Research Article
Postoperative Bleeding after Gastric Endoscopic Submucosal Dissection in Patients on Anticoagulant Therapy, Especially Warfarin and DOAC
Tsukasa Furuhata*, Shu Hoteya, Toshiro Iizuka, Akihiro Yamada, Kosuke Nomura, Yasutaka Kuribayashi, Daisuke Kikuchi, Akira Matsui, Satoshi Yamashta and Toshifumi Mitani
*Corresponding author: Tsukasa Furuhata, Departments of Gastroenterology, Toranomon Hospital, 2-2-2 Toranomon, Minato-ku, Tokyo, Japan
Published: 14 Sep, 2018
Cite this article as: Furuhata T, Hoteya S, Iizuka T, Yamada
A, Nomura K, Kuribayashi Y, et al.
Postoperative Bleeding after Gastric
Endoscopic Submucosal Dissection
in Patients on Anticoagulant Therapy,
Especially Warfarin and DOAC. Clin
Surg. 2018; 3: 2102.
Abstract
Background and Aims: It is controversial whether anticoagulant therapy increases the risk of
bleeding after Endoscopic Submucosal Dissection (ESD). The aim of this study is to evaluate the
effects of anticoagulant therapy on gastric ESD.
Methods: Patients who underwent gastric ESD in Toranomon Hospital between April 2005 and
July 2017 were enrolled. The risk of post-ESD bleeding was evaluated using multivariate cox
proportional hazards analysis.
Results: Of 1979 patients enrolled, 49 were taking anticoagulant agent; 25 discontinued warfarin and
switched to heparin alternative therapy during ESD, whereas 24 discontinued DOAC (Direct oral
anticoagulant). During 2 months of the entire observational period, post-ESD bleeding occurred in
88 patients (4.4%): 80 patients without any anticoagulant agent (4.1%), 8 patients with anticoagulant
agent (9.1%); 4 patients with a warfarin (16.0%), 4patients with DOAC (16.7%). In multivariate
analysis, anticoagulant therapy [hazards ratio (HR) 6.24, 95% confidence interval (CI): 2.93–13.27],
warfarin (HR4.19, 95% CI: 2.10–16.62), DOAC (HR6.71, 95% CI: 2.39-18.82) were independent risk
factors of post-ESD bleeding.
Conclusion: Because anticoagulant therapy significantly increases the risk of post-ESD bleeding, it
should be necessary to strictly management at least two weeks after ESD.
Keywords: ESD, Anticoagulant, DOAC, Warfarin, Gastric tumor
Introduction
Endoscopic Submucosal Dissection (ESD) is accepted as a highly curative resection for early
gastric cancer without metastasis. Compared with Endoscopic Mucosal Resection (EMR), ESD has
curative advantages because of the high rate of R0 resection, even in patients with ulceration or
difficult gastric location [1-4]. However, in contrast with its superior curability, ESD is associated
with higher rates of procedure-related complications; this remains an important issue to be resolved.
The rate of post-ESD bleeding in the stomach has been reported to range between 0% and 15.6%
[5-7], which is higher than the rate of post-ESD bleeding in the colon (1.5% to 6.6%) 5 {Terasaki,
2014 #26, [7-9]} and esophagus (0% to 5.2%) [5,10,11]. Previous studies have shown that tumor
location, size, and non-coagulated vessels exposed on the ESD ulcer bed are significantly associated
with a higher rate of post-ESD bleeding in the stomach [12,14-16]. Moreover, our recent study
showed that heparin alternative therapy and multiple antithrombotics significantly increases the
risk of post-ESD bleeding and heparin alternative therapy may not decrease thromboembolic
events. In recent years, there is some report that supports our result [15-17]; on the other hand,
there are a few informative data on the relationship between post-ESD bleeding and DOAC (Direct
oral anticoagulant). Nagata et al. reported that the risk of post endoscopy GI bleeding was higher in
warfarin than DOAC users, however this study did not include information on the lesion location
and specific size, lesion morphology, lesion histopathology, and timing of post-ESD bleeding 15.
Therefore, endoscopists face the practical difficulty of deciding how best to manage patients taking
anticoagulants during the perioperative period of gastric ESD. The aim of the present study was to
evaluate the effects of anticoagulant therapy especially warfarin and DOAC on bleeding after gastric
ESD.
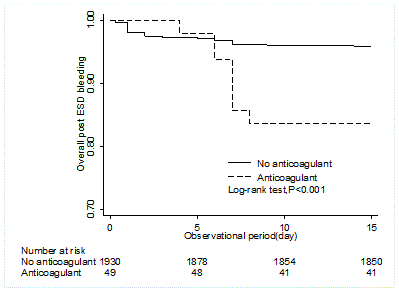
Figure 1
Figure 1
Kaplan-Meier curves for overall post ESD bleeding between patient
with no anticoagulant agent and warfarin.
Methods
Subjects
Patients who underwent gastric ESD in Toranomon Hospital
between April 2005 and July 2017 were enrolled in the present
study, with the exception of patients with a past history of upper
Gastrointestinal (GI) tract surgery, including esophagectomy or
gastrectomy, were except in this study. Patients were also excluded
from the study if ESD was performed simultaneously in two or
more portions of the stomach. Indications for ESD were based on
endoscopic and pathological findings. Patients were also excluded
if the polyglycolic acid sheet sheets were placed over the surgical
wound. To investigate potential risk factors for postoperative
bleeding after ESD, the following variables were analyzed: age, sex,
comorbidities that could affect bleeding (e.g. central neurological
disease, pulmonary disease, cardiovascular disease, renal disease,
hepatic disease, hypertension, and diabetes mellitus), the use of
anticoagulant agents (discontinuation of warfarin and replaced
heparin alternative therapy, discontinued DOAC), the tumor size,
tumor location (upper, middle, or lower one-third of the stomach,
anterior wall, posterior wall, lesser curvature, or greater cuvature of
the stomach), pathological factors (cancer or adenoma, histologic
depth, ulcer presence), and operation time. The indications for ESD
were determined on the basis of endoscopic findings, including
chromo endoscopy with indigo carmine dye, and biopsy. The criteria
for gastric ESD used in Toranomon Hospital are those published by
the Japanese Gastric Cancer Association [18,19]. All patients provided
written informed consent to undergo the proposed procedure. The
study itself was approved by the hospital ethics committee.
ESD procedure
The ESD procedure was performed using a hook knife (KD-
620LR; Olympus Medical Science, Tokyo, Japan), flex knife (KD-
630L; Olympus Medical Science), and dual knife (KD-650Q;
Olympus Medical Science) through a two-channel scope equipped
with multi bending and water jet functions (GIF-2TQ260M;
Olympus Medical Science) or a single-channel endoscope (Q260J
Olympus Medical Science). A soft transparent hood (D-201-13404;
Olympus Medical Science) was attached to the tip of the endoscope to
obtain good, direct endoscopic views of the submucosal layer. Marker
dots were placed on the normal mucosa approximately 5 mm from
the tumor margin to indicate the safety margins. After submucosal
injection of glycerol solution (10% glycerol and 5% fructose; Chugai
Pharmaceutical, Tokyo, Japan) containing a small amount of indigo
carmine and epinephrine, a mucosal incision was made outside the
marker dots. Hyaluronic acid solution was added to the glycerol
solution being injected if mucosal elevation was insufficient due to
ulceration of the lesion or massive fibrosis of the submucosal layer.
After mucosal incision, the submucosal layer was dissected directly
to obtain a perfect specimen, and complete en bloc resection was
performed. Hemostatic forceps (HDB2422W; Pentax, Tokyo, Japan)
in soft coagulation mode were used to control bleeding during the
procedure. In all cases, prophylactic coagulation of visible vessels on
the mucosal defect was performed right after ESD with hemostatic
forceps or with EZ clips (HX-610-090/HX-610-090S/HX-610-135,
Olympus Medical Systems).
ESD was usually performed in conscious patients sedated with a
mixture of diazepam (5 mg to 20 mg) and pethidine hydrochloride
(35–70). However, if the procedure was expected to exceed 2 hr,
patients were administered general anesthesia.
Patients started drinking water and a liquid diet the day after
the ESD procedure if there were no signs of perforation or post-
ESD bleeding. A proton pump inhibitor (PPI) or Vonoprazan was
administered in all patients on the day of ESD, and was regularly
continued for 60 days after ESD. In most cases, second-look
endoscopy was performed on Day 7 after ESD, and hospital discharge
was decided if there was no stigma of bleeding (Forest type I or IIa)
on the artificial ESD ulcer. In cases in which massive intra-procedural
bleeding occurred or was considered as at high risk for post-ESD
bleeding, second-look endoscopy was performed within 3 days after
ESD. If a clinical episode of melena or hematemesis occurred during
the post-ESD period, emergency endoscopy was performed, with
therapeutic endoscopic hemostasis, if necessary, using hemostatic
forceps or hemostatic clips. The method used to achieve hemostasis
was selected on the basis of the bleeding condition. Efforts to achieve
hemostasis were continued until active bleeding had stopped for
several seconds, including after flushing with water, or the visible
vessels had disappeared.
Management of anticoagulant treatment
If patients were being treated with oral anticoagulant agents,
such as warfarin or DOAC (dabigatran, rivaroxaban, apixaban,
or edoxaban), the prescribing doctors were consulted as to how
best to manage anticoagulant therapy during the peri-ESD period.
The patients who were taking warfarin before ESD, warfarin was
discontinued 3 days to 4 days, and treated with intravenous heparin
as an alternative therapy. The patients who were taking DOAC before
ESD, DOAC was discontinued 1day to 2 days, whereas patients at high
risk of cardiovascular events were treated with intravenous heparin
as an alternative only before ESD. Unfractionated heparin was used
as the heparin alternative therapy, and continuous administration
of heparin was initiated and controlled to keep the activated partial
thromboplastin time (aPTT) extended 1.5- to 2.5-fold that of the
control. Heparin was stopped 6 hr before ESD. In patients on warfarin,
heparin was discontinued when the prothrombin time-international
normalized ratio (PT-INR) level reached approximately 1.5–2.5.
DOAC was restarted within 2 days after ESD when hemostasis was
confirmed by the absence of symptoms of gastrointestinal bleeding or
no significant decrease in hemoglobin level.
Definitions of post-ESD bleeding
In the present study, the patient observed for 2 months, post-
ESD bleeding was defined as an episode of hematemesis/melena, or
a decrease in hemoglobin levels (>2 g/dL). All patients meeting these
criteria underwent emergency Esophagogastroduodenoscopy (EGD),
and endoscopic hemostatic procedures were performed when active
bleeding (Forest type I) or stigmata of potential bleeding (Forest type
IIa) was observed. Preventive hemostasis for visible vessels without
the clinical criteria of bleeding at second-look endoscopy was not
included post-ESD bleeding.
Evaluation of resected specimens
Gastric neoplasms were categorized according to location (i.e.
upper, middle, or lower one-third of the stomach, anterior wall,
posterior wall, lesser curvature, or greater cuvature of the stomach).
Tumor size and ulceration were determined histopathologically,
and the size of the resected specimen was measured at its maximum
diameter.
Statistical analysis
Unless indicated otherwise, data are expressed as the mean ± SD.
Mean quantitative values among groups were compared statistically
using analysis of variance followed by the t-test.
These categorical variables were compared statistically using
the Chi-squared test or Fisher’s exact test in univariate analysis. The
effectiveness of anticoagulant on the post ESD bleeding was compared
using the log-rank test, Kaplan-Meier method, univariate and
multivariate cox proportional hazards ratio (HR) and 95% confidence
interval (CI). Two-tailed P<0.05 was considered significant. All data
analyses were conducted using Stata version 14 (Stata Corp, College
Station, TX, USA).
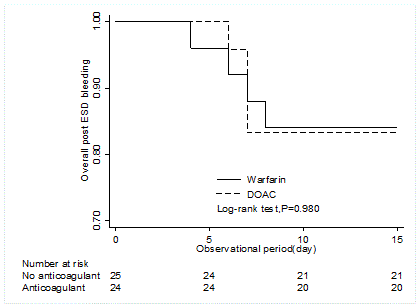
Figure 2
Figure 2
Kaplan-Meier curves for overall post ESD bleeding between patient
with warfarin and DOAC.
*No patient occurred post ESD bleeding aftet 2 weeks of ESD
Table 1
Table 1
Clinicopathologic characteristics of the patients and univariate analysis of the relationship between post-endoscopic submucosal dissection (ESD) bleeding
and various clinicopathologic factors unless indicated otherwise, data are given as the mean ± SD or as the number of patients in each group.
Table 2
Table 2
Clinical characteristics and outcomes of gastric endoscopic submucosal dissection (ESD) according to antithrombotic use unless indicated otherwise, data
are given as the mean ± SD or as the number of patients in each group, with percentages in parentheses. OR, odds ratio; CI, confidence interval; CR, coefficient of
regression.
Results
In all, 1979 patients were enrolled in the study (Table 1 and 2).
Mean patient age was 68.6 years; many patients had comorbidities,
including cardiovascular disease (7.0%) and central neurological
disease (4.4%), which required anticoagulant therapy. Of the 1979
patients, 49 (2.5%) were being treated with anticoagulant agents.
Table 3 provides details of the anticoagulant agents. During the peri-
ESD period, about half of these 49 patients discontinued warfarin and
were switched to heparin during ESD, of the other half of the patients
discontinued DOAC.
During 2 months of the entire observational period, post-ESD
bleeding occurred in 88 patients (4.4%) within 2 weeks after ESD.
The timing of post-ESD bleeding tended to be later in patients on
anticoagulant therapy compared with patients who were not on any
antithrombotic agent (6.5 ± 1.2 days vs. 3.5 ± 3.4 days after ESD, logrank
test: P<0.001). However, the timing of post-ESD bleeding was
equivalent in patients on warfarin compared with patients who were
on DOAC (6.3 ± 1.7 days vs. 6.8 ± 0.5 days after ESD, log-rank test:
P=0.980) (Figure 1 and 2).
Relationships between post-ESD bleeding and various
clinicopathological factors were examined by univariatecox
proportional hazardsanalysis (Table 1). Significant relationships
were identified between post-ESD bleeding and seven factors:
age, anticoagulant use, tumor location (middle and lower onethird
of the stomach), tumor size, the presence of an ulcer in the
tumor, and operation time, whereas age and tumor location in the
middle one-third of the stomach was associated with a low rate of
bleeding. With regard to anticoagulant therapy, warfarin and DOAC
were significantly related to post-ESD bleeding. Multivariate cox
proportional hazardsanalysis was then performed on these factors to
determine which factors remained associated with post-ESD bleeding
(Table 4). Anticoagulant agents, hepatic disease, tumor location in the
lower one-third of the stomach, and operation time were significantly
related to post-ESD bleeding. Of these factors, the HR was higher for
anticoagulant therapy (HR=6.24, 95% CI 2.93–13.27), warfarin and
DOAC were as same significantly related to post-ESD bleeding.
Next, we examined relationships between clinical outcomes of
ESD and subgroups based on anticoagulant type (Table 2). If patients
who were not using anticoagulant agents were set as the reference
group, the HR of post-ESD bleeding were significantly higher for
patients on anticoagulant therapy (HR=3.98, 95% CI 1.92–8.24),
those who discontinued warfarin (HR=4.19, 95% CI 1.53–11.46),
and DOAC (HR=4.14, 95% CI 1.51–11.32). Similarly, the odds ratios
(ORs) of blood transfusion were significantly higher for patients on
anticoagulant therapy (ORs=9.08, 95% CI 1.91–43.19). Furthermore,
the timing of post-ESD bleeding tended to be later in patients on
anticoagulant therapy compared with patients who were not on any
antithrombotic agent (6.5 ± 1.2 days vs. 3.5 ± 3.4 days after ESD;
coefficient of regression (CR) 3.01, 95% CI 0.60–5.43, if patients not on
antithrombotic agents were set as the reference group). The length of
hospital stay was almost 1.5 times longer in patients on anticoagulant
therapy compared with patients not on any antithrombotic agent
(15.6 ± 6.3 vs. 10.0 ± 5.4 days, respectively), especially longer in
patient on heparin alternative therapy (18.3 ± 5.2 days). Perforation
occurred in 34 patients (1.7%). All perforations were cured without
surgery by conservative treatment with intravenous administration
of antibiotics, PPI and by withholding oral intake for a few days.
Thromboembolism and major bleeding other than GI bleeding did
not occur during the peri-ESD period in any patients in this study.
Table 3
Table 4
Table 4
Multivariate analysis of factors related to post-endoscopic submucosal dissection bleeding CI, confidence interval.
Discussion
The present study provides detailed information regarding the
relationship between bleeding after gastric ESD and anticoagulant
therapy in a large cohort of 1979 patient, including 49(2.5%) who
were being treated with anticoagulant agents. Multivariate analysis
identified that anticoagulant increased the risk of post-ESD bleeding.
A possible explanation for this is that anticoagulants, not only
anticoagulant action, which may have anti-platelet activity through
inhibiting the production or direct inhibition of thrombin. Patients
with anticoagulant agent have an antithrombotic effect equivalent to
that of multiple antithrombotic drugs, so the post-ESD bleeding risk
is high.
Compared with patients who were not on anticoagulant therapy,
hemorrhage occurred later (on around Day 7 after ESD) in patients
on anticoagulant therapy. For patient with warfarin, during the switch
from heparin to warfarin, a certain degree of overwrapped action of
these two drugs might cause a stronger antithrombotic effect in a
later period and consequently caused delayed bleeding. The time to
peak onset of the DOAC is 0.5 hr to 4 hr and the half-life is 5 hr to
13 hr, however there are individual differences in coagulability and
renal function, so, and the anticoagulation effect might be differed for
individuals, even if the same amount of DOAC is administered. Since
DOAC was restarted within 2 days after ESD in the present study, it
takes time for blood concentration to equilibrium, so that the post-
ESD bleeding occurred later. Although the number of patients was
limited and could not monitor the blood concentration of the DOAC
in the present study, it could not elucidate about this.
There are several limitations in this study. Firstly, this study is a
retrospective design from a single high volume center of endoscopy,
and thus the results obtained needs to be confirmed by further studied
of prospective type. Another limitation is that we could not evaluate
the relationship between post-ESD bleeding and there starting time of
antithrombotic. The restarting times were too broad to make a clear
threshold or criteria of the restarting time for assessing if it influenced
the risk of post-ESD bleeding.
Conclusion
Perioperative management of anticoagulation is a challenging clinical issue that requires balancing the risk of acute thromboemobolism against perioperative bleeding. Because anticoagulant, especially warfarin and DOAC increases post-ESD bleeding, it should be necessary to strictly management at least two weeks after ESD.
Ethical Standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. Informed consent or substitute for it was obtained from all patients for being included in the study.
References
- Hoteya S, Iizuka T, Kikuchi D, Yahagi N. Benefits of endoscopic submucosal dissection according to size and location of gastric neoplasm, compared with conventional mucosal resection. J Gastroenterol Hepatol. 2009;24(6):1102-6.
- Lian J, Chen S, Zhang Y, Qiu F. A meta-analysis of endoscopic submucosal dissection and EMR for early gastric cancer. Gastrointest Endosc. 2012;76(4):763-70.
- Watanabe K, Ogata S, Kawazoe S, Watanabe K, Koyama T, Kajiwara T, et al. Clinical outcomes of EMR for gastric tumors: historical pilot evaluation between endoscopic submucosal dissection and conventional mucosal resection. Gastrointest Endosc. 2006;63(6):776-82.
- Oka S, Tanaka S, Kaneko I, Mouri R, Hirata M, Kawamura T, et al. Advantage of endoscopic submucosal dissection compared with EMR for early gastric cancer. Gastrointest Endosc. 2006;64(6):877-83.
- Toyonaga T, Man-i M, East JE, Nishino E, Ono W, Hirooka T, et al. 1,635 Endoscopic submucosal dissection cases in the esophagus, stomach, and colorectum: complication rates and long-term outcomes. Surg Endosc. 2013;27:1000-8.
- Oda I, Suzuki H, Nonaka S, Yoshinaga S. Complications of gastric endoscopic submucosal dissection. Dig Endosc. 2013;25(Suppl 1):71-8.
- Saito Y, Uraoka T, Yamaguchi Y, Hotta K, Sakamoto N, Ikematsu H, et al. A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest Endosc. 2010;72(6):1217-25.
- Fujishiro M, Yahagi N, Kakushima N, Kodashima S, Muraki Y, Ono S, et al. Outcomes of endoscopic submucosal dissection for colorectal epithelial neoplasms in 200 consecutive cases. Clin Gastroenterol Hepatol. 2007;5(6):678-83.
- Saito Y, Uraoka T, Matsuda T, Emura F, Ikehara H, Mashimo Y, et al. Endoscopic treatment of large superficial colorectal tumors: a case series of 200 endoscopic submucosal dissections (with video). Gastrointest Endosc. 2007;66(5):966-73.
- Kagemoto K, Oka S, Tanaka S, Miwata T, Urabe Y, Sanomura Y, et al. Clinical outcomes of endoscopic submucosal dissection for superficial Barrett's adenocarcinoma. Gastrointest Endosc. 2014;80(2):239-45.
- Isomoto H, Yamaguchi N, Minami H, Nakao K. Management of complications associated with endoscopic submucosal dissection/ endoscopic mucosal resection for esophageal cancer. Dig Endosc. 2013;25 Suppl 1:29-38.
- Okada K, Yamamoto Y, Kasuga A, Omae M, Kubota M, Hirasawa T, et al. Risk factors for delayed bleeding after endoscopic submucosal dissection for gastric neoplasm. Surg Endosc. 2011;25(1):98-107.
- Yosuke Tsuji, Ken Ohata, Takafumi Ito, Hideyuki Chiba, Tomohiko Ohya, Toshiaki Gunji, et al. Risk factors for bleeding after endoscopic submucosal dissection for gastric lesions. World J Gastroenterol. 2010;16(23):2913-7.
- Takizawa K, Oda I, Gotoda T, Yokoi C, Matsuda T, Saito Y, et al. Routine coagulation of visible vessels may prevent delayed bleeding after endoscopic submucosal dissection--an analysis of risk factors. Endoscopy. 2008;40(3):179-83.
- Nagata N, Yasunaga H, Matsui H, Fushimi K, Watanabe K, Akiyama J, et al. Therapeutic endoscopy-related GI bleeding and thromboembolic events in patients using warfarin or direct oral anticoagulants: results from a large nationwide database analysis. Gut. 2018;67(10):1805-12.
- Jaruvongvanich V, Sempokuya T, Wijarnpreecha K, Ungprasert P. Heparin-Bridging Therapy and Risk of Bleeding After Endoscopic Submucosal Dissection for Gastric Neoplasms: a Meta-Analysis. J Gastrointest Cancer. 2018;49(1):16-20.
- Dong J, Wei K, Deng J, Zhou X, Huang X, Deng M. Effects of antithrombotic therapy on bleeding after endoscopic submucosal dissection. Gastrointest Endosc. 2017;86(5):807-16.