Research Article
Effectiveness of Decompressive Suboccipital Craniectomy for Cerebellar Infarction
Yoshio Suyama1,2, Shinichi Wakabayashi1, Hiroshi Aihara1, Yusuke Ebiko1, Hiroshi Kajikawa1 and Ichiro Nakahara2*
1Department of Neurosurgery, Suiseikai Kajikawa Hospital, Japan
2Department of Comprehensive Strokology, Fujita Health University School of Medicine, Japan
*Corresponding author: Ichiro Nakahara, Department of Comprehensive Strokology, Fujita Health University School of Medicine, 1-98 Dengakugakubo, Kutsukake, Toyoake, Aichi 470-1192, Japan
Published: 14 Jul, 2018
Cite this article as: Suyama Y, Wakabayashi S, Aihara
H, Ebiko Y, Kajikawa H, Nakahara
I. Effectiveness of Decompressive
Suboccipital Craniectomy for Cerebellar
Infarction. Clin Surg. 2018; 3: 2019.
Abstract
Objective: The adaptation of surgical treatments for cerebellar infarction that have occupied
lesions remains subject to discussion. We investigated effectiveness of Decompressive Suboccipital
Craniectomy (DSC) for cerebellar infarction and poor prognostic factors affecting surgical results.
Materials and Methods: From October 2006 to June 2017, 14 consecutive patients (12 males, 2
females; age, 42–84 years; mean age ± standard deviation, 65 ± 12 years) admitted to our hospital
and underwent DSC under at an admission or clinical course in hospitalization following inclusion
criteria: 1) level of consciousness below Glasgow Coma Scale (GCS) 13 or 2) brainstem compression
and/or obstructive hydrocephalus due to brain edema by cerebellar infarction. Ventricular drainage
was performed simultaneously or later by surgeons’ decision.
Results: After 90 days, 12 of the 14 patients survived (85.7%), 10 (71.4%) were independent (modified
Rankin scale [mRS] ≤ 2) and four (28.6%) were completely dependent or dead. Comparison between
good and poor prognosis demonstrated that the factors affecting prognosis were lesions other than
cerebellar infarction (p<0.01) and obstructive hydrocephalus (p<0.05).
Conclusion: Early DSC for cerebellar infarction may be advisable for cerebellar infarction in
patients with GCS 13 or worse before advancement of hydrocephalus. Poor prognosis is inevitable
in patients causing otherinfarcts other than cerebellum and patients who have already accompanied
obstructive hydrocephalus at the time of surgery.
Keywords: Cerebellar infarction; Decompressive suboccipital Craniectomy; Ventricular drainage; Outcome
Introduction
Cerebellar infarction and associated brain edema due to brainstem compression or obstructive hydrocephalus causes consciousness disturbance. The mortality rate when Decompressive Suboccipital Craniectomy (DSC) is not performed is reported to be 84%. There are reports that DSC is effective, but patient selection and the timing of operation remain unknown. We studied 14 patients who underwent DSC for cerebellar infarction and reviewed the literature on indication, timing of surgical intervention and good prognostic factors [1-3].
Materials and Methods
We studied 14 consecutive patients who underwent DSC for cerebellar infarction between
October 2006 and June 2017 (10 years, 9 months) at our institution. Emergent surgery was indicated
if any of the following were observed at an admission or after hospitalization.
1) Level of consciousness below Glasgow Coma Scale (GCS) 13
2) Brainstem compression and/or obstructive hydrocephalus due to brain edema by cerebellar
infarction
For patients the above indications, we evaluated their general condition to ascertain whether
there was any obstacle to general anesthesia and the prone position for approximately four hours.
Then, after acquiring informed consent from patient’s family, we elected to perform emergency
surgery. A skin incision was added to the median longitudinal incision from 2 cm to 3 cm above
the inion to the fifth cervical vertebra process level, and an approximately 8 cm inverted T-shaped
transverse incision was made on the parieto occipital region. Suboccipital Craniectomy was
performed as far as possible, and laminectomy of the first cervical
vertebra was done as an option based on preoperative images. An
inverted Y-shaped incision was made in the dura mater and internal
decompression, such as removal of infracted brain and hemorrhagic
infarction, was performed. In addition to external decompression,
artificial dura mater made of GORE-TEX membrane was used for
duraplasty. Ventricular Drainage (VD) was performed in cases when
hydrocephalus was observed before surgery or when it was expected
to occur even after DSC by Intraoperative findings and the surgeon
judged it necessary.
We retrospectively evaluated patient age, sex, time from
onset until surgery, infarction at other sites, and pathology of
cerebellar infarction, hemorrhagic infarction, and complication
of hydrocephalus and infarction volume by the patient’s record.
Statistically significant differences between these factors and
prognosis were examined. The volume of the infracted lesion was
measured on the MRI diffusion-weighted image using AZE virtual
place Fujin Raijin 360 (AZE, Tokyo, Japan).Prognosis was evaluated
90 days after onset and expressed according to the modified Rankin
Scale (mRS).
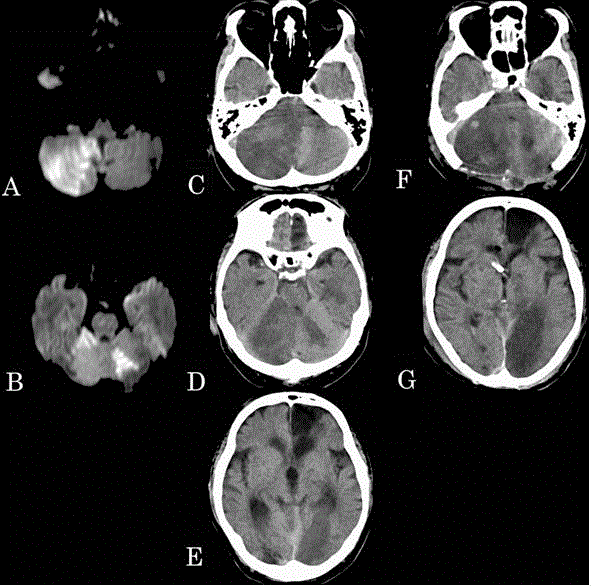
Figure 1
Figure 1
(A, B) Magnetic resonance imaging at admission showed a fresh
infarction in the right posterior inferior cerebellar artery and bilateral superior
cerebellar artery region. (C-E) Computed Tomography (CT) taken 20 hrs after
onset of cerebellar infarction shows compression of the brainstem and acute
obstructive hydrocephalus, as well as cerebral infarction in the occipital lobe.
(F,G) Emergency decompressive suboccipital craniectomy and ventricular
drainage were performed. Although the decompression effect is observed
adequately on CT, the cerebral infarction of the left occipital lobe is clearer.
Results
Table 1 shows the details of 14 patients (age range, 42 years to 84
years; mean ± standard deviation [SD], 65 ± 12; 12 males). Cerebellar
infarction was caused by cardiogenic cerebral embolism in eight
patients, atherothrombotic cerebral infarction in six and unknown
cause in one. Hemorrhagic infarction was found in nine patients.
The Posterior Inferior Cerebellar Artery (PICA), Superior Cerebellar
Artery (SCA) and Anterior Inferior Cerebellar Artery (AICA) were
involved in twelve, five and one patients, respectively. Seven patients
had multiple artery involvement among these three dominant arteries
perfusing the cerebellum. Among these, three patients had infarction
in the Posterior Cerebral Artery (PCA) and Middle Cerebral
Artery (MCA) territories (two and one, respectively). Brainstem
compression findings and/or hydrocephalus was observed on CT or
MRI in all cases before DSC as a result.
The time required from onset to surgery was 16:00 - 157:10
hours: minutes (mean ± SD, 60:30 ± 44:21). The volume of the
infracted lesion immediately before DSC was 33.4 to 104.7 ml3
(mean ± SD, 64.3 ± 19.2 ml3). There were no complications due to
the surgical procedure. Prognostic mRS after 90 days was mRS ≤ 2 in
10 patients (71.4%) who did not require total assistance and mRS ≥
3in four patients (28.6%) who had required total assistance or died.
Severe cerebral infarction of the occipital lobe was the cause of poor
prognosis in two patients (Cases 7 and 12). In addition, two patients
died, one with severe heart failure (Case 5) and one with brainstem
hemorrhage (Case 8). Comparing the good and poor prognosis
groups, there was no significant difference in age, sex, time from
onset to operation, pathology of cerebellar infarction, hemorrhagic
infarction and myocardial infarction. Significant differences were
found between patients with infarction in other areas than cerebellum
and obstructive hydrocephalus combined (Table 2). Representative
examples of poor good prognoses are presented below.
Figure 2
Figure 2
(A) Magnetic resonance imaging at admission shows fresh
infarcts in the left posterior inferior cerebellar artery area. (B,C) Hemorrhagic
cerebellar infarction was confirmed on computed tomography on day five
after onset. The brainstem is compressed and a part of the fourth ventricle
is obscured. (D,E) Emergency decompressive suboccipital craniectomy and
ventricular drainage were performed. Decompression effect is observed
adequately on the computed tomography.
Representative Case Presentations
Case 7: Poor prognosis case
A 67-year-old man presented with dysarthria. Fresh infarction
was observed in the right PICA and SCA, and left SCA region son
MRI at admission (Figure 1A and 1B). Consciousness at arrival to
the hospital was assessed as GCS14. Dysarthria was recognised and
National Institutes of Health Stroke Scale (NIHSS) score was 3 points.
The patient became somnolent on the day after onset, and parenthesis
appeared in the right upper limb. Therefore, immediate head CT
was performed, which confirmed compression of the brainstem by
cerebellar infarction, new cerebral infarction in the left PCA region,
acute obstructive hydrocephalus, in addition to known old infarction
in the left frontal lobe (Figure 1C–1E). Preoperatively, consciousness
was rated as GCS12. Because the therapeutic indication criteria were
met, we performed emergency DSC and VD after obtaining informed
consent (Figure 1F and 1G). After surgery, the patient's conscious
state improved to GCS13. However, cerebral infarction extended to
the left thalamus postoperatively due to the left PCA involvement and
presented severe cognitive impairment. He required full assistance
of activity of daily living and was transferred to a long-term care
hospital; mRS was 5 at discharge from our hospital.
Case 10: Good prognosis case
An84-year-old man with chronic sustained a trial fibrillation
presented to our hospital with nausea and occipital pain.
Consciousness at arrival to the hospital was rated as GCS15. Mild
dysarthria was recognised and NIHSS score was 1 point. MRI showed
fresh infarction in the left PICA area at admission (Figure 2A).
Hemorrhagic cerebellar infarction was confirmed on head CT five
days after onset (Figure 2B and 2C). Consciousness score decreased
to JCS 10 and GCS 13 on day seven after onset. DSC and VD were
performed, and consciousness recovered to GCS 15 postoperatively
(Figure 2D and 2E). Postoperative course was good, VD was removed
a week later, and he was discharged from our hospital with mRS2.
Table 1
Discussion
Discussion continues regarding the adaptation of surgical
operations to cases of cerebellar infarction accompanied by cerebellar
edema and consciousness disorder. The frequency of cerebellar
infarction with cerebellar edema and symptoms is reported to
be 17% to 54% [4]. In addition to cytotoxic edema caused by the
cerebral infarction itself, the mechanism of this condition includes
vasogenic edema due to natural recanalization of occluded vessels,
as well as hemorrhagic changes, direct compression of the brainstem
and upward herniation of the superior vermis cerebelli through the
tentorial notch, downward herniation of the cerebellar tonsils through
the foramen magnum and obstructive hydrocephalus, resulting in
disturbance of consciousness [4-6]. However, surgical treatment for
such patients remains controversial.
In the American Heart Association/American Stroke Association
(AHA/ASA) guideline, surgical treatment for cerebellar infarction
is described as follows: class I, Level of Evidence B, ‘Suboccipital
craniectomy with dural expansion should be performed in patients
with cerebellar infarctions who deteriorate neurologically despite
maximal medical therapy’. On the other hand, according to Guidelines
for Stroke Treatment in Japan 2015, there is sufficient evidence to
perform DSC in cases of cerebral infarction of the unilateral cerebral
hemisphere caused by MCA occlusion(Grade A) [3]. However,
there is insufficient evidence to support surgical treatment for
cerebellar infarction (GradeC1), although many reports have found
that decompression surgery is effective for cerebellar infarction [7].
Feely et al. [1] reported on 55 patients with cerebellar infarction who
became comatose. The mortality rate was 84% when DSC was not
performed, but it was 28% when surgery was performed, and DSC
was reported to be effective [1-3]. Ogasawara et al. [3] reported on
10 patients with cerebellar infarction whose consciousness was
deteriorated. As a result of DSC, seven of the 10 patients had a good
recovery. Tsitsopoulos et al. [2] examined 32 patients who underwent
DSC, and 17(53.1%) had a good prognosis.
The preoperative state of consciousness is reported important as
a factor related to poor prognosis, and DSC improves prognosis in
half of comatose patients or in patients without brainstem infarction.
Regarding age, the density study, which examined broad cerebral
infarction in the MCA region, shows surgical effectiveness in patients
younger than 70 years [2,3,8,9]. However, in patients with cerebellar
infarction, Tsitsopoulos et al. [10] reported that age did not affect
prognosis even if patients were older than 70 years. Regarding the
state of consciousness when deciding on surgery, Ogasawara et al.
[3] reported that a case of deterioration to somnolence surely will
become coma afterwards, and recommended DSC when a level
of consciousness falls down to somnolence. Therefore, in cases of
progressive consciousness disorder, it may be desirable to perform
surgical intervention more quickly. More recently, Recently, Kim
et al. [11] conducted a retrospective-matched case-control study on
efficacy of DSC for patients with cerebellar infarction. As a result,
better clinical outcome was obtained in patients with 1) initial GCS
score >= 9, 2) without clinical deterioration within 72 hr from the
onset, and 3) infarction volume ratio between 0.25 and 0.33 by their
radiological criteria, and 4) no brainstem infarction. However, as
they mentioned, SDC only by volume ratio without deterioration of
consciousness has the risk of refusal of the patient or their family.
Furthermore, the volume ratio is calculated by manual drawing using
brain CT, as they also mentioned as their limitation, in contrast with
our study.
Comparable results as previous reports were obtained in ten
patients out of 14 (71.4%) in our series [1-3]. Factors of the poor
prognosis include combined infarction in other areas than cerebellum
and obstructive hydrocephalus. Regarding the former, more than half
of the causes of cortical infarction are accompanied by persistent a
trial fibrillation. Surgical indication should be withheld in case poor
prognosis is anticipated. Co morbidities such as heart failure may
also define prognosis for the postoperative course as in our case.
Brainstem involvement is another issue that should be discussed.
In our report, the prognosis was poor in a patient with brainstem
bleeding due to hemorrhagic infarction. Some reports mentioned
that combined brainstem infarction as a cause of poor prognosis and
reported that surgical intervention improves prognosis in patients
without brainstem infarction. Another factor affecting prognosis
was obstructive hydrocephalus [2,3,12]. Even though there was no
significant difference in time from onset to surgery, early surgical
intervention may be advisable in patients with signs of hydrocephalus
at the time of somnolence.
As a surgical method, there is no definite conclusion on whether
to perform DSC add VD to hydrocephalus or only perform VD.
Among 42 patients with cerebellar infarction, Rieke et al. [13]
evaluated 20 undergoing conservative treatments, 15 undergoing VD
and 7undergoing DSC. According to their study, VD is recommended
in patients with stupor due to hydrocephalus while DSC should be
performed in comatose cases with brainstem compression. Januss et
al. [14] compared 34 patients undergoing DSC and 14 undergoing
VD among 84 patients with acute cerebellar infarction with mass
effect on head CT, and found no difference between VD and DSC. On
the other hand, severe sequel, such as consciousness disturbance and
hemiplegic, have occurred frequently when only VD is performed.
There also is a report that the level of consciousness worsens even if
only VD is performed for the first time by additional risk for upward
tentorial herniation [3,8,15-17]. Limitations of this study include
its retrospective nature and the small number of patients from a
single facility [18]. The validity of our results should be assessed in a
multicentre study with more cases.
Table 2
Conclusion
We reported on 14 patients undergoing DSC for cerebellar infarction. We suggested that there is a high possibility that a good prognosis will be obtained by DS if there is no infarction other than in the cerebellum and complications of the whole body do not occur in the case of progressive consciousness disturbance.
References
- Feely MP. Cerebellar infarction. Neurosurgery. 1979;4(1):7-11.
- Tsitsopoulos PP, Tobieson L, Enblad P, Marklund N. Clinical outcome following surgical treatment for bilateral cerebellar infarction. Acta Neurol Scand. 2011;123(5):345-51.
- Ogasawara K, Koshu K, Nagamine Y, Fujiwara S, Mizoi K, Yoshimoto T. [Surgical decompression for massive cerebellar infarction]. No Shinkei Geka. 1995;23(1):43-8.
- Jüttler E, Schweickert S, Ringleb PA, Huttner HB, Köhrmann M, Aschoff A. Long-term outcome after surgical treatment for spaceoccupying cerebellar infarction. Experience in 56 patients. Stroke. 2009;40(9):3060-6.
- Hornig CR, Rust DS, Busse O, Jauss M, Laun A. Space occupying cerebellar infarction. Clinical course and prognosis. Stroke. 1994;25(2):372-4.
- Wijdicks EFM, Sheth KN, Carter BS, Greer DM, Kasner SE, Kimberly WT, et al. Recommendations for the management of cerebral and cerebellar infarction with swelling. A statement for healthcare professional from a the American heart association/American stroke association. Stroke. 2014;45(4):1221-38.
- Shinohara Y, Yanagihara T, Abe K. Japanese guideline for management of stroke 2015:Cerebral infarction/ Transient ischemic attack (TIA); Published in issue; July-August, 2011.
- Khan M, Polyzoidis KS, Adegbite ABO, McQueen JD. Massive cerebellar infarction: “Conservative” management. Stroke. 1983;14(5):745-51.
- Juttler E, Schwab S, Schmiedek P, Andreas Unterberg, Michael Hennerici, Johannes Woitzik, et al. Decompressive surgery for the treatment of malignant infarction of the middle cerebral artery (DESTINY). A Randomized, controlled trial. Stroke. 2007;38:2518-25.
- Tsitsopoulos PP, Tobieson L, Enblad P, Marklund N. Clinical outcome following surgical treatment for bilateral cerebellar infarction. Acta Neurol Scand. 2011;123(5):345-51.
- Kim MJ, Park SK, Song J, Oh SY, Lim YC, Sim SY, et al. Preventive Suboccipital Decompressive Craniectomy for cerebellar infarction. A retrospective-matched case-control study. Stroke. 2016;47(10):2565-73.
- Tchopev Z, Hiller M, Zhuo J, Betz J, Gullapalli R, Sheth KN. Prediction of poor outcome in cerebellar infarction by diffusion MRI. Neurocrit Care. 2013;19(3):276-82.
- Rieke K, Krieger D, Adams H, Aschoff AB, Meyding-Lamade UC, Hacke W. Therapeutic strategies in space-occupying cerebellar infarction based on clinical neuroradiological and neurophysiological dat. Cerebroasc Dis. 1993;3:45-55.
- Januss M, Krieger D, Hornig C, Schramm J, Busse O. Surgical and medical management of patients with massive cerebellar infarctions: results of the German-Austrian Cerebellar Infarction Study. J Neurol 1999;246(4):257-64.
- Andoh T, Sakai N, Yamada H, Hattori T, Miwa Y, Hirata T, et al. [Cerebellar infarction: analysis of 33 cases]. No Shinkei Geka. 1990;18(9):821-8.
- Auer LM, Auer T, Sayama I. Indications for surgical treatment of cerebellar haemorrhage and infarction. Acta Neurochir (Wien). 1986;79(2-4):74-9.
- Laun A, Busse O, Calatayud V, Klug N. Cerebellar infarcts in the area of the supply of the PICA and their surgical treatment. Acta Neurochir (Wien). 1984;71(3-4):295-306.
- Hara Y, Hosoda K, Ohta K. Upward transtentorial herniation associated with severe posterior fossa subarachnoid hemorrhage due to vertebral artery dissecting aneurysm: A report of two cases. Japanese journal of neurosurgery. 2007;16(5):423-7.