 breast cancer but the existing method of SLNB needs improving. For the sake of limited financial and health resources, the most widely used technique might take account of both safe and convenient. MB mapping method was convenient and proved not as accurate as combined with radioisotope. We adopted unenhanced chest CT to assist SLNB using only MB as the tracer. In China, unenhanced chest CT was cheap, convenient and mandatory for breast cancer patients to excluding lung metastasis and this test was included in the social insurance system. FNR is an index of SLNB for quality control and it was influenced by surgical skills, tracer and injection method [8,22]. For the purpose of limit FNR, we used unenhanced chest CT to locate suspected LNs pre-operatively to assist SLNB. According to the featured criterion of LNs in CT images, we could easily grade LNs and find SLNs with MB mapping during surgery. When the LN was graded 3 in CT images, no false negative node was found. At the beginning of SLNB in our institution, 190 cases were adopted SLNB followed by ALND. FNR and IR were both similar between three doctors. IRs was more than 90% and FNRs were all less than 7%. The possible bias of surgical skill between doctors in this study could be excluded. In the context of tracers, MB was profoundly proved safe and easily obtainable for almost all patients over the world. MB caused serious toxic adverse events only when it was administered with high dose of 7.5 mg/kg [23]. During SLNB, we used only 2 ml of 1% MB and its safety has been established even if the patient was pregnant [24]. MB was commonly injected around the areola under the skin from 2006 because IR of this method was reported to be higher. However, as previously reported, FNR of parenchymal injection was lower than superficial injection and it was safer since superficial injection might cause skin necrosis [25,26]. In our results, both IR and FNR of parenchymal injection showed no difference with superficial injection (χ²=0.05 and 0.18, p=0.97 and 0.91) and it didn't cause skin necrosis. Both of pathologic N-stage distribution and complications showed no difference between three methods for SLNB (χ²=2.67 and 6.75, p=0.85 and 0.35) [27-29].
Figure 3
Table 1
Conclusion
As reported previously, the only positive SLN rarely happened in the fourth SLN and the total number of SLNs harvested during surgery will markedly limit FNR. Moreover, Axelsson and colleagues suggested that less than 5 LNs during ALND increased axillary recurrence and decreased 5-year OS. So, we supposed that 3~5 of SLNs were the optimal number for accuracy and safety of SLNB. It has been found that parenchymal injection of MB caused undefined mass. We considered that 4 ml of 1% MB was too much and 2 ml of 1% MB used in this study did not cause any mass during follow-up. After all, we believe MB is a reliable and most convenient tracer for SLNB and it will allow maximizing breast cancer patients benefitting from SLNB. Unenhanced chest CT could be an efficacious assistance to improve SLNB using only MB injected in only one location and this might be an alternative "standard" procedure in developing countries.
Limitations of the Study
After all, there are several drawbacks in this study. Firstly, it was a retrospective study. However, patients involved in this study were arranged consecutively by their doctors. SLNB has become the standard procedure for early breast cancer, it is impossible to do any randomized prospective study to get the true FNR. Until now, there is no other tracer on a par with MB. A lot of clinical researches compared tracers with MB, but they are more expensive than MB, such asindocyanine green and enhancement agent [27]. Secondly, enhanced CT scan could significantly improve the accuracy of evaluation of LNs, but it is expensive and not convenient. Thirdly, there were significant different distribution in subtypes and LVI status (Table 1). This was due to pathology development because the “unknown” status was most seen in patients treated by the second doctor and she was the senior surgeon. In view of SNB technology and survival, the shortcomings did not affect successful rate and excellent survival.
Role of Funding Source
This study was supported by Sciences and Technology Program of Zhejiang Bureau of Public Health (No.2015KYB139) and this item was in the charge of the corresponding author.
Acknowledgement
We are extremely obliged to all patients who are brave and respectable. We also appreciated the research team of the First Affiliated Hospital, Zhejiang University School of Medicine.
References
- Fan L, Strasser-Weippl K, Li JJ, St Louis J, Finkelstein DM, Yu KD, et al. Breast cancer in China. Lancet Oncol. 2014;15(7):e279-89.
- Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66(2):115-32.
- Wood WC. Progress from clinical trials on breast cancer. Cancer. 1994;74(9):2606-9.
- Rao R, Euhus D, Mayo HG, Balch C. Axillary node interventions in breast cancer: a systematic review. JAMA. 2013;310(13):1385-94.
- Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP, Costantino JP, et al. Sentinel-lymph-node resection compared with conventional axillary-lymph-node dissection in clinically node-negative patients with breast cancer: overall survival findings from the NSABP B-32 randomized phase 3 trial. Lancet Oncol. 2010;11(10):927-33.
- Olson JA, McCall LM, Beitsch P, Whitworth PW, Reintgen DS, Blumencranz PW, et al. Impact of immediate vs. delayed axillary node dissection on surgical outcomes in breast cancer patients with positive sentinel nodes: results from American College of Surgeons Oncology Group Trials Z0010 and Z0011. J Clin Oncol. 2008;26(21):3530-5.
- Cox CE. Lymphatic mapping in breast cancer: combination technique. Ann Surg Oncol. 2001;8(9):67S-70S.
- Hindié E, Groheux D, Brenot-Rossi I, Rubello D, Moretti JL, Espié M. The sentinel node procedure in breast cancer: nuclear medicine as the starting point. J Nucl Med. 2011;52(3):405-14.
- Shirah GR, Bouton ME, Komenaka IK. Occurrence of prolonged injection site mass with methylene blue but not isosulfan blue after the sentinel node procedure. Arch Surg. 2011;146(2):137-41.
- Motomura K, Izumi T, Tateishi S, Tamaki Y, Ito Y, Horinouchi T, et al. Superparamagnetic iron oxide-enhanced MRI at 3 T for accurate axillary staging in breast cancer. Br J Surg. 2016;103(1):60-9.
- Abe H, Schmidt RA, Sennett CA, Shimauchi A, Newstead GM. US-guided core needle biopsy of axillary lymph nodes in patients with breast cancer: why and how to do it. Radiographics. 2007;27:S91-9.
- Tan H, Yang B, Wu J, Wana S, Gu Y, Li W, et al. Localization and Evaluation of Sentinel Lymph Node in Breast Cancer from Computed Tomographic Lymphography. J Comput Assist Tomogr. 2011;35(3):367-74.
- Kinne DW. The surgical management of primary breast cancer. CA Cancer J Clin. 1991;41(2):71-84.
- Kohrt HE, Olshen RA, Bermas HR, Goodson WH, Wood DJ, Henry S, et al. New models and online calculator for predicting non-sentinel lymph node status in sentinel lymph node positive breast cancer patients. BMC Cancer. 2008;8:66.
- van la Parra RF, Peer PG, Ernst MF, Bosscha K. Meta-analysis of predictive factors for non-sentinel lymph node metastases in breast cancer patients with a positive SLN. Eur J Surg Oncol. 2011;37(4):290-9.
- World Medical Association declaration of Helsinki. Recommendations guiding physicians in biomedical research involving human subjects. JAMA. 1997;277(11):925-6.
- Yuen S, Yamada K, Goto M, Sawai K, Nishimura T. CT-based evaluation of axillary sentinel lymph node status in breast cancer: value of added contrast-enhanced study. Acta Radiol. 2004;45(7):730-7.
- East JM, Valentine CS, Kanchev E, Blake GO. Sentinel lymph node biopsy for breast cancer using methylene blue dye manifests a short learning curve among experienced surgeons: a prospective tabular cumulative sum (CUSUM) analysis. BMC Surg. 2009;9:2.
- Chetty U, Chin PK, Soon PH, Jack W, Thomas JS; Edinburgh Breast Unit. Combination blue dye sentinel lymph node biopsy and axillary node sampling: the Edinburgh experience. Eur J Surg Oncol. 2008;34(1):13-6.
- Gradishar WJ, Anderson BO, Balassanian R. Breast Cancer Guideline, Version 2. ST-2-3. National Comprehensive Cancer Network®. 2016.
- Giuliano AE, Kirgan DM, Guenther JM, Morton DL. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Ann Surg. 1994;220(3):391-8.
- Diepstraten SC, Sever AR, Buckens CF, Veldhuis WB, van Dalen T, van den Bosch MA, et al. Value of preoperative ultrasound-guided axillary lymph node biopsy for preventing completion axillary lymph node dissection in breast cancer: a systematic review and meta-analysis. Ann Surg Oncol. 2014;21(1):51-9.
- Tummers QR, Schepers A, Hamming JF, Kievit J, Frangioni JV, van de Velde CJ, et al. Intraoperative guidance in parathyroid surgery using near-infrared fluorescence imaging and low-dose Methylene Blue. Surgery. 2015;158(5):1323-30.
- Gropper AB, Calvillo KZ, Dominici L, Troyan S, Rhei E, Economy KE, et al. Sentinel lymph node biopsy in pregnant women with breast cancer. Ann Surg Oncol. 2014;21(8):2506-11.
- Reintgen M, Kerivan L, Reintgen E, Swaninathan S, Reintgen D. Breast Lymphatic Mapping and Sentinel Lymph Node Biopsy: State of the Art: 2015. Clin Breast Cancer. 2016;16(3):155-65.
- Lee JH, Chang CH, Park CH, Kim JK. Methylene blue dye-induced skin necrosis in immediate breast reconstruction: evaluation and management. Arch Plast Surg. 2014;41(3):258-63.
- Ahn SK, Kim MK, Kim J, Lee E, Yoo TK, Lee HB, et al. Can We Skip Intra operative Evaluation of Sentinel Lymph Nodes? Nomogram Predicting Involvement of Three or More Axillary Lymph Nodes before Breast Cancer Surgery. Cancer Res Treat. 2017;49(4):1088-96.
- Zakaria S, Degnim AC, Kleer CG, Diehl KA, Cimmino VM, Chang AE, et al. Sentinel lymph node biopsy for breast cancer: how many nodes are enough? J Surg Oncol. 2007;96(7):554-9.
- Axelsson CK, Mouridsen HT, Zedeler K. Axillary dissection of level I and II lymph nodes is important in breast cancer classification. The Danish Breast Cancer Cooperative Group (DBCG). Eur J Cancer. 1992;28A(8-9):1415-8.