Research Article
Leg Ulcer Treatment using an Innovative Biodegradable Dressing not Requiring Replacements
Ewa Bieniek1*, Karolina Skołucka-Szary2, Andrzej Lewiński1,3 and Sylwester Piaskowski2
1Department of Endocrinology and Metabolic Diseases, Polish Mother’s Memorial Hospital – Research Institute, Poland
2Celther Polska Sp. z o.o. Inwestycyjna 7, Poland
3Department of Endocrinology and Metabolic Diseases, Medical University of Lodz, Poland
*Corresponding author: Ewa Bieniek, Department of Endocrinology and Metabolic Diseases, Polish Mother’s Memorial Hospital – Research Institute, Rzgowska St. No 281/289, 93-338, Lodz, Poland
Published: 16 Aug, 2018
Cite this article as: Bieniek E, Skołucka-Szary K, Lewiński
A, Piaskowski S. Leg Ulcer Treatment
using an Innovative Biodegradable
Dressing not Requiring Replacements.
Clin Surg. 2018; 3: 2071.
Abstract
The aim of this study was to evaluate the efficacy of the DibuCell Active dressing, the fully
biodegradable dressing based on dibutyrylchitin, in the treatment of venous ulcers. The dressing
undergoes full degradation within the wound. It does not require to be replaced (what significantly
reduces the traumatization of newly formed tissues) and the only thing that is needed is to add yet
another layer of the dressing once the previous one has been degraded.
Evaluation of the effectiveness of the dressing was performed based on the analysis of results
obtained during the prospective, multicentre, randomized, single-blinded clinical trial ULCERUS
1/2014. Statistical analysis was performed using data obtained from 137 patients. The control group
was treated with the Biatain Ag dressing, while the study group with both the DibuCell Active and
Biatain Ag dressings.
In the control group, 38.24% of ulcers were healed, while in the study group ‒ 34.78%. Comparing
only the subset of normotensive patients, the study showed statistically significant differences
(p<0.05) in the rate of ulcer healing process between these two groups. The rate of the healing
process of ulcers was higher in the group of patients treated with the DibuCell Active and Biatain
Ag dressings compared to patients treated only with the Biatain Ag dressing.
Adding the investigated dressing to standard therapy results in beneficial acceleration of the
healing process. Addition of the DibuCell Active dressing to standard therapy seems to improve the
treatment efficacy, especially in the case of large and extensive ulcers.
Keywords: Biodegradable dressings; Chitin dibutyrate; Chronic venous insufficiency; Venous ulcers
Introduction
Venous ulcers are especially common in the population of elders [1-3]. The therapies applied in most cases are regarded expensive and of dubious effectiveness. Treatment of ulcers occurring as a consequence of chronic venous insufficiency requires highly specialized and multidisciplinary approach. Increasing knowledge in this area leads to increasingly effective treatment of this common disease that affects 0.3% adult population in the countries of Western Europe [4,5]. The best wound care strategy is based on the TIME framework. It includes the proper tissue preparation, control over the infections of wound, maintaining the proper wound moisture and stimulation of epidermal growth. Back in the sixties of the 20th century, the first generation of dressings had been designed. They were basically the polymer films made of polyethylene, polypropylene or polyester. They were, however, impermeable for moisture. As a result, this feature contributed to accumulation of body fluids upon the wound surface and thus hindered the healing process itself. Subsequently, the second generation of dressings, made of pure polyurethane or carboxymethylcellulose and polyisobutylene (hydrocolloid dressings), was introduced. Their main drawback, adherence to newly formed tissues, lead to tissue damage as the dressing was replaced. The third generation of dressing materials is based on hydrophilic polyurethane films and hydrogels [6]. Moistened hydrogel dressings have better mechanical properties, elasticity and very good biocompatibility [7,8]. Similarly to their predecessors, they also need to be replaced regularly, which brings about considerable, although minimized in relation to older types of dressing, traumatization of newly regenerated tissues. Elimination of this problem became possible following the progress in such scientific fields as biotechnology and chemistry, due to the fact that innovative biodegradable dressing DibuCell Active has been developed. This dressing, unlike the older ones, is enzymatically degraded directly in the wound environment. It does not need to be replaced on regular basis and the only further treatment steps required are to add an additional layer of the dressing once the previous one has been degraded in the wound. This procedure is continued until the wound is completely healed. Lack of the need to replace the dressing considerably facilitates and simplifies the treatment. The healing of ulcerations occurring during the chronic venous insufficiency should also consider the need for systemic administration of antibiotics, analgesics and nutritional preparations, especially in specific situations. An attention is paid to patient's education and secondary prevention including modification of lifestyle. It is also necessary to mention the treatment of all accompanying comorbidities, which may render the treatment of ulcerations more difficult and increase the risk of its eventual recurrence. Cardiovascular diseases (including hypertension) deserve special attention mainly due to their widespread occurrence. Moreover, some medications used in therapeutic management of hypertension have beneficial effects on the healing process of ulcers. This is especially the case of calcium antagonists (such as unarisine and nifedipine). These drugs inhibit the calcium ion channels in vascular smooth muscle cells, abolishing strong and long-lasting contraction and stenosis of small blood vessels. They also inhibit blood platelets aggregation and reveal slight antihistamine effect. What is more, nifedipine is proved to have beneficial effects on the area of ulcer, leading to its faster reduction, what is explained by improved skin circulation [9,10].
Material and Methods
The effectiveness of the DibuCell Active dressing was evaluated
based on the analysis of results obtained during the prospective,
multicentre, randomized, single-blinded clinical trial ULCERUS
1/2014. The dressing is undergoing enzymatic degradation within
the wound. Therefore, there is no need to replace the dressing. It
is to be left in the wound until it becomes completely degraded (by
means of enzymes present physiologically within the wound), and
all that is needed is just mere addition of its new additional layer.
Such application scheme is repeated until the wound is completely
healed. The dressing serves as a scaffold for migrating cells and newly
created tissues. It stabilizes the newly created tissue in early stages of
its development and then undergoes degradation. Biodegradability of
the dressing, determining its uniqueness, results from the properties
of material called chitin dibutyrate (DBC) ‒ a material of which it is
made of. This biopolymer has been developed in the Celther Polska.
DBC is a diester derivative of chitin, a polymer which can be quite
commonly found in the natural environment (fungi, exoskeleton of
insect). Chitin is characterized by biocompatibility, biodegradability,
lack of toxicity, adsorptive properties, and mechanical strength [11-
13] and it positively affects the migration, adhesion, and proliferation
of fibroblasts [14-17]. This in turn may positively affect the key
stages of the healing process, i.e. granulation and epidermal growth.
DBC is a biocompatible polysaccharide enzymatically degrading to
simple sugars naturally occurring in human body [18]. The DibuCell
Active dressing is of highly porous structure, consisting of a network
of intermittent micro channels. Variety of their sizes and open
porosity determines the characteristic structure of the dressing.
This structure ensures an optimally humid environment within the
wound. On the one hand, it allows the excretion excess to be removed
from the wound and, on the other hand, protects the wound from
excessive drying. It also ensures proper gas exchange and acts as a
scaffold for migrating connective tissue cells. In vitro experiments
have proved no propagation of bacteria growth on the investigated
dressing. As required by the ISO 10993 standard, the hereby tested
DibuCell Active dressing has positively passed the biocompatibility
test, in vitro cytotoxicity test, in vivo irritation and sensitization
tests, hemocompatibility studies and tests of genotoxicity as well as
preclinical studies on diabetic mouse model with induced wounds.
The study population consisted of adult subjects with flat and shallow
venous ulcers of 5-50 cm2, without any clinical signs of infection,
located within the lower limb region (between ankle and knee). One
hundred and sixty patients were enrolled in the study and randomized
into two groups: the study group (treated with the DibuCell Active
and Biatain Ag dressings) and the control group (treated only with
the Biatain Ag dressing), both involving 80 patients. The main criteria
for inclusion to the clinical trial ULCERUS 1/2014: the presence of
flat and shallow ulcers, without the clinical features of infection, after
prior surgical or enzymatic cleansing, localized within the lower
limb (between the ankle and the knee), size 5-50cm2; duration of
ulceration not shorter than 6 weeks and not longer than 46 weeks,
the ankle brachial index in the range of 0.8 - 1.1 or the toe pressure
above 50 mmHg. The main exclusion criteria were the following:
ulcers with active infection, contaminated and with a large amount of
exudate; dry wounds, necrobiosis lipoidica diabeticorum, current or
previous cancer disease in the last 5 years (with the exception of preinvasive
cervical cancer or properly treated skin cancers other than
melanoma). Of all 160 subjects enrolled in the study, those patients
for which at least one episode of significant non-compliance with
the study protocol was noted were excluded from the final statistical
analysis. Ultimately, the final analysis was performed upon 68
(49.6%) patients from the control group treated with only the Biatain
Ag dressing and 69 (50.4%) patients from the study group treated
with both the DibuCell Active and Biatain Ag dressings, making
together 137 subjects from both groups (Table 1). In both groups, all
patients were treated according to the TIME strategy and the current
state of the art. The wound dressings were applied on flat and shallow
ulcers following their surgical and/or enzymatic debridement. In the
study group, the Biatain Ag dressing was applied during each visit in
the active phase of the study as a second layer covering the primary
layer of the DibuCell Active dressing. The DibuCell Active dressing
undergoes spontaneous degradation within the wound (the time of
degradation depends, among others, on the amount of exudate).
Where the structure of dressing became obliterated, another layer of
the DibuCell Active dressing was applied with a maximum margin
of 0.5 cm overlapping the previous dressing layer and/or the edges of
the wound. Such application scheme was repeated until the wound
became completely healed or until the end of the study. The Biatain
Ag dressing was replaced regularly, at time intervals recommended
by manufacturer's instructions. In the study group, it was used as the
secondary dressing (not directly in contact with the wound). In the
control group, the Biatain Ag dressing was used as both the primary
and secondary dressing. This study design gave the opportunity to
ensure the same standards of medical care in both groups of patients.
The secondary dressing used in the study group was the same as the
primary and secondary dressings used in the control group. It allowed
showing differences between these two groups. All other components
of the treatment were identical in both groups. Once the dressings
were applied to the limb with ulcer a multilayer compression
system (JOBST Comprifore® 4 layer) was applied. During the stage
of patients’ qualification to the study, which was based on physical
examinations and interview, the ankle-brachial pressure index value
and the USG Doppler examinations of lower limbs were used to
exclude any possible contraindications of the above described scheme
of treatment. At the each visit, a 0.9% NaCl solution and Octenisept
(Schülke) were used in all subjects to clean the wound. All patients
were subjects to interview and physical examinations. The patient's
follow-up period in the study was approx. 11 weeks, during which each
patient took part in 12 visits (plus any additional visits if necessary).
The healing rate and the rate of reduction of ulcer surface area were
analysed. In addition, the fraction of completely healed wounds in
both groups was also assessed. Statistical significance was inferred for
α=0.05 in the case of all statistical tests used in the study. The normal
distribution of random variables was tested using the Shapiro-Wilk
test. The analysis of outliers was made using the Cook’s distance
method. Analysis of primary endpoints: healing rate and reduction
of surface area in time: depending on the variable distribution and its
type, a Student’s t-test or U Mann-Whitney or ANOVA or Kruskal
Wallis tests were performed; percentage of healed ulcers: a percentage
calculation was performed along with a confidence interval in
individual groups along with the chi-square test.
The clinical trial ULCERUS 1/2014 was accepted by the Bioethics
Committee (Approval No. RNN/189/14/KE). The study protocol
was conformed to the ethical guidelines of the 1975 Declaration of
Helsinki. All patients participating in the clinical trial, prior to the
start of the clinical trial, have accepted informed consent, including
the agreement of clinical trial results publication.
Table 1
Figure 1
Figure 2
Figure 2
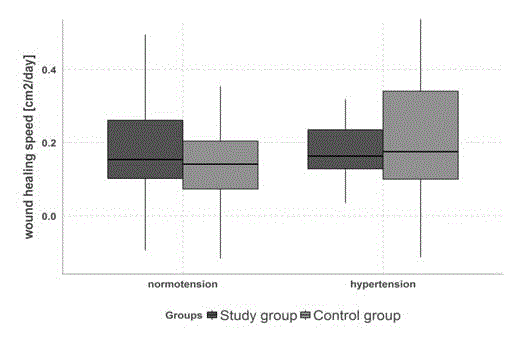
Change of wound healing speed according to level of blood
pressure in study and control group.
Results
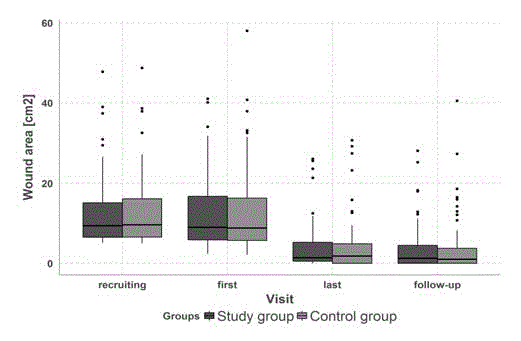
No statistically significant differences in the rate of healing of ulcers, the rate of reduction of their surface area (Figure 1, Table 1), and the fraction of healed ulcers were found between the patients treated with both the DibuCell Active and Biatain Ag dressings and the patients treated with the Biatain Ag dressing only (p>0.05). In the control group, the median rate of healing was 0.157 cm2/day while in the control group it was 0.156 cm2/day. Median change of the wound area between the first and the last visit of the active stage of the study in the control and the study group was 75.5% and 77.9%, respectively. In the control group, 38.24% (95%CI lower bound 26.7%, upper bound 50.8%) of ulcers were healed, while in the study group this fraction reached 34.78% (95% CI lower bound 23.7%, upper bound 47.2%), significance level was 0.67 (Table 3). Statistically significant differences (p<0.05) in the rate of ulcer healing process were, however, found comparing only the subset of normotensive subjects. Among normotensive subjects from the control group, the median rate of ulcer healing was 0.14 cm2/day, while in the study group (with the DibuCell Active dressing included); the healing rate was 0.21 cm2/ day (Figure 2).
Table 2
Table 2
Change of the wound area between the recruiting visit and the follow-up visit in the study and control groups.
Discussion
The treatment of lower leg ulcers, as well as other chronic
wounds, is a difficult and expensive process. This is implied by both
the underlying changes as well as by the general state of patents.
Primary causes that have led to the development of ulcer cannot often
be removed. Therefore, they exert their adverse effects even once the
lesion has been healed, leading often to relapses. One of the ulcer
development risk factors unable to be modified is patient's age. The
incidence of venous pathologies increases with the duration of life.
The problem of varicose veins of the lower limbs concerns 40% of
men and 50% of women at the age of 70 years or older. A similar
relationship is observed in the case of trophic changes of the skin
and ulcers [9]. Elderly people often suffer from the individual multi
morbidity phenomenon. It consists in the increased incidence of
various diseases. Diseases, the incidence of which increases with age
other than those mentioned above, include diabetes, atherosclerosis,
arterial hypertension, cancer, and psychiatric disorders [19]. All these
accompanying diseases should be considered during the treatment
process. The ULCERUS 1/2014 clinical trial, in which the efficacy and
safety of the DibuCell Active dressing was investigated, was conducted
in the elderly population (according to WHO criterion, the early oldage
starts at the age of 60 years), which comprised 70% of all subjects
enrolled in the study. In the analysis, it was thus necessary to take the
effect of the above mentioned accompanying diseases on the healing
process into account.
Even though no significant differences in the rate of the ulcer
healing process were observed when all subjects treated with both
the DibuCell Active and Biatain Ag dressings (i.e. from the study
group) were compared to all subjects from the control group treated
only with the Biatain Ag dressing (Figure 1, Table 3), significant
differences were, however, revealed when only the subset of
normotensive patients from both these groups were compared. The
study showed statistically significant differences (p<0.05) in the rate
of ulcer healing process comparing the normotensive subjects from
the study group to those of the control group (Figure 2). Patients
were divided into two abovementioned subsets (i.e. normotensive
patients vs. hypertensive patients) based on the blood pressure
measurement performed during the first recruiting visit. The goal
of such measurement was to assess the prevalence of hypertension
in the population enrolled in the clinical trial. Hypertension has
a huge effect on the treatment process and general condition of
patients and due to rare occurrence of any symptoms of this disease,
some patients may not be aware of its presence at all. The analysis
was based on the value of arterial pressure measured during the first
recruiting visit, since the arterial pressure values obtained during the
subsequent visits as well as their potential changes (optimization)
may have resulted from started or intensified treatment of arterial
hypertension following its identification during the first visit. Thus,
subjects declaring arterial hypertension comprised 27% of the study
group and 35% of the control one. However, elevated values of arterial
pressure during the first recruiting visit were observed among 42% of
subjects from the study group and 34% of the control one. Subjects
with high arterial pressure values without any treatment comprised
27% of the study group and 23% of the control one, and those treated
for hypertension comprised 17% and 10%, respectively (Table 1).
The study showed statistically significant differences (p<0.05) in
the rate of ulcer healing between the normotensive subjects (blood
pressure measured during the first recruiting visit) from the control
group and respective subset of normotensive subjects (blood pressure
measured at the same time) from the study one (Figure 2). This
leads to the conclusion that the DibuCell Active dressing is more
effective in the group of normotensive patients (blood pressure
<140/90 mmHg). At the same time, the lack of any advantage of the
DibuCell Active/Biatain Ag combined treatment over the treatment
with the Biatain Ag dressing only among patients with hypertension
results most likely from the disproportion in the distribution of
patients in individual groups. As far as the blood pressure analysis
data is concerned, it should be noted that in the control group more
patients were treated for hypertension. In the study group, during
the incidental blood pressure measurement, higher blood pressure
values were found. Thus, there were more patients with suboptimal
treatment in this group. In addition, subjects from the study group
were more frequently unaware of the disease than subjects from
the control group, owing to which they were not treated (Table 1).
Consequently, subjects from the study group may have presented
worse overall condition. Potentially, they may have suffered from
more aggravated atherosclerosis and vascular remodeling resulting
from the adverse effects of hypertension. This may have led to worse
functionality of internal organs and worse blood supply to the skin
resulting in impaired healing process. Patients with hypertension
without any hypertensive treatment also lacked the potentially
beneficial effect exerted by antihypertensive drugs on the process of
ulcer healing. The literature suggests a beneficial effect of calcium
channel blockers on the course of the ulcer healing process, especially
in the case of nifedipine [10,20,21]. This drug inhibits progression
of atherosclerosis and possesses antioxidant properties [22]. This
may improve the skin oxidation and blood supply and accelerate
the ulcer healing process, as a consequence. Moreover, some of the
medications used in the treatment of hypertension are known to
have endothelial and vasoprotective effects, as well. These include
the Angiotensin Converting Enzyme Inhibitors (ACEI), Angiotensin
Receptor Antagonists AT1 (sartans). These drugs, irrespective of their
hypotensive effects, trigger a broad spectrum of endothelial changes
[23]. This may lead to improved blood supply and nourishment of the
skin and acceleration of the ulcer healing process.
In view of the disproportions in the distribution of hypertension,
the disease awareness, blood pressure values and the effect of
medications between the study and control group, it seems apparent
that all of them may have influenced the results of the ULCERUS
1/2014 trial, especially among patients with elevated blood pressure.
Lack of any effect of addition of the DibuCell Active dressing to
standard therapy in the subset of hypertensive patients from the
study group seems to be related to poorer general condition of
these patients. Patients comprising this subset, when compared
to respective counterparts from the control group, presented
higher values of blood pressure, were less likely to be aware of the
disease, more often they were untreated, or their treatment was
suboptimal. As a consequence, the condition of their circulatory
system and especially the condition of their blood vessels was poorer,
possibly contributing to poorer blood supply of the skin, hindering
furthermore the process of healing. At the same time, it should be
noted that both the median as well as the distribution of all results
of hypertensive patients in the study group are better comparing to
respective values of normotensive patients from the control group
(Figure 2). Considering these data, the DibuCell Active dressing
seems to have beneficial effects on the course of the healing process
also in the subset of patients with abnormal blood pressure values.
An objection may be raised against the above described analysis,
that it has been performed based on only one blood pressure
measurement (during the first recruiting visit). The diagnosis of
hypertension under such circumstances is subject to error resulting
from the fact that the group of hypertensive patients may also have
included patients suffering from the so-called white coat hypertension.
It should be emphasized that in the light of current opinions, the
decision to implement an antihypertensive treatment should depend
not only on the value of blood pressure, but also on the presence and
nature of target organ damages, other risk factors as well as other
accompanying comorbidities. Considering the information that the
values of blood pressure obtained during the subsequent visits of the
clinical trial may have been subject to change due to introduced or
optimized antihypertensive treatment, it seems that the selection of
time point for blood pressure values measurement and analysis was
optimal. This study shows that while treating leg ulcers, efforts should
be made to achieve optimal blood pressure values. The inclusion
or optimization of pharmacotherapy may not only translate into
improved general condition of a given patient, but also to improved
time course of the lesion healing process, as well.
In assessing the rate of the healing process, attention should also
be paid to the initial area of the ulcer. During the qualification stage
of the study, the median ulcer area in the control group was 9.63
cm2 (min=5 cm2, max=48.75 cm2), during the first visit, following
the proper cleaning of wounds, the median surface area was 8.81
cm2 (min=2.125 cm2, max=58 cm2). In the study group, the median
ulcer area during the recruiting visit was 9.38 cm2 (min=5.13 cm2,
max=47.75 cm2), during the first visit, following the proper cleaning
of wounds, the median surface area was 9.0 cm2 (min=2.38 cm2,
max=41.0 cm
Moreover, the wounds in the study group, treated with the
DibuCell Active and Biatain Ag dressings, were, in fact, larger and
with lower therapeutic potential therefore more difficult to be treated.
At the end of the study, the median ulcer area in the control group
was 1.81 cm2 (min=0 cm2, max=30.63 cm2) while in the study group
it was 1.38 cm2 (min=0 cm2, max=26.0 cm2) (Table 2). Taking the
above presented results into account as well as the fact that similar
therapeutic effects were achieved in both groups, it seems that adding
the DibuCell Active dressing into standard therapy (especially in the
case of large ulcers) may improve the treatment effectiveness.
Table 3
Conclusion
The scientific progress determining the knowledge and understanding of the pathophysiology of venous ulcers and their healing process, together with the development of biomaterials and biotechnology, provides new therapeutic possibilities. Work that has been done in these fields, resulted in development of the biodegradable dressing made of chitin dibutyrate (DBC) called the DibuCell Active dressing. This dressing undergoes full degradation within the wound environment. There is no need to change the dressing and the only necessary thing is to add its additional layer once the previous one (placed in the wound) has been fully degraded. This minimizes the traumatization of newly created tissues and facilitates and simplifies the treatment process. The ULCERUS 1/2014 clinical trial showed that addition of the DibuCell Active dressing to standard therapy increases the therapeutic efficacy of the treatment of ulcers occurring during the chronic venous insufficiency among normotensive patients when compared to those from the control group and exerts beneficial effects on the rate of the wound healing process in the case of hypertensive patients. Results of this study emphasize the need for control and possible stabilization of blood pressure in the group of patients treated for chronic wounds. Moreover, the addition of the DibuCell Active dressing to standard therapy, especially in the case of large ulcers, may improve the effectiveness of the treatment.
Article Summary
• DibuCell Active undergoes enzymatic degradation within
wound environment; therefore it does not require to be replaced.
• The aim of the study was to evaluate the efficacy of the
DibuCell Active dressing, the fully biodegradable dressing based on
dibutyrylchitin, in the treatment of venous ulcers.
• Addition of the DibuCell Active dressing to standard
therapy seems to improve the treatment efficacy and eliminates the
traumatization of newly formed tissues.
Acknowledgment
Funding: The study was sponsored by Polish Agency for Enterprise Development, Grant No. POIG.01.04.00-10-020/10.
Disclosure
Conflict of interests: Ewa Bieniek- employed as medical
consultant in Celther Polska;
Sylwester Piaskowski- employed as project coordinator and
President in Celther Polska;
Karolina Skołucka-Szary- employed as project coordinator in
Celther Polska.
Author contributions (%)
Ewa Bieniek (40%) - the conception and the design of the study,
acquisition of the data, analysis and interpretation of data, drafting
the article, final approval of the version to be submitted.
Karolina Skołucka-Szary (25%) - the conception and the design
of the study, analysis and interpretation of data, revising the article
critically for important intellectual content, final approval of the
version to be submitted.
Andrzej Lewiński (10%) - analysis and interpretation of data,
revising the article critically for important intellectual content, final
approval of the version to be submitted.
Sylwester Piaskowski (25%) - the conception and the design of the
study, analysis and interpretation of data, revising the article critically
for important intellectual content, final approval of the version to be
submitted.
References
- Marola S, Ferrarese A, Solej M, Enrico S, Nano M, Martino V. Management of venous ulcers: State of the art. Int J Surg. 2016;33:132-4.
- Adeyi A, Muzerengi S, Gupta I. Leg ulcers in older people: a review of management. Br J Med Pract. 2009;2(3):21-23.
- Agale SV. Chronic leg ulcers: epidemiology, aetiopathogenesis, and management. Ulcers. 2013;9.
- Jawień A, Grzela T, Ochwat A. Prevalence of chronic venous insufficiency (CVI) in men and women of Poland. Multicenter cross-sectional study of 40095 patients. Phlebology. 2003;3:110-22.
- Adhikari A, Criqui M, Wooll V, Denenberg JO, Fronek A, Langer RD, et al. The epidemiology of chronic venous diseases. Phlebology. 2000;15(1):2-18.
- Queen D, Orsted H, Sanada H, Sussman G. A dressing history. Int Wound J. 2004;1(1):59-77.
- Singh B, Pal L. Development of sterculia gum based wound dressings for use in drug delivery. Eur Polym J. 2008;44(10):3222-30.
- Zhu J. Bioactive modification of poly(ethylene glycol) hydrogels for tissue engineering. Biomaterials. 2010;31(17):4639-56.
- Jawień A, Szewczyk M, Kaszuba A, Gaciong Z, Krasiński Z, Wroński J, et al. Guidelines for the management of chronic venous leg ulceration. Recommendations of a multidisciplinary expert group [in Polish]. Leczenie Ran. 2011;8:59-80.
- Samy W, Elgindy N, El-Gowelli HM. Biopolymeric nifedipine powder for acceleration of wound healing. Int J Pharm. 2012;422(1-2):323-31.
- Kumar MNVR. A review of chitin and chitosan applications. React Funct Polym. 2000;46(1):1-27.
- Alsarra IA. Chitosan topical gel formulation in the management of burn wounds. Int J Biol Macromol. 2009;45(1):16-21.
- Silva C, Pereira J, Ramalho A, Pais A, Sousa J. Films based on chitosan polyelectrolyte complexes for skin drug delivery: development and characterization. J Memb Sci. 2008;320:268-79.
- Muzzarelli R. Chitins and chitosan for the repair of wound skin, nerve, cartilage and bone. Carbohydr Polym. 2009;76(2):167-82.
- Azuma K, Izumi R, Osaki T, Ifuku S, Morimoto M, Saimoto H, et al. Chitin, chitosan, and its derivatives for wound healing: old and new materials. J Funct Biomater. 2015;6(1):104-42.
- Blasinska A, Drobnik J. Effects of nonwovens mats of di-o-butyrylchitin and related polymers on the process of wound healing. Biomacromolecules. 2008;9(3):776-82.
- Kiekens P, Szosland L, Krucińska I, Schoukens G, Błasińska A, Chilarski A, et al. Novel dibutyrylchitin dressing materials stimulating wound healing. AUTEX 2006 - NC State University - Conference Proceedings. 2006.
- Skołucka-Szary K, Ramięga A, Piaskowska W, Janicki B, Grala M, Rieske P, et al. Synthesis and physicochemical characterization of chitin dihexanoate - a new biocompatible chitin derivative - in comparison to chitin dibutyrate. Mater Sci Eng C Mater Biol Appl. 2016;60:489-502.
- Brettschneider C, Leicht H, Bickel H, Dahlhaus A, Fuchs A, Gensichen J, et al. Relative impact of multimorbid chronic conditions on health-related quality of life – results from the MultiCare Cohort Study. PLoS ONE. 2013;8(6):e66742.
- Agrawal V, Kaushal G, Gupta R. Randomized controlled pilot trial of nifedipine as oral therapy vs. topical application in the treatment of fissure-in-ano. Am J Surg. 2013;206(5):748-751.
- Bhaskar H, Udupa S, Udupa L. Effect of nifedipine and amlodipine on dead space wound healing in rats. Indian J Exp Biol. 2005;43:294-296.
- Hernandez RH, Armas-Hernandez MJ, Velasco M, Israili ZH, Armas-Padilla MC. Calcium antagonists and atherosclerosis protection in hypertension. Am J Ther. 2003;10:409-14.
- Dézsi CA. Differences in the Clinical Effects of Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers: A Critical Review of the Evidence. Am J Cardiovasc Drugs. 2014;14(3):167-73.