Research Article
The Degree of Vascular Leak of Hydroxyethyl Starch in Severe Hemorrhagic Shock in Rats
Takashi Hitosugi1*, Toshiyuki Saito2 and Yoshiyuki Oi2
1Department of Dental Anesthesiology, Kyushu University, Japan
2Department of Anesthesiology, Nihon University Graduate School of Dentistry, Japan
*Corresponding author: Takashi Hitosugi, Department of Dental Anesthesiology, Kyushu University, 3-1- 1 Maidashi, Higashi-ku, Fukuoka city, Fukuoka 812-8582, Japan
Published: 14 Jul, 2018
Cite this article as: Hitosugi T, Saito T, Oi Y. The Degree of
Vascular Leak of Hydroxyethyl Starch
in Severe Hemorrhagic Shock in Rats.
Clin Surg. 2018; 3: 2021.
Abstract
Aim of the Study: In circulatory shock, colloid solution is beneficial to effectively maintain blood
pressure. The HES solution is characterized by its mean Molecular Weight (MW), concentration,
Degree of Substitution (DS) and the pattern of the substitution (C2/C6). To find the ideal character
of the HES to administer to the patients in shock, we studied how the HES is retained in the vascular
vessels in the shock rats using three types of HES bound with Fluorescein-Isothiocyanate (FITCHES)
with a spectrophotometer-computer detection system to determine the rate of elution of the
HES from the microvascules.
Methods: We adopted the model of a severe hemorrhagic shock with a Mean Arterial Pressure
(MAP) of 50 mmHg in rats. We infused one of three types of FITC-HES; HES-A (MW 150-200 kDa,
DS: 0.6-0.68, C2/C6=8), HES-B (MW 175 kDa to 225 kDa, DS: 0.45 to 0.55, C2/C6=6) or HES-C
(MW 550 kDa to 850 kDa, DS: 0.7 to 0.8, C2/C6=5). The FITC-HES retention rates in the image of
the intravital microscope.
Results: MAP transiently increased toward the baseline value after infusion but then progressively
decreased in all groups. HES-C remained in the A2 arteriole longer than HES-A (P=0.022). HES-C
was eluted from the blood vessels more slowly than HES-A (P=0.028). We did not find any statistical
difference in MAP among groups and the rate of disappearance from the vessels between HES-A,
HES-B and HES-C.
Conclusion: HES of high MW was retained in the blood vessel longer those HES with low MW. In
the hypovolemia, the retention rate was more dependent to the MW than that in the normovolemia.
Other factors as DS and C2/C6 showed less effect in the severe hypovolemia.
Keywords: Decompensated hemorrhage; Hydroxyethyl starch; Rats; Shock; Intravital microscopy
Introduction
Significance: Hypovolemia in bleeding decreases cardiac output and tissue oxygen supply. Both
the duration and extent of tissue hypoperfusion determine the severity of cellular damage, which
should be kept to a minimum with timely volume substitution.
Background: Critical hemorrhagic shock requires most effective treatment for volume expansion
in a limited time. Previous studies showed that colloid solutions are superior to crystalloid solutions
for the management of blood volume [1-3]. There are hypo-, iso- and hypertonic crystalloids, and
human albumin, dextran, gelatin and Hydroxyethyl Starch (HES) in colloid solutions. HES is a
popular regimen for hypovolemia among the colloids [4]. The pharmacological effects of HES depend
on the number of starch particles in the vascular space, which produce Colloid Osmotic Pressure
(COP). The number of the starch particles relates the velocity of the decay and the elution of the
particle through the vascular wall. Therefore, we think that it is helpful to observe the concentration
of the HES in the blood vessel, and the leakage of the HES to the outside of the blood to assess the
retention of HES in the blood circulation.
The study’s aims: In this study, the model of severe hemorrhagic shock was chosen which
actually occurs in severe critical injuries. We used intravital microscopy to observe the distribution
and concentration of the HES particles. To observe the HES, the HES is bound with Fluorescein-
Isothiocyanate (FITC-HES) to produce chemiluminescence. We adopted three types of HES with
known molecular weight. The fascia in the cremaster muscle of the rat was chosen to observe the
vessels distributing an area of muscular fascia by using intravital
microscopy [5-7]. We could evaluate macromolecular concentration
and leak in the vessels in the cremaster muscle fascia [7-10].
Figure 1
Figure 1
Color image of leaking hydroxyethyl starch particles bound with fluorescein-isothiocyanate (FITC-HES), seen under the intravital microscope over time.
The particular amount of FITC-HES was shown to have leaked out from the capillary blood vessels in the cremaster muscle a few seconds after the FITC-HES
administration.
Methods
The study was approved by the Committee for Ethical Review
of Animal Experiments at Nihon University and was conducted
according to the National Institutes of Health guidelines for the
use of experimental animals. Male Sprague-Dawley rats (5 ± 1 wk
old, weighing 200 ± 10 g) were purchased from Sankyo Laboratory
Co. Ltd (Tokyo, Japan). The rats were maintained under controlled
conditions at a temperature of 23 ± 3oC with a relative humidity of 55
± 15% and a 12:12 h light-dark cycle. They were used for experiments
following acclimatization of at least 14 days to these conditions, and
fasted for 24 h before the experiments, but had free access to water.
We produced Fluorescein Isothiocyanate Conjugated
Hydroxyethyl Starch (FITC-HES) as described by Thomas et al. [11]
from non-commercial HES for experimental use (Ajinomoto Pharm.
Co., Ltd. Japan). We used three types of HES with different characters:
HES-A (MW 150 kDa to 200 kDa, DS 0.6 to 0.68, C2/C6=8:1), HES-B;
MW 175 kDa to 225 kDa, DS 0.45 to 0.55, C2/C6=6:1) and HES-C
(MW 550 kDa to 850 kDa, DS 0.7 and 0.8, C2/C6=5:1). The volume of
distribution of FITC-HES was extrapolated to Time 0 of the natural
logarithms of the fluorescence derived from plasma samples obtained
at the three time points.
The procedure was performed according to a previously
established method, shown to produce hemorrhage shock [12,13].
Briefly, anesthesia was induced with intraperitoneal sodium
pentobarbital (1 g/kg). A venous line was taken at the ventral tail
vein with a 24-gauge needle to provide continuance of anesthesia
with pentobarbital (40 mg/kg/h to 90 mg/kg/h). Normal saline was
administered as the maintenance fluid at the rate of 2.5 ml/kg/h to
5 ml/kg/h. The infusion rate was adjusted to maintain a stable light
plane of anesthesia based on the previous criteria [12]. Animals were
placed on a warming pad to maintain body temperature between 37oC
to 38oC, which was monitored by a rectum thermometer. The trachea
was intubated with a polyethylene catheter (internal diameter: 2.1
mm) to support spontaneous breathing with the room air.
A silicone cannula was inserted into the left carotid artery to allow
continuous monitoring of blood pressure via a pressure transducer
(BP-308 ETI, OMRON Corporation, Tokyo, Japan). The femoral
artery was catheterized for blood withdrawal. For the observation
of intravital microscopy, the cremaster muscle was prepared [5].
When the anesthetized rats were lying with the cremaster flat on the
microscope slide they were allocated into one of three experimental
groups; re-infusion with HES-A, re-infusion with HES-B, or reinfusion
with HES-C, all in saline solution.
In all groups based on the previous criteria as described,
during anesthesia, after the Baseline (BL) readings were obtained,
hemorrhage was induced by rapid withdrawal of blood for 3 to 5
minutes to obtain a Mean Arterial Pressure (MAP) of 50 mmHg
[12]. Additional blood was withdrawn as required to maintain the
blood pressure at 50 mmHg throughout the hypotensive period until
decompensation occurred. Severe shock state was defined as the point
at which blood pressure could no longer be maintained without reinfusion
of previously withdrawn blood. After a 30-min stabilization
period followed. During this period, areas of measurement were
selected randomly to assess microcirculatory variables, and four
points were fixed for observation. The first two points were the points
of 100 μm from the A2 and V2 wall to the outside, who's A2 and V2
were classified according to their order of branching as previously
described [5]. The other two points were inside A2 and V2. The first
sets of measurements (baseline) were recorded. The animals were
randomly allocated to one of three groups, according to the type
of solution used for resuscitation: 6% HES-A (n=20), 6% HES-B
(n=21) and 6% HES-C (n=20) at the 1: 1 ratio of the blood volume
withdrawn. Initial experiments with skeletal muscle were performed
without additional blood or saline resuscitation, and observations
were continued at 5-minute intervals until the animal died. In
subsequent experiments, rats were further resuscitated by return of
previously withdrawn blood and additional crystalloid as 0.9% NaCl
as required maintaining MAP at or near BL values. Observations
were then continued for additional hours or until death occurred.
In the experiment with rats receiving HES, the first 5 animals died
<1 minute before measurements could be made, and we aborted this
protocol.
The brightness of the illumination at the four points was
measured for a 30-min baseline period (t = -30 to 0 min) and for
an experimental period of 120-mins (t=0 to 120 min) in all groups.
The retention of HES was evaluated by the contrasting density of the
brightness of fluorescence on the image stored on a PC.
For the observation of the intravital fluorescent microscopy,
the animal on the warming pad was transferred to the modified
stage of a BX51WI Olympus fluorescent microscope (Olympus
Co. Ltd., Tokyo, Japan). This was equipped with a tungsten lamp
for transmitted light and a mercury arc lamp for epi-illumination
fluorescent light microscopy. A filter cube interposed into the path of
epi-illumination of the cremaster microcirculation. The extravasation
of FITC-HES was measured by determining the changes in integrated
optical intensity by image analysis.
The FITC-HES represented relative changes in retention and
permeability as previously described [9]. In brief, ΔI=1 – (Ii – Io)/
Ii, where ΔI is the change in light intensity; Ii is the light intensity
inside the vessel, and Io is the light intensity outside the vessel. Each
experimental frame was digitized into a 512 × 512 charge- coupled
device (CCD) (DP70; Olympus Co. Ltd., Tokyo, Japan).
We started the infusion of FITC-HES throughout the tail
vein to the blood circulation. FITC-HES are normally retained in
vasculature; therefore, epi-illumination allows the cremaster muscle
microcirculation to be clearly visualized [2]. Images of the preparation
were monitored by using a CCD camera (DP70; Olympus Co. Ltd.,
Tokyo, Japan), displayed on a high-resolution monitor (Olympus
Co. Ltd., Tokyo, Japan), and the images saved to a Hard Disk Drive
(HDD) (Olympus Co. Ltd., Tokyo, Japan) for later off-line analysis.
Four areas of cremaster muscle microcirculation (random
location) were recorded by using epi-illumination to include two
A2 (30-70 μm) arterioles and V2 (35-120 μm) venuls in each animal
[5]. FITC-HES leakage from the A2 and V2 vessels were recorded
every 5 minutes and assessed off-line by using an Olympus personal
computer (Olympus Co. Ltd., Tokyo, Japan) and image analysis
software package (MetaMorph®, Molecular devices, Sunnyvale, CA,
USA). Each of the 3-5 areas selected were intermittently exposed
to fluorescent light for a maximum of 15s at each 5-min interval
to present photodynamic effects [2]. Macromolecular leakage was
assessed based on the contrasting density of the brightness of FITCHES
fluorescence on the image stored on a computer. Permeability
was measured by light intensity taken at two different sites, one within
the vessel and the other adjacent to the vessel, within the same area.
All data are expressed as mean ± SE. One-way analysis of variance
was used to compare the means of different treatments. If significance
was identified, individual comparisons were subsequently made by
Tukey-Kramer test to determine the site of significance within the
data sets. The differences were considered significant when P <0.05 in
use of the two tails of the test.
Figure 2
Figure 2
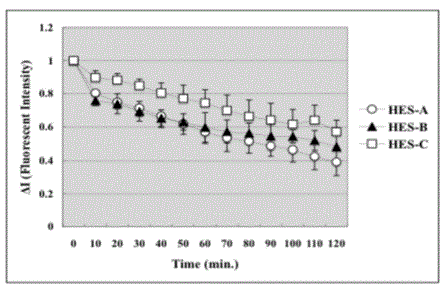
Retention of three groups of HES inside V2 blood vessel. ΔI means
the value of fluorescent intensity at each time point compared to that at time
zero. There was no significant difference among the three groups of the
experiment. Data are presented as means ± SE in each group.
Figure 3
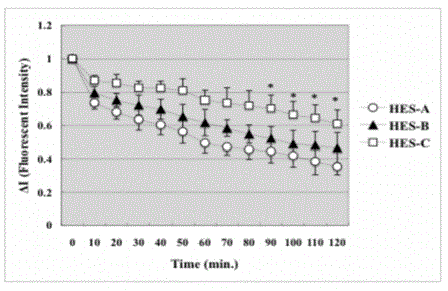
Figure 3
Retention of three groups of HES inside A2 blood vessel. *P <0.05,
significant difference between HES-A and HES-C. ΔI means the value of
fluorescent intensity at each time point compared to that at time zero. Data
are presented as means ± SE in each group.
Figure 4
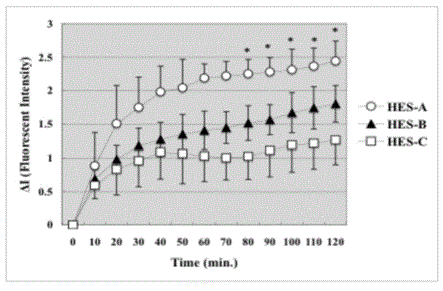
Figure 4
Fluorescent Intensity of HES outside V2. Fluorescent Intensity of
HES-A (HES with the lowest molecular weights) increased the most rapidly
outside the venula. The increase of HES-C was the smallest. The increase
of HES-B was between HES-A and HES-C. *P<0.05 significantly different
between HES-A and HES-C (statistical method ANOVA). Data are presented
as means ± SE for 20 rats in each group.
Results
We used 20 rats in the experiment with HES-A, HES-B and 21 rats
with HES-C. We could successfully observe that FITC-HES leaked
out from the blood vessels. The interstitial space of the cremaseter
muscle became bright after the start of re-infusion. FITC-HES leaked
out from the blood vessels within one minute after the FITC-HES reinfusion
(Figure 1).
The rates of retention of FITC-HES in the arteriolar and venular
blood vessels were different among groups (Figure 2 and 3). Figure 2
is a graphic presentation of the change of the retention of FITC-HES
inside V2 blood vessel after the FITC-HES infusion over a 120-min
period. HES-C was found to have decreased steadily. The fluorescent
intensity of HES-C became two third of that at zero point in two
hours. The retention rates of HES-A, -B and -C at 120 minutes point
were 39% ± 6.8%, 48% ± 8.9% and 57% ± 9.2% of baseline values,
respectively. However, there were no significant differences among
the retention rates of three groups.
The retention rate of FITC-HES inside A2 blood vessel after a
120-min shock period is shown in Figure 3. In the arteriole, too, the
FITC-HES decreased gradually in all groups, although the degrees
were different. HES-A decreased most remarkably in the arteriole. The
retention rates of HES-A, -B and -C at 120 minutes point were 35%
± 10.2%, 46% ± 9.8% and 60% ± 7.2% of baseline values, respectively.
The decrement of fluorescent intensity of HES-C (P=0.038) was
significantly smaller than HES-A. The retention rate of HES-C was
significantly larger than HES-A after 90 minutes point.
The detection of the FITC-HES outside the V2 venules was also
examined (Figure 4). The fluorescent intensity of HES-A outside
the V2 venules increased the most rapidly. Apparently the increase
of HES-C was the smallest, and the increase of HES-B was between
HES-A and HES-C. The increments of fluorescent intensity of HES-A
(P=0.039) was significantly larger than HES-C.
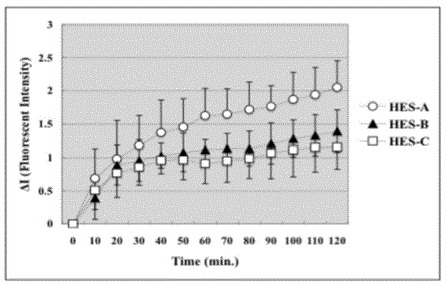
The detection of the FITC-HES outside the A2 arteries is shown
Figure 5. The fluorescent Intensity of HES-A apparently increased the
most rapidly. However, no significant difference was found anywhere
in the figure among the groups in this study.
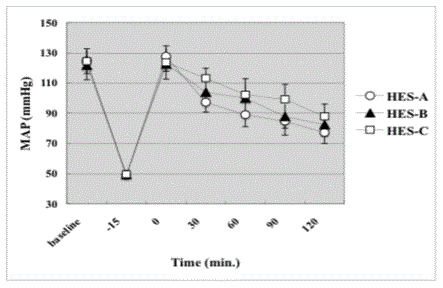
The change of MAP of each group is shown in Figure 6. No
important difference was noted among Baseline (BL) values in groups
receiving HES-A, HES-B and HES-C solution. The blood withdrawal
caused similar decrement of blood pressure in all groups. The blood
withdrawal decreased MAP to 42.3% ± 1% of Baseline (BL) in HES-B
and 40.9% ± 1% of the BL in HES-A and HES-C. After infusion of
HES-A, HES-B and HES-C solution, MAP transiently increased to
the BL value, but then progressively decreased again to about 65% of
BL in all groups. No significant difference was found anywhere in the
Figure between groups.
Figure 5
Figure 5
Fluorescent Intensity of HES outside A2. Fluorescent Intensity of
HES-A outside arteriole increased the most rapidly. The increase of HES-C
was the smallest. The increase of HES-B was between HES-A and HES-C.
However, no significant difference was found anywhere among the groups in
the figure. Data are presented as means ± SE in each group.
Figure 6
Figure 6
The change of Mean Arterial Pressure (MAP). The MAP transiently
increased toward baseline after the start of infusion after the shock. But the
MAP progressively decreased again down to about 65% of the baseline in all
groups. There was no significant difference among three groups at any time
point in the study. Data are presented as means ± SD in each group.
Discussion
Each HES solution is characterized by its concentration, MW, DS
and C2/C6, where DS means the percentage of hydroxyethyl residue
in the glucose subunits, and C2/C6 means the pattern of substitution
of the hydroxyethyl residue [1]. Hydroxyethylation is mandatory
because the natural starches are unstable and are rapidly hydrolyzed
in the circulating amylase. The hydroxyethyl residues, especially
when bound at the C2 carbon position of glucose, hinder the
plasma-amylase to degrade the glucose polymers, hence increasing
the intravascular half-life of the HES particle. As a result, a higher
MW range, extensive DS value and high C2/C6 ratio result in slower
elimination.
In the previous our study in normovolemia, however, the HES
with middle MW with a molecular degradation at a certain rate was
thought to keep the molecular concentration higher, and was retained
within the blood vessel longer than other HES with lesser degradation
in microcirculation. It was not necessary to infuse high MW HES, and
necessary to consider DS and the pattern of the substitution. Another
study showed compared with HES 200/0.5, the use of HES 130/0.4
could significantly improve internal organ perfusion and tissue
oxygenation in patients undergoing liver surgery with a relatively
large amount of blood loss [14]. Two recent meta-analyses concluded
that a goal-directed approach to maintaining tissue perfusion reduces
mortality, postoperative organ failure and surgical complications in
high-risk surgical patients [15,16]. In this study, the elimination of
HES from the blood vessel in hypovolemic condition was elucidated.
Our current result was simple because the larger HES particles could
stay within the blood vessel longer [17,18]. The MW of HES-A and
-B were 175 and 200 kDa on average, respectively. Their DS and the
pattern of the location of hydroxyethyl substitution (C2/C6 ratio)
were 0.64 and 0.5, and 0.5 and 0.6, respectively. In the previous
study, the possible explanation why HES-B was retained within the
vessel space significantly longer was the difference of MW of 2.5 kDa
although it is small, the difference of DS values and the difference
of the C2/C6 ratio [1,3,17-23]. We hypothesized on the fact that the
certain amount of medium sized HES with low DS can be divided
rapidly but at a certain rate so as to keep a constant osmotic pressure in
order to keep it longer in the space. The relatively decreased DS value
may have led to an increased metabolic degradation, which might
have been counteracted by the increased C2/C6 ratio, preventing
a too rapid decrease in the plasma. Anyway, in the previous study
in normovolemia, we found a difference in half lives in the vascular
space between the two HESs of middle MW. Although the reason
should be elucidated in the future, the fact is that we obtained a
result where the HESs was retained in the vessel for a time length
which related to the molecular weight. We can only assume that the
vascular integrity was different between the states in normovolemia
and in hypovolemia [24]. Otherwise, the titer of circulating amylase
would have been diminished by the dilution after the large amount
of rapid re-infusion. Further investigation is required. There are
several types of HES in the world. The properties of HES were very
widely. In all research, HES of high MW showed long term volume
effect. Although the HES of high MW have a beneficial effect, it is also
reported that any HES causes several side effects. There are reports of
tissue storage, plasma accumulation, and renal dysfunction [25-28].
HES of high MW are also reported to produce decreased hemostatic
conditions [29-32]. Large HES with high DS induce significant
increase in prothrombin time and a greater decrease in factor VIII
[13]. In 2013, In the statement from the Co-ordination Group for
Mutual Recognition and Decentralized Procedures–Human (CMDh),
healthcare professionals are informed to consider that ’HES solutions
should be used at the lowest effective dose for the shortest period
of time. Treatment should be guided by continuous hemodynamic
monitoring so that the infusion is stopped as soon as appropriate
hemodynamic goals have been achieved.’ and ‘HES solutions should
only be used for the treatment of hypovolemia due to blood loss when
crystalloids alone are not considered sufficient’ [33]. The therapeutic
target is to achieve normovolemia, not infusing beyond.
To solve the hemostatic derangement, the some studies report on
the new synthesis of a middle sized HES with low SD and high C2/C6
ratio [34-38]. In our study, one of the two compounds of middle sized
HES was retained in the vascular space statistically not different from
HES with high MW. We recognized that middle size HES might exert
a similar volume effect as the large HES does.
Conclusion
The retention of the HES in the blood vessel in the hypovolemia simply depended on its molecular weight. The largest-sized HES of the three stayed in the blood vessel the longest. The middle-sized HES of mean MW 200 kDa with DS 0.5 and C2/C6=6 stayed midway between the other two. The smallest HES of mean MW 175 kDa with DS 0.65 was found to be eliminated the quickest. The retention rate of the middle-sized HES was different in our previous study of normovolemia.
References
- Boldt J, Suttner S. Plasma substitutes. Minerva Anestesiol. 2005;71(12):741-58.
- Jones SB, Whitten CW, Despotis GJ, Monk TG. The influence of crystalloid and colloid replacement solution in acute normovolemic hemodilution; a preliminary survey of hemostatic markers. Anesth Analg. 2003;96(2):363-8.
- Boldt J. Fluid choice for resuscitation of trauma patient: a review of the physiological, pharmacological and clinical evidence. Can J Anaesth. 2004;51(5):500-13.
- Ferreira EL, Terzi RG, Silva WA, de Morraes AC. Early colloid replacement therapy in a near-fatal model of hemorrhagic shock. Anesth Analg. 2005;101(6):1785-91.
- Hutchins PM, Goldstone J, Wells R. Effects of hemorrhagic shock on the microvasculature of skeletal muscle. Microvasc Res. 1973;5(2):131-140.
- Brown NJ, Pollock KJ, Bayjoo P, Reed MW. The effect of cryotherapy on the cremaster muscle microcirculation in vivo. Br J Cancer. 1994;69(4):706-10.
- Brookes ZL, Brown NJ, Reilly CS. Response of the rat cremaster microcirculation to hemorrhage in vivo: differential effects of intravenous anesthetic agents. Shock. 2002;18(6):542-8.
- Miller FN, Joshua IG, Anderson GL. Quantitation of vasodilator-induced macromolecular leakage by in vivo fluorescent microscopy. Microvasc Res. 1982;24(1):56-67.
- Bekker AY, Ritter AB, Duran WN. Analysis of microvascular permeability to macromolecules by video-image digital processing. Microvasc Res. 1989;38(2):200-16.
- Menger MD, Pelikan S, Steiner D, Messmer K. Microvascular ischemia-reperfusion injury in striated muscle: significance of “reflow paradox”. Am J Physiol. 1992;263(6):H1901-6.
- Thomas E, Jones G, de Souza P, Wardrop C, Wusteman F. Measuring blood volume with fluorescent-labeled hydroxyethyl starch. Crit Care Med. 2000;28(3):627-31.
- Cryer HM, Gosche J, Harbrecht J, Anugian G, Garrison N. The effect of hypertonic saline resuscitation on response to severe hemorrhagic shock by the skeletal muscle, intestinal, and renal microcirculation systems: seeing is believing. Am J Surg. 2005;190(2):305-13.
- Hitosugi T, Saito T, Suzuki S, Kubota I, Shoda E, Shimizu T, et al. Hydroxyethyl starch: the effect of molecular weight and degree of substitution on intravascular retention in vivo. Anesth Analg. 2007;105(3):724-8.
- Standl T, Burmeister MA, Schroeder F, Currlin E, Schulte am Esch J, Freitag M, et al. Hydroxyethyl starch (HES) 130/0.4 provides larger and faster increases in tissue oxygen tension in comparison with prehemodilution values than HES 70/0.5 or HES 200/0.5 in volunteers undergoing acute normovolemic hemodilution. Anesth Analg. 2003;96(4):936-43.
- Gurgel ST, do Nascimento P Jr. Maintaining tissue perfusion in high-risk surgical patients: a systematic review of randomized clinical trials. Anesth Analg. 2011;112(6):1384-91.
- Hamilton MA, Cecconi M, Rhodes A. A systematic review and meta-analysis on the use of preemptive hemodynamic intervention to improve postoperative outcomes in moderate and high-risk surgical patients. Anesth Analg. 2011;112(6):1392-402.
- Imm A, Carlson RW. Fluid resuscitation in circulatory shock. Crit Care Clin. 1993;9(2):313-33.
- Funk W, Baldinger V. Microcirculatory perfusion during volume therapy. A comparative study using crystalloid or colloid in awake animals. Anesthesiology. 1995;82(4):975-82.
- Schaeffer Jr RC, Bitrick MS, Holberg III WC, Katz MA. Macromolecular transport across endothelial monolayers. Int J Microcirc Clin Exp. 1992;11(2):181-201.
- Yacobi A, Stoll RG, Sum CY, Lai CM, Gupta SD, Hulse JD. Pharmacokinetics of hydroxyethyl starch in normal subjects. J Clin Pharmacol. 1982;22(4):206-12.
- Persson J, Grande PO. Volume expansion of albumin, gelatin, hydroxyethyl starch, saline and erythrocytes after haemorrhage in the rat. Intensive Care Med. 2005;31(2):296-301.
- James MF, Latoo MY, Mythen MG, Mutch M, Michaelis C, Roche AM, et al. Plasma volume changes associated with two hydroxyethyl starch colloids following acute hypovolemia in volunteers. Anaesthesia. 2004;59(8):738-42.
- Treib J, Haass A, Pindur G, Seyfert UT, Treib W, Graer MT, et al. HES 200/0.5 is not HES HES 200/0.5. Influence of the C2/C6 hydroxyethylation ratio of hydroxyethyl starch (HES) on hemorheology, coagulation and elimination kinetics. Thromb Haemost. 1995;74(6):1452-6.
- Childs EW, Udobi KF, Hunter FA, Dhevan V. Evidence of transcellular albumin transport after hemorrhagic shock. Shock. 2005;23(6):565-70.
- Silk MR. The effect of dextran and hydroxyethyl starch on renal hemodynamics. J Trauma. 1966;6(6):717-23.
- Nohe B, Burchard M, Zanke C, Eichner M, Krump-Konvalinkova V, Kirkpatrick CJ, et al. Endothelial accumulation of hydroxyethyl starch and functional consequence on leukocyte-endothelial interactions. Eur Surg Res. 2002;34(5):364-72.
- Dieterich HJ, Weissmüller T, Rosenberger P, Eltzschig HK. Effect of hydroxyethyl starch on vascular leak syndrome and neutrophil accumulation during hypoxia. Crit Care Med. 2006;34(6):1775-82.
- London MJ, Franks M, Verrier ED, Merrick SH, Levin J, Mangano DT. The safety and efficacy of ten percent pentastarch as a cardiopulmonary bypass priming solution. A randomized clinical trial. J Thorac Cardiovasc Surg. 1992;104(2):284-96.
- Thompson WL, Gadsden RH. Prolonged bleeding times and hypofibrinogenemia in dogs after infusion of hydroxyethyl starch and dextran. Transfusion. 1965;5(5):440-6.
- Villarino ME, Gordon SM, Valdon C, Potts D, Fish K, Uyeda C, et al. A cluster of severe postoperative bleeding following open heart surgery. Infect Control Hosp Epidemiol. 1992;13(5):282-7.
- Claes Y, Van Hemelrijck J, Van Gerven M, Arnout J, Vermylen J, Weidler B, et al. Influence of hydroxyethyl starch on coagulation in patients during the perioperative period. Anesth Analg. 1992;75(1):24-30.
- Strauss RG, Stansfield C, Henriksen RA, Villhauer PJ. Pentastarch may cause fewer effects on coagulation than hetastarch. Transfusion. 1988;28(3):257-60.
- European Medicines Agency. Hydroxethyl starch solutions for infusion. 2013.
- Madjdpour C, Dettori N, Frascarolo P, Burki M, Boll M, Fisch A, et al. Molecular weight of hydroxyethyl starch: is there an effect on blood coagulation and pharmacokinetics? Br J Anaesth. 2005;94(5):569-76.
- Van der Linden PJ, De Hert SG, Deraedt D, Cromheecke S, De Decker K, De Paep R, et al. Hydroxyethyl starch 130/0.4 versus modified fluid gelatin for volume expansion in cardiac surgery patients: the effect on perioperative bleeding and transfusion needs. Anesth Analg. 2005;101(3):629-34.
- Langeron O, Doelberg M, Ang ET, Bonnet F, Capdevilla X, Coriat P. Voluven, a lower substituted novel hydroxyethyl starch (HES 130/0.4), causes fewer effects on coagulation in major orthopedic surgery than HES 200/0.5. Anesth Analg. 2001;92(4):855-62.
- Gallandat Huet RC, Siemons AW, Baus D, van Rooyen-Butijn WT, Haagenaars JA, van Oeveren W, et al. A novel hydroxyethyl starch (Voluven) for effective perioperative plasma volume substitution in cardiac surgery. Can J Anaesth. 2000;47(12):1207-15.
- Von Roten I, Madjdpour C, Frascarolo P, Burmeister MA, Fisch A, Schramm S, et al. Molar substitution and C2/C6 ration of hydroxyethyl starch: influence on blood coagulation. Br J Anaesth. 2006;96(4):455-63.