Research Article
Closed Incision Negative Pressure Therapy for Laparotomy Wounds: A Review
Fowler AL* and Barry MK
Department of Surgery, National University of Ireland, Ireland
*Corresponding author: Fowler AL, Department of Surgery, National University of Ireland, Galway, Ireland
Published: 20 Sep, 2018
Cite this article as: Fowler AL, Barry MK. Closed Incision
Negative Pressure Therapy for
Laparotomy Wounds: A Review. Clin
Surg. 2018; 3: 2123.
Abstract
Background: Open abdominal surgery is associated with high rates of wound complications.
Surgical site infection (SSI) carries a significant burden to the healthcare system and the patient and is
associated with prolonged length of stay, delayed treatment and high rates of readmission. Negative
pressure wound therapy over closed incisions (ciNPWT) is a novel approach to prevention of SSI.
We reviewed the outcomes of studies comparing ciNPWT and standard therapy on laparotomy
wounds to assess the efficacy of the current evidence base.
Aim: To assess the effect of negative pressure wound therapy used over closed incisions in open
abdominal surgery.
Methods: Search of relevant terms was conducted on PubMed and Google Scholar to identify
relevant studies published between Jan 2006 - Dec 2017. Studies were chosen based on specific
inclusion and exclusion criteria. An additional search was conducted to identify epidemiology, risk
factors and cost-burden of SSI. Articles were screened to assess demographics, study design and
outcomes.
Results: Five retrospective and four prospective randomised controlled trials were identified for
inclusion, totalling 1470 participants. 454 received ciNPWT and 1016 received standard treatment.
Studies assessed a mix of surgeries (4=colorectal, 1=pancreaticoduodenectomy, 1=gynaecologic,
2=mixed). ciNPWT was statistically significant in reducing SSI in 8 of 9 studies (p<0.05).
Conclusion: ciNPWT is a promising therapy for reducing the rate of SSI in open abdominal surgery
however; its effect on other wound complications is unclear. Further multicenter, prospective
studies are needed to assess cost-benefit, appropriate patient-selection and assess efficacy of closed
incision negative pressure therapy in open abdominal surgery.
Keywords: Negative pressure wound therapy; Laparotomy; Open abdominal surgery; Surgical site infection; Complication
Introduction
Postoperative wound issues are a frequent cause of surgical morbidity and a large-scale health
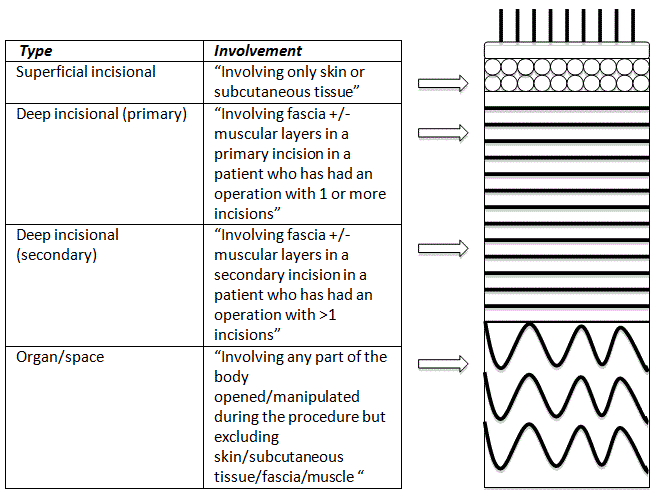
problem [1-5]. Wound issues can range from superficial surgical site infection to deep surgical site
infection, organ space infection and complete wound dehiscence (Figure 1) [6]. Hospital associated
infections affect one in twenty patients, with surgical site infection (SSI) being one of the most
common causes [7].
SSI represents a significant healthcare cost both directly and indirectly [3]. Studies in the UK
found that treatment for SSI costs between £814 and £6626 per patient [4]. Wound complications
cause high rates of morbidity, prolonged postoperative length of stay and increased readmission
rates. Furthermore, it can lead to a delay in further treatment in those undergoing adjuvant chemoand
radiotherapy [4]. A significant mortality risk of 3% exists for those with surgical site infection,
which is directly related to SSI occurrence in 75% of cases [8].
Current perioperative guidelines for the prevention of SSI in Ireland include hair removal with
clippers, correct usage and timing of antibiotic prophylaxis, use of appropriate skin preparation
and maintenance of perioperative normothermia and normoglycemia [7]. Similar guidelines are
replicated worldwide [6]. Despite these methods, rates of SSI remain high. To date, research has
been conducted to further reduce SSI with techniques such as application of topical antimicrobial
agents, use of debriding agents, wound protectors and various dressing types to prevent infection
and stimulate wound healing [9-13].
The incidence of wound complications after laparotomy is
amongst the highest in surgical practice particularly in colorectal
surgery, where incidence of SSI is found to be as high as 45% [2,14-
16]. SSI occurs in the absence of perforation, representing a risk to
any patient undergoing bowel resection [17]. The most frequent
causative organisms are those found in the gastrointestinal tract,
suggesting that infection develops from contamination of the wound
by intraluminal organisms [5]. Mechanisms to clear pathogens from
the wound have been proposed to reduce the incidence of surgical
site infection [5].
Negative pressure wound therapy (NPWT) is a model of therapy
used in wound care since the 1990s and was originally introduced
as a novel method of management for chronic open wounds [18,19].
NPWT consists of a pump attached to foam or gauze dressing via a
tubing system. The device generates either intermittent or continuous
negative pressure ranging from 20-125 mmHg and collects fluid away
from the wound [16,20]. Its mechanism of action includes reducing
oedema, promoting blood flow and angiogenesis, draining exudate
and contracting wound edges [21-24]. Its success in this area has lead
to its introduction in other areas of wound care such as acute open
wounds [25], particularly in the setting of trauma and more recently,
in closed postoperative incisions. Initially trialled in orthopaedic
surgery, “incisional NPWT” has shown promising results in reduction
of SSI rates across vascular, cardiothoracic, plastic and abdominal
surgery [26-29].
Multiple studies and reviews have been conducted to assess
efficacy, cost-effectiveness and overall treatment benefit of closed
incision NPWT (ciNPWT) [9,15,16,20,23,26-34]. To our knowledge,
this is the first literature review to assess the use of ciNPWT in open
abdominal surgery.
Aims and Objectives
Primary aim
To assess the effect of negative pressure wound therapy used over
closed incisions in open abdominal surgery.
Secondary aims
1) To assess the impact of SSI in abdominal surgery 2) to assess
current risk-reduction strategies in prevention of SSI in abdominal
surgery 3) to assess cost-savings associated with the use of ciNPWT 4)
to assess potential complications associated with the use of ciNPWT
in abdominal surgery.
Materials and Methods
Data search
PubMed and Google Scholar search engines were used to identify
relevant studies published between Jan 2006 - Dec 2017. The search
was conducted from November 2017 - January 2018. Keywords
included the terms “incisional negative pressure wound therapy”,
“vacuum therapy”, “vacuum assisted closure”, “closed incision”,
“closed wound”, “topical negative pressure”, “negative pressure
therapy”, “NPWT”, “INPWT”, “Prevena”, “PICO”, combined with
terms; “abdominal surgery”, “laparotomy”, “abdomen”, “general
surgery”.
A further search was performed to identify risk-factors and
epidemiology of surgical site infection using the terms; “surgical site
infection”, “SSI”, “surgical site event”, “wound complication(s)”,
“surgical wound dehiscence”, “prevention”.
Additional search terms were identified from the keywords of
relevant articles identified. A “snowball” search method was utilised
(continued search of relevant references found in the literature).
Systematic, literature reviews and meta-analysis in the area of interest
were also identified for comparison.
Inclusion criteria
Studies were included if they met the following search criteria:
1) Studies comparing closed incisional negative pressure wound
therapy (ciNPWT) to standard dressings in abdominal surgery, 2)
study participants >18 years, 3) human studies, 4) studies in English,
5) full-text articles, 6) primary/secondary end-point of surgical site
event (SSE)/surgical site infection (SSI) (Table 1).
Exclusion criteria
Exclusion criteria included 1) negative-pressure wound therapy
over open wounds, 2) use of negative pressure wound therapy
in wound management in other surgical specialties, 3) negative
pressure wound therapy for existing wound issues or abdominal wall
reconstruction, 4) preclinical or animal studies, 5) paediatric studies,
6) non-comparative study design (Table 1).
Data extraction
Following removal of duplicate records, articles were screened for
relevance. Abstracts and full articles deemed eligible were assessed.
Remaining publications were read to ensure they met inclusion and
exclusion criteria. The references of suitable articles were searched for
additional articles that may fit the criteria. Data of interest included 1)
year of publication 2) inclusion/exclusion criteria 3) study design 4)
postoperative follow-up 5) patient characteristics 6) type of surgery 7)
incidence and type of postoperative wound complications.
Results
Initial search of the databases identified 4714 results. After
removing duplicates and application of inclusion and exclusion
criteria, a total of nine studies remained. Studies are described in
Table 2.
The review identified five retrospective and four prospective trials
containing a total of 1470 participants. Of those, 454 received NPWT
versus 1016undergoing conventional treatment. Vacuum-assisted
closure (V.A.C, KCI, San Antonio, TX) was the most common type
of negative pressure therapy to be administered (n=4). All studies
followed patients for a minimum of 30 days (n=9) Five studies assessed
NPWT in those at high risk of SSI, including abdominal oncologic
resection for colorectal, pancreatic and peritoneal disease, oncologic
pancreaticoduodenectomy, oncologic gynaecologic resection and
bowel resection/stricturoplasty for Crohn’s disease. Four studies
assessed open colorectal surgery only. Duration of NPWT ranged
from 3 to 7 days. Four studies assessed solely elective surgery, with the
rest assessing a mix of elective and emergency surgery. Most studies
assessed clean or clean-contaminated operations (n=7).
Across all studies, 118 patients receiving NPWT experienced
postoperative wound complications, with the majority being
superficial infections requiring oral antibiotic therapy only. All
studies found NPWT to prevent surgical site infection and this was
statistically significant in 8 of 9 studies (p<0.05).
Discussion
Impact of surgical site infection
Surgical site infections affect approximately 25% of those
undergoing surgical procedures and are the second most common
healthcare associated infection [38]. Wound complications,
specifically SSI, are common after open abdominal surgery resulting
in some of the highest rates amongst surgical specialties [14,32,39,40].
The economic burden of surgical site complications is huge, both
from direct cost and from impact on length of stay and readmission
rates [1,26]. A study by Wick et al. demonstrated a 30-day SSI rate
of 18.9% in colorectal surgery; the presence of SSI doubling the risk
of readmission. This was associated with a median cost of $12835
per readmission [1]. Indirect cost is also accrued from the longterm
impact of surgical site events (SSE), which leads to morbidity
in the form of chronic wound issues and incisional hernia [41]. The
impact is apparent both on the healthcare system and on the patient,
with many reporting adverse effects on their quality of life following
wound complications and is most evident in those requiring further
surgical intervention [42].
Identifying risk factors for SSI
Multiple strategies have been developed to target high-risk
patients, identify operations and recognise intraoperative factors,
which may lead to the development of wound complications. These
are described in Table 3.
Willy et al. performed a literature review across surgical specialties
and identified patient-related, operation-related and specialtyrelated
factors leading to the development of SSI. Comorbidities
most frequently cited as risk factors for SSI include male gender,
diabetes mellitus, ASA grade ≥ 3, BMI ≥ 30, hypoalbuminemia,
renal disease, active alcoholism, smoking, corticosteroid use and
immunosuppression. Surgical factors include prolonged operation
time, emergency operation, re-operation and increased intraoperative
blood loss. They also identified certain operations carrying a higher
risk of wound issues; in general surgery, this included any open
procedures and incisional hernia repair [38].
Specific to colorectal surgery, Wick et al. analysed 1646 patients
undergoing colon or rectal resection and identified BMI ≥30
(p=0.008), platelet count <150/microl (p=0.021), age >55 (p=0.045)
and operation duration >180 min (p=0.034) as statistically significant
risk factors for SSI [14]. Similar findings have been replicated by
other authors [26,27]. These results are useful in identifying high-risk
patients, but relatively small patient cohorts in these studies could not
be used to develop a risk reduction tool.
Using data obtained from the American College of Surgeons
National Surgical Quality Improvement Programme (ACS-NSQIP),
Walraven et al. proposed a risk scoring system (Site Infection Risk
Score) to identify patient related and procedure related predictors
for SSI. Whilst this was validated, it was found to be limited in its
application in routine practice [43]. A similar, easy-to-use predictive
tool was also developed by Hedrick et al but has not been validated
in practice [44]. To date, a clinically useful tool to target those at risk
still remains to be seen.
Current prevention mechanisms
National Institutes of Health have recognised the need to decrease
SSE and have developed guidelines for the reduction of postoperative
surgical site occurrences. In 2006, the US Centers for Medicare &
Medicaid Services developed the Surgical Care Improvement Project,
which aimed to reduce SSI incidence and associated morbidity
[45]. This introduced seven perioperative measures including
administration of prophylactic intravenous antibiotics within 1 hr
of incision, correct antibiotic selection, cessation of prophylactic
antibiotics within 24 hr, appropriate hair removal, perioperative
euglycemia, normothermia and removal of urinary catheters within
48 hr. Similar guidelines have been introduced in Ireland and much
of this practice is now routine [7].
In addition to this, the development of the ACS-NSQIP has been
found to reduce SSI rates and costs associated with them. Analysis
of cost-savings in one centre found a reduction of 2.5 million dollars
attributable to a decline in SSI rates over a two year period [46]. A tenhospital
collaborative formed in Tennessee looked at all NSQIP data
across ten hospitals over two separate periods. They reported a decline
in deep incisional SSI (RR 0.82, 95% CI 0.63 to 1.06) and a significant
decrease in superficial site infections of 18.9% (RR 0.81, 95% CI 0.71
to 0.92) due to introduction of SSI programmes. They proposed that
this was potentially limited by a “Hawthorne effect” and that decreases
in SSI could not be directly attributable to improvement programmes
alone, but are more likely due to awareness of practice [47].
The true impact of these reduction measures on the rate of SSI is
controversial and remains uncertain [48,49]. Alternative methods to
reduce wound issues have yielded mixed results with limited efficacy.
These include use of gentamycin-soaked sponges placed in the
surgical wound [10], use of subcutaneous drains [11], use of wound
protectors [12,13], and use of topical antimicrobials [50]. As yet, no
single method has been found to be useful in prevention of SSI in
those undergoing open abdominal surgery.
Use of negative pressure wound therapy in closed
incisions
Since its introduction in 1997, NPWT has shown promising
results in the treatment of chronic and otherwise difficult-to-manage
wounds [18,19]. Its efficacy was confirmed in a meta-analysis
published by Cochrane, which assessed several randomised controlled
trials comparing ciNPWT to conventional wound management
across specialties and demonstrated improvement in wound size and
reduction in time to healing with NPWT [51].
Studies assessing the mechanism of NPWT suggest effectiveness
through a number of methods. Firstly, increased lymphatic clearance
leads to reduced tissue oedema and removal of excess fluid from
the wound. Contraction of wound edges decreases lateral wound
tension thereby preventing stress at the incision site, improving
scar appearance and risk of wound breakdown. Finally, regions
of hypoperfusion at the wound edges caused by NPWT stimulate
production of angiogenic vascular endothelial growth factor
increasing blood flow to the wound and deposition of granulation
tissue [21,22,24].
The success of NPWT has led to its introduction in other wound
management scenarios, initially in the setting of acute open wounds
and most recently as a preventative measure over closed surgical
incisions. Use of ‘incisional’ NPWT was first trialled in orthopaedic
surgery by Gomoll et al. [23]. They demonstrated a reduction in SSI
with use of incisional vacuum-assisted therapy in high-risk patients
undergoing a multitude of orthopaedic procedures including revision
hip arthoplasty, femoral and tibial fracture fixation, and foot and ankle
trauma [23]. Closed-incision negative pressure wound therapy has
since shown promising results across a range of surgical specialties
[28,29,33].
Most recently, ‘closed-incision Negative Pressure Wound
Therapy (ciNPWT) has been introduced in open abdominal surgery.
Current guidelines recommend covering incisions with a sterile
wound dressing prior to removal of surgical drapes at the end of the
procedure, leaving the dressing intact for 48 hrs [7]. Laparotomy
poses an increased risk of SSI and for this reason the mechanism
of NPWT provides potential for the successful prevention of
surgical site complications in this setting. Additional prophylactic
benefits including sealing of the wound to exogenous bacteria and
contamination are particularly useful in the setting of laparotomy
with stoma formation [16,32].
Current evidence base for ciNPWT use in open abdominal
surgery
Studies assessing the efficacy of ciNPWT in open abdominal
surgery have demonstrated overall rates of SSI ranging from
14.1% to 28.0% [31,37]. Eight of the nine trials identified showed a
statistically significant reduction in SSI associated with the use of
ciNPWT. Moreover, most infections in those treated with NPWT
were superficial and did not require further intervention. O’Leary
et al. assessed 50 patients undergoing elective or emergency open
abdominal procedures, with results showing only two SSI in the
NPWT treatment group, all of which were superficial infection versus
8 in the control group [33]. Li et al. also found promising results
in patients undergoing mostly elective open operations including
colorectal resection, primary ventral hernia repair and intraabdominal
tumour resection. They demonstrated a decrease in SSI
rate of 3.0% vs. 23.7% (p=0.031) with only one superficial infection
in the ciNPWT group [37]. These studies included only small patient
cohorts and so cost savings and overall benefit of NPWT across all
patient groups is difficult to ascertain.
Most ciNPWT studies identified included only patients with
risk factors for SSI, including malignancy, smoking history,
immunosuppression, malnutrition, emergency surgery and
inflammatory bowel disease and obesity [26,27,30,32,35-37]. Zaidi et
al. analysed use of NPWT in clean contaminated or contaminated
open colorectal surgery and demonstrated a reduction in SSI, with
only two patients in the ciNPWT developing wound infection post
operatively [32]. Selvaggi et al. conducted a study assessing use of
ciNPWT in Crohn’s patients undergoing elective bowel resection or
stricturoplasty and found that NPWT was protective against SSI in
this patient cohort (OR 0.21 95%CI 0.15-0.5, P=0.005). Moreover use
of PICO dressings was found to prevent infectious SSE in patients
on corticosteroid therapy (p=0.001) [31]. Positive results were also
demonstrated by Blackham et al. who studied high-risk oncology
patients undergoing elective surgery for colorectal, pancreatic and
peritoneal malignancy. They found not only a reduction in SSI
(6.7% versus 19.5% p=0.015) but also a decline in the rate of total
complications (24% vs. 35.6% p=0.050). Of interest, they noted a
higher rate of deep incisional infections in those in the NPWT group
(n=5 vs. n=0). This was not statistically significant but may represent
systemic physiological stresses resulting from decreased perfusion
of the fascia, which cannot be prevented by use of incisional NPWT
[26].
Additional benefits proven in the studies included a reduction in
length of stay in patients treated with ciNPWT, as demonstrated by
O’Leary et al. (6.1 days versus 14.7 days, p=0.019) and replicated in
a study by Pellino et al. (7.1 vs. 12 days, p=0.001) [30,33]. There was
also a reduction in rates of seroma in patients undergoing colorectal
resection (8% vs. 40%, p=0.02) and in patients undergoing bowel
resection or stricturoplasty for Crohn’s disease [31].
Only one study did not find a statistical benefit to use of ciNPWT
in laparotomies. Lynam et al. performed a retrospective review of
patients undergoing laparotomy for gynaecological malignancy
[35]. Whilst there was a reduction in SSI in the NPWT, no statistical
difference was demonstrated (p=0.40). It was noted that as a
retrospective study, selection bias existed and those receiving NPWT
were more likely to be obese (30.67 in control group versus 41.29
in NPWT group, p<0.001) [35]. This still represents a potential
therapeutic benefit as reduction was still seen even in this high-risk
group.
No adverse effects of ciNPWT were reported in any study
analysed. In previous studies, such as that by Conde-Green et al.,
intact skin has become excoriated, irritated or bruised if exposed
directly to the polyurethane foam of the NPWT dressing [9]. This
was not found to be the case in any study assessed and is reduced
by a barrier between the skin and the foam, which is found in most
pre-made ciNPWT dressings [27]. Burkhart et al. found that those
with iVAC dressing were more likely to have a pancreatic fistula;
however those selected for VAC therapy were at increased risk of
fistula formation [36]. There is also a theoretical risk that the device
may conceal an SSI however cellulitis of the surrounding skin should
be visible with most ciNPWT dressings. Selvaggi et al. found ciNPWT
to be well tolerated by patients, included those sent home with the
device, of which none required unscheduled follow-up to manage the
device or dressing [31].
Multiple systematic reviews and meta-analysis have been
produced to assess incisional negative-pressure wound management.
Most have pooled data from all specialties and have included caseseries,
observational studies as well as randomised controlled
trials leading to heterogeneity of data [20,34,52,53]. Strugala et al.
assessed use of PICO across orthopaedic, abdominal, colorectal
and obstetric specialties, including 6 observational studies and 10
randomised trials [53]. They demonstrated a significant reduction
in SSI across all specialties (RR 0.32, P<0.0001). Sandy-Hodgetts
et al. analysed eight studies including two open abdominal surgery
trials and found results in favour of ciNPWT in preventing SSI but
conflicting results for reduction of wound dehiscence and seroma.
They concluded benefits of ciNPWT in those deemed high-risk [20].
Similar results were published in a meta-analysis by Semsarzadeh et
al, which demonstrated a risk reduction (RR) of 29.4% with the use
of NPWT [52]. Scalise et al. also found a decrease in the incidence
of infection, sero-haematoma and re-operation rates with the use of
closed-incision NPWT [34]. Only one systematic review from Pellino
et al. focused on open abdomen trials and analysed colorectal studies
specifically. They included five studies with a total of 493 patients of
whom 147 received closed incision NPWT. They concluded in favour
of use of ciNPWT in colorectal surgery patients [16].
Future considerations for ciNPWT use
To date, ciNPWT has shown potential as a safe and effective
treatment modality however the setting for its use remains unclear.
The major downside is the associated cost of closed-incision negative
pressure wound management systems, which are more expensive
than traditional standard wound management. The PICO disposable
incisional vacuum unit costs between $300 - $600 (PICO, Smith &
Nephew, London, UK), and the Prevena system costs $500 (V.A.C.
therapy KCI, San Antonio, TX). These costs can be somewhat
defended when considering SSI can result in a mean increase in
healthcare costs of 115% [3] but cost-effectiveness of ciNPWT in
open abdominal surgery is yet to be established.
Recent advice from the National Institute for Health and Care
Excellence recommends use of PICO for those at risk of developing
surgical site infection or for treatment surgical site complications.
Specifically, PICO is a cost effective alternative to standard care for
closed surgical incision. Whilst the cost of PICO ranges from £126.88
- £145.68 per dressing, they suggest that the additional cost can be
offset by the potential reduction in surgical site infection, particularly
in those at high risk where the cost of surgical site infection in a
general surgical patient can be as high as £14,000 [54,55].
As aforementioned, identification of those at high-risk is also
unclear. Many studies have proposed those at increased risk of
SSI and most likely to benefit from ciNPWT including presence of
diabetes mellitus [27], incision length >20 cm, ASA >2 grade (37),
age >65 yrs and stoma formation [2]. Willy et al. proposed consensus
recommendations and an algorithm to identify those most likely to
benefit from NPWT however this is yet to be rationalised in clinical
practice [38]. More work is needed in this area to identify those who
would benefit most from ciNPWT.
PICO and Prevena systems have been developed specifically for
use in closed-incision situations however limitations to their use
have been established. The Prevena system contains a canister, which
can be cumbersome for patient use and can only hold 45 ml of fluid.
It is also significantly more expensive than standard conventional
therapy. The PICO dressing becomes ineffective if saturated with over
200 ml of exudate. Furthermore, neither system has clearly defined
‘discontinuation criteria’ nor this may vary with the patient and type
of incision. These points represent potential pitfalls of the devices and
their usage [33,34].
Little prospective data is currently available to assess use of
ciNPWT and this is a potential area of development. At present, two
trials are on-going which may shed more light on use of incisional
vacuum therapy. The NEPTUNE trial is a single-institution,
prospective, randomised, open blinded endpoint trial, which aims
to recruit approximately 300 patients undergoing elective open
colorectal surgery to either Prevena therapy or standard wound
management. The primary outcome measure is SSI within the first
30 post-operative days, with secondary aims including length of stay,
number of readmissions, cost and need for public health care.
The PONIY trial is a similar randomized, controlled observer
multicenter clinical trial assessing the use of Prevena in open elective
colorectal surgery.
Figure 1
Table 1
Table 2
Table 3
Conclusion
ciNPWT is a promising therapy for use in open abdominal surgery. It has been shown to have potential in reducing the rate of SSI in this area however, its effect on other areas of wound complications remains uncertain. Due to the potential for publication selection bias and heterogeneity of data, no conclusions can be drawn as to which patient cohorts will most benefit from this therapy. Further multicenter, prospective studies are needed to assess cost-benefit, appropriate patient-selection, duration of treatment and efficacy of closed incision negative pressure therapy in open abdominal surgery.
Acknowledgment
The authors would like to acknowledge Dr. James Brown, PhD, MA (Ed), (Lecturer, Discipline of Surgery, School of Medicine, National University of Ireland, Galway), for his input in editing this review.
Clinical Image
- Wick EC, Shore AD, Hirose K, Ibrahim AM, Gearhart SL, Efron J, et al. Readmission rates and cost following colorectal surgery. Diseases of the colon and rectum. 2011;54(12):1475-9.
- Konishi T, Watanabe T, Kishimoto J, Nagawa H. Elective colon and rectal surgery differ in risk factors for wound infection: results of prospective surveillance. Annals of surg. 2006;244(5):758-63.
- Broex EC, van Asselt AD, Bruggeman CA, van Tiel FH. Surgical site infections: how high are the costs? J Hosp Infect. 2009;72(3):193-201.
- Coello R, Charlett A, Wilson J, Ward V, Pearson A, Borriello P. Adverse impact of surgical site infections in English hospitals. J Hosp Infect. 2005;60(2):93-103.
- Mihaljevic AL, Schirren R, Muller TC, Kehl V, Friess H, Kleeff J. Postoperative negative-pressure incision therapy following open colorectal surgery (Poniy): study protocol for a randomized controlled trial. Trials. 2015;16:471.
- Anderson DJ, Podgorny K, Berrios-Torres SI, Bratzler DW, Dellinger EP, Greene L, et al. Strategies to prevent surgical site infections in acute care hospitals: 2014 update. Infection control and hospital epidemiology. 2014;35(6):605-27.
- Preventing Surgical Site Infections. Key Recommendations for Practice 2012.
- Awad SS. Adherence to surgical care improvement project measures and post-operative surgical site infections. Surg Infect (Larchmt). 2012;13(4):234-7.
- Conde-Green A, Chung TL, Holton LH 3rd, Hui-Chou HG, Zhu Y, Wang H, et al. Incisional negative-pressure wound therapy versus conventional dressings following abdominal wall reconstruction: a comparative study. Ann Plast Surg. 2013;71(4):394-7.
- Bennett-Guerrero E, Pappas TN, Koltun WA, Fleshman JW, Lin M, Garg J, et al. Gentamicin-collagen sponge for infection prophylaxis in colorectal surgery. N Engl J Med. 2010;363(11):1038-49.
- Watanabe J, Ota M, Kawamoto M, Akikazu Y, Suwa Y, Suwa H, et al. A randomized controlled trial of subcutaneous closed-suction Blake drains for the prevention of incisional surgical site infection after colorectal surgery. Int J Colorectal Dis. 2017;32(3):391-8.
- Zhang MX, Sun YH, Xu Z, Zhou P, Wang HX, Wu YY. Wound edge protector for prevention of surgical site infection in laparotomy: an updated systematic review and meta-analysis. ANZ J Surg. 2015;85(5):308-14.
- Edwards JP, Ho AL, Tee MC, Dixon E, Ball CG. Wound protectors reduce surgical site infection: a meta-analysis of randomized controlled trials. Ann Surg. 2012;256(1):53-9.
- Wick EC, Vogel JD, Church JM, Remzi F, Fazio VW. Surgical site infections in a "high outlier" institution: are colorectal surgeons to blame? Dis Colon Rectum. 2009;52(3):374-9.
- Shen P, Blackham AU, Lewis S, Clark CJ, Howerton R, Mogal HD, et al. Phase II Randomized Trial of Negative-Pressure Wound Therapy to Decrease Surgical Site Infection in Patients Undergoing Laparotomy for Gastrointestinal, Pancreatic, and Peritoneal Surface Malignancies. J Am Coll Surg. 2017;224(4):726-37.
- Pellino G, Sciaudone G, Selvaggi F, Canonico S. Prophylactic negative pressure wound therapy in colorectal surgery. Effects on surgical site events: current status and call to action. Updates Surg. 2015;67(3):235-45.
- Taylor GD, Kirkland TA, McKenzie MM, Sutherland B, Wiens RM. The effect of surgical wound infection on postoperative hospital stay. Can J Surg. 1995;38(2):149-53.
- Argenta LC, Morykwas MJ. Vacuum-assisted closure: a new method for wound control and treatment: clinical experience. Annals of plastic surgery. 1997;38(6):563-76.
- Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W. Vacuum-assisted closure: a new method for wound control and treatment: animal studies and basic foundation. Ann Plast Surg. 1997;38(6):553-62.
- Sandy-Hodgetts K, Watts R. Effectiveness of negative pressure wound therapy/closed incision management in the prevention of post-surgical wound complications: a systematic review and meta-analysis. JBI Database System Rev Implement Rep. 2015;13(1):253-303.
- Wilkes RP, Kilpad DV, Zhao Y, Kazala R, McNulty A. Closed incision management with negative pressure wound therapy (CIM): biomechanics. Surg Innov. 2012;19(1):67-75.
- Kilpadi DV, Cunningham MR. Evaluation of closed incision management with negative pressure wound therapy (CIM): hematoma/seroma and involvement of the lymphatic system. Wound Repair Regen. 2011;19(5):588-96.
- Gomoll AH, Lin A, Harris MB. Incisional vacuum-assisted closure therapy J Orthop Trauma. 2006;20(10):705-9.
- Erba P, Ogawa R, Ackermann M, Adini A, Miele LF, Dastouri P, et al. Angiogenesis in wounds treated by microdeformational wound therapy. Ann Surg. 2011;253(2):402-9.
- Cirocchi R, Birindelli A, Biffl WL, Mutafchiyski V, Popivanov G, Chiara O, et al. What is the effectiveness of the negative pressure wound therapy (NPWT) in patients treated with open abdomen technique?. J Trauma Acute Care Surg. 2016;81(3):575-84.
- Blackham AU, Farrah JP, McCoy TP, Schmidt BS, Shen P. Prevention of surgical site infections in high-risk patients with laparotomy incisions using negative-pressure therapy. Am J Surg. 2013;205(6):647-54.
- Bonds AM, Novick TK, Dietert JB, Araghizadeh FY, Olson CH. Incisional negative pressure wound therapy significantly reduces surgical site infection in open colorectal surgery. Dis Colon Rectum. 2013;56(12):1403-8.
- Pauli EM, Krpata DM, Novitsky YW, Rosen MJ. Negative pressure therapy for high-risk abdominal wall reconstruction incisions. Surg Infect (Larchmt). 2013;14(3):270-4.
- Matatov T, Reddy KN, Doucet LD, Zhao CX, Zhang WW. Experience with a new negative pressure incision management system in prevention of groin wound infection in vascular surgery patients. J Vasc Surg.. 2013;57(3):791-5.
- Pellino G, Sciaudone G, Candilio G, De Fatico GS, Landino I, Della Corte A, et al. Preventive NPWT over closed incisions in general surgery: Does age matter?. Int J Surg. 2014;12(Supplement 2):S64-S8.
- Selvaggi F, Pellino G, Sciaudone G, Corte AD, Candilio G, Campitiello F, et al. New advances in negative pressure wound therapy (NPWT) for surgical wounds of patients affected with Crohn's disease. Surg Technol Int. 2014;24:83-9.
- Zaidi A, El-Masry S. Closed-incision negative-pressure therapy in high-risk general surgery patients following laparotomy: a retrospective study. Colorectal Dis. 2017;19(3):283-7.
- O'Leary DP, Peirce C, Anglim B, Burton M, Concannon E, Carter M, et al. Prophylactic Negative Pressure Dressing Use in Closed Laparotomy Wounds Following Abdominal Operations: A Randomized, Controlled, Open-label Trial: The P.I.C.O. Trial. Ann Surg. 2017;265(6):1082-6.
- Scalise A, Calamita R, Tartaglione C, Pierangeli M, Bolletta E, Gioacchini M, et al. Improving wound healing and preventing surgical site complications of closed surgical incisions: a possible role of Incisional Negative Pressure Wound Therapy. A systematic review of the literature. Int Wound J. 2016;13(6):1260-81.
- Lynam S, Mark KS, Temkin SM. Primary Placement of Incisional Negative Pressure Wound Therapy at Time of Laparotomy for Gynecologic Malignancies. Int J Gynecol Cancer. 2016;26(8):1525-9.
- Burkhart RA, Javed AA, Ronnekleiv-Kelly S, Wright MJ, Poruk KE, Eckhauser F, et al. The use of negative pressure wound therapy to prevent post-operative surgical site infections following pancreaticoduodenectomy. HPB (Oxford). 2017;19(9):825-31.
- Li PY, Yang D, Liu D, Sun SJ, Zhang LY. Reducing Surgical Site Infection with Negative-Pressure Wound Therapy After Open Abdominal Surgery: A Prospective Randomized Controlled Study. Scand J Surg. 2017;106(3):189-95.
- Willy C, Agarwal A, Andersen CA, Santis G, Gabriel A, Grauhan O, et al. Closed incision negative pressure therapy: international multidisciplinary consensus recommendations. Int Wound J. 2017;14(2):385-98.
- Edwards JR, Peterson KD, Andrus ML, Dudeck MA, Pollock DA, Horan TC. National Healthcare Safety Network (NHSN) Report, data summary for 2006 through 2007, issued November 2008. Am J Infect Control. 2008;36(9):609-26.
- Watanabe A, Kohnoe S, Shimabukuro R, Yamanaka T, Iso Y, Baba H, et al. Risk factors associated with surgical site infection in upper and lower gastrointestinal surgery. Surg Today. 2008;38(5):404-12.
- van Ramshorst GH, Eker HH, Hop WC, Jeekel J, Lange JF. Impact of incisional hernia on health-related quality of life and body image: a prospective cohort study. Am J Surg. 2012;204(2):144-50.
- Di Cristofaro L, Ruffolo C, Pinto E, Massa M, Antoniutti M, Cagol M, et al. Complications after surgery for colorectal cancer affect quality of life and surgeon-patient relationship. Colorectal disease. 2014;16(12):O407-19.
- van Walraven C, Musselman R. The Surgical Site Infection Risk Score (SSIRS): A Model to Predict the Risk of Surgical Site Infections. PloS one. 2013;8(6):e67167.
- Hedrick TL, Sawyer RG, Friel CM, Stukenborg GJ. A method for estimating the risk of surgical site infection in patients with abdominal colorectal procedures. Dis Colon Rectum. 2013;56(5):627-37.
- Rosenberger LH, Politano AD, Sawyer RG. The surgical care improvement project and prevention of post-operative infection, including surgical site infection. Surg Infect (Larchmt). 2011;12(3):163-8.
- Ingraham AM, Richards KE, Hall BL, Ko CY. Quality improvement in surgery: the American College of Surgeons National Surgical Quality Improvement Program approach. Adv Surg. 2010;44:251-67.
- Guillamondegui OD, Gunter OL, Hines L, Martin BJ, Gibson W, Clarke PC, et al. Using the National Surgical Quality Improvement Program and the Tennessee Surgical Quality Collaborative to improve surgical outcomes. J Am Coll Surg. 2012;214(4):709-14;
- Stulberg JJ, Delaney CP, Neuhauser DV, Aron DC, Fu P, Koroukian SM. Adherence to surgical care improvement project measures and the association with postoperative infections. Jama. 2010;303(24):2479-85.
- Pastor C, Artinyan A, Varma MG, Kim E, Gibbs L, Garcia-Aguilar J. An increase in compliance with the Surgical Care Improvement Project measures does not prevent surgical site infection in colorectal surgery. Dis Colon Rectum. 2010;53(1):24-30.
- McHugh SM, Collins CJ, Corrigan MA, Hill AD, Humphreys H. The role of topical antibiotics used as prophylaxis in surgical site infection prevention. J Antimicrob Chemother. 2011;66(4):693-701.
- Suissa D, Danino A, Nikolis A. Negative-pressure therapy versus standard wound care: a meta-analysis of randomized trials. Plast Reconstr Surg. 2011;128(5):498e-503e.
- Semsarzadeh NN, Tadisina KK, Maddox J, Chopra K, Singh DP. Closed Incision Negative-Pressure Therapy Is Associated with Decreased Surgical-Site Infections: A Meta-Analysis. Plast Reconstr Surg. 2015;136(3):592-602.
- Strugala V, Martin R. Meta-Analysis of Comparative Trials Evaluating a Prophylactic Single-Use Negative Pressure Wound Therapy System for the Prevention of Surgical Site Complications. Surg Infect (Larchmt). 2017;18(7):810-9.
- Excellence NIfHaC. PICO negative pressure wound therapy for closed surgical incision wounds 2018.
- Excellence NIfHaC. NICE support for commissioning for surgical site infection 2013.