Review Article
ECMO in Lung Transplantation: A Review
Vanessa M Bazan1 and Joseph B Zwischenberger2*
1Department of Surgery, University of Texas at Austin, USA
2Department of Surgery, University of Kentucky, USA
*Corresponding author: Joseph B Zwischenberger, Department of Surgery, University of Kentucky, 800 Rose Street, MN264, Lexington, Kentucky 40536-0298, USA
Published: 13 Jul, 2018
Cite this article as: Bazan VM, Zwischenberger JB. ECMO
in Lung Transplantation: A Review. Clin
Surg. 2018; 3: 2016.
Abstract
Use of Extracorporeal Membrane Oxygenation (ECMO) for cardiopulmonary support has been rapidly expanding over the last 10 years in the adult population, particularly in lung transplantation. Increased demand for organs has caused longer wait list times leading to increased mortality, prompting the use of ECMO prior to, during, and post lung transplantation. Technological advances in circuit components, improved cannulation techniques, and the development of dual lumen cannulas have improved outcomes of ECMO patients in the setting of lung transplantation.
Introduction
Extracorporeal Membrane Oxygenation (ECMO) uses an extracorporeal circuit consisting of
a drainage cannula, blood pump, oxygenator, and reinfusion cannula to support cardiopulmonary
function in critically ill patients with severe respiratory and/or cardiac failure, unresponsive to
conventional treatment [1]. The use of ECMO in this heterogeneous patient population has produced
a variety of modifications to the ECMO circuit. In1995, the array of ECMO terminology in the
literature led Zwischenberger and Bartlett to introduce the standardizing term “Extracorporeal Life
Support (ECLS)” to encompass all prolonged extracorporeal support techniques [2]. Unfortunately,
the umbrella term ECMO is still universally used.
ECMO is not a curative treatment, but rather temporary life support initiated to optimize
care for critically ill patients expected to recover in weeks to months after receiving supportive or
curative treatment, or lung transplant. ECMO is considered for patients with respiratory and/or
cardiac failure with an estimated 50% mortality risk, but in most cases ECMO is not utilized until a
perceived 80% mortality risk is reached [3]. Mortality risk measurements are patient specific, based
largely on the cause and progression of respiratory and/or cardiac failure, other organ dysfunction,
and age. Patient selection and the risk/benefit analysis should target good stewardship of scarce
donor organ resources.
The only long-term treatment for irreversible end stage respiratory failure is lung transplantation.
Conventional support for end stage respiratory failure is mechanical ventilation until the time of
lung transplantation [4]. If the patient does not achieve sufficient oxygenation and carbon dioxide
removal by mechanical ventilation, ECMO can serve as a bridge to lung transplantation [4]. ECMO
minimizes progressive injury from high pressure and/or volume ventilation, optimizing the clinical
condition of the patient pre-transplantation. ECMO use in lung transplantation has expanded
to include all of the following scenarios: pre-transplantation rescue for acute cardiopulmonary
failure resulting in hypoxia or hypercarbia, a means to facilitate ambulation for pre-transplantation
rehabilitation, intraoperative maintenance of cardiopulmonary support, rescue for post-operative
infection or primary graft dysfunction, and bridge to re-transplantation. The recent expansion of
ECMO applications in lung transplantation has been driven by technological advances in circuit
equipment, development of new cannulation techniques, and emerging evidence supporting the
use of ECMO in adults.
While ECMO has been the standard of care for neonates with severe cardiopulmonary failure
over the past 30 years, ECMO use in adults has endured setbacks. The first successful use of ECMO
in an adult was reported by Hill et al. [5] in 1972, for the treatment of acute respiratory distress
syndrome (ARDS). The patient was placed on ECMO for 3 days and recovered. Five years later,
Vilthet al. [6] reported the first use of ECMO as a bridge to lung transplantation, where a patient
bridged on ECMO underwent bilateral lung transplant, but died 10 days post-transplantation from
infection. In the second reported case of ECMO as a bridge to lung transplantation (1982), the
patient experienced Primary Graft Dysfunction (PGD) after being weaned from ECMO. ECMO was
reinserted as a bridge to re-transplantation but they died due to complications related to tracheostomy
[7]. These early experiences combined with disappointing results from
two randomized control adult ECMO trials dampened enthusiasm
in adults [8,9]. In fact, many considered ECMO a contraindication
to lung transplantation [10]. Ten years later, successful use of adult
ECMO as a bridge to lung transplantation was reported by Jurmann
et al. [11] in 2 patients on mechanical ventilation that developed
PGD and was bridged on ECMO to re-transplantation. Despite this
success, the clinical community remained unconvinced of the value
of adult ECMO.
Adult ECMO was reinvigorated as a form of adult cardiopulmonary
support in 2009, following the publication of the CESAR trial. The
CESAR trial (conventional ventilatory support vs. extracorporeal
membrane oxygenation for severe adult respiratory failure), was a
randomized control trial that included 180 patients in respiratory
failure assigned to either ECMO center referral or conventional
treatment locally. The authors reported 63% of patients in the ECMO
center referral group survived to 6 months compared to only 43% in
the conventional treatment group (relative risk 0.69; 95% CI 0.05–
0.97, p=0.03) [12]. Although the CESAR trial has been criticized for
limitations such as intent-to-treat analysis, proponents argue that the
strengths of the study outweigh its weaknesses, and is considered to
be the sentinel paper on adult ECMO [12,13]. Another driving force
behind the expansion of adult ECMO is the success of ECMO support
in H1N1 influenza patients in 2009-2010. Noah et al. [14] found a
22.7% survival benefit for critically ill, propensity matched, H1N1
patients in the ECMO referral group over the conventional treatment
group (relative risk 0.51; 95% CI 0.31-0.81, p=0.008).
This new evidence supporting the benefits of adult ECMO,
combined with technological advances and improved cannulation
techniques has led to the utilization of ECMO in lung transplantation.
We discuss below the ECMO circuitry, the various ECMO
configurations, and the application of ECMO pre-transplant,
intraoperatively, and post-transplant.
Table 1
The ECMO Circuit
ECMO builds on the technical foundation of Cardiopulmonary Bypass (CPB) [15]. Unlike CPB, ECMO is a closed circuit that can be used pre-, intra-, and post-operatively, supporting cardiopulmonary functions for days to weeks [3]. The ECMO circuit withdraws deoxygenated blood from the venous system through a drainage cannula, pumps the blood through an oxygenator where oxygenation and carbon dioxide removal occurs, returning the blood to venous (veno-venous for gas exchange) or arterial (veno-arterial for cardiopulmonary support) circulation through a reinfusion cannula.
Oxygenators
Early oxygenators exposed blood to gas directly either by agitation or infusion of oxygen bubbles, [16] damaging blood products to limit CPB use to a few hours for cardiac surgery. Development of the membrane oxygenator in the 1960s, created a barrier between the blood and gas, significantly reducing damage to blood products increasing the time that could safely be spent on an extracorporeal circuit [17]. Silicone membrane or polypropylene hollow fiber membrane oxygenators were used from the time the original ECMO circuit was popularized in the 1980s until the early 2000s [18]. Both, however, had significant deficiencies. Silicone membranes had inefficient gas exchange and polypropylene hollow fiber membranes had significant plasma leak. These limitations prompted the development of polymethylpentene (PMP) used in most modern hollow fiber membrane oxygenators. PMP is a gas permeable material which overcomes the deficiencies of inefficient gas exchange and plasma leak encountered when using earlier membrane materials. In addition, PMP oxygenators have low resistance, fast priming, and have been shown to function for weeks before needing to be replaced [19]. Low resistance PMP membranes such the interventional lung assistant, iLA®, or Novalung®, (Xenios, Heilbronn, Germany) are also effective for low flow carbon dioxide removal in patients with end stage hypercapneic respiratory failure awaiting lung transplantation [20].
Cannulas
The ECMO circuit requires a drainage cannula to carry deoxygenated
blood to the oxygenator and a separate reinfusion cannula
to deliver oxygenated blood to the patient. Lung transplantation
candidates are preferentially cannulated to increase ambulation
and reduce risk of complications. Cannulas can be single or double
lumen. Single lumen cannulas require separate insertion sites for the
drainage cannula and the reinfusion cannula. Single lumen cannulas
can be placed using a cut down method in which the vessel is surgically
exposed and the cannula is visually placed. Another cannulation
method combines cut down and Seldinger techniques, using a cut
down to visualize and access the vessel followed by insertion of a
percutaneous guide wire over which serial dilations are performed
followed by cannula placement. A final method involves thoracotomy
for cannulation of the right atrium and aorta. Common cannulation
sites include the femoral vein, internal jugular vein, subclavian vein,
femoral artery, and subclavian artery (Table 1).
In contrast, double lumen cannulas can be utilized for venovenous
ECMO through single central venous access. The OriGen®
dual lumen cannula(DLC) (OriGen Biomedical, Austin, TX), Avalon
Elite® DLC (Maquet Cardiovascular, San Jose, CA) and Nova Port
twin® DLC (Xenios, Heilbronn, Germany)are double lumen cannulas
with both drainage and infusion ports to help separate desaturated
venous drainage from oxygenated reinfused blood returning to the
central venous circulation (Figure 1). Adult DLCs range from 23 to
32 French (OriGen® DLC), 20 to 31 French (Avalon Elite®) and 18 to
24 French (Nova Port twin®), where 3 French is equivalent to 1 mm
diameter [21-23]. Size determination is based on body habitus and
cardiac index (cardiac output divided by body surface area) of the
patient. Right jugular Avalon cannulas are inserted 31 cm into the
central vascular system using the Seldinger technique percutaneously
placed in the vessel using a guide wire and serial dilations under
fluoroscopic and/or echocardiographic insertion guidance [24].
Fluoroscopic guidance is the preferred technique for insertion, but
echocardiography can be used for placement and aid final positioning
[25]. Cannula can be stabilized by suture ligatures (usually two) or
a surrounding Dacron graft sewn to the skin and tied around the
cannula.
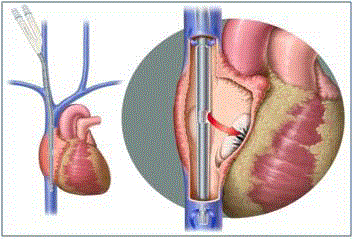
Figure 1
Figure 1
Avalon Elite® is a bicaval dual lumen veno-venous ECMO catheter
inserted via the right internal jugular vein. Cannula drains blood from the
superior and inferior vena cava. Oxygenated blood is returned into the right
atrium via infusion port pointing toward the tricuspid.
Pumps
Roller pumps and centrifugal pumps are currently used in ECMO circuits to control blood flow. There has been a shift away from roller pumps because stress/strain on circuit raceway tubing results in wear and subsequent spallation. The tubing fragments are carried with the blood and can be a source of microemboli [26]. This risk necessitates frequent changing of raceway tubing. Alternatively, centrifugal pumps do not require raceway tubing, eliminating the risks associated with wear. Another advantage of centrifugal pumps is that they are after load sensitive whereas roller pumps are not. This means that if a kink or other occlusion occurs upstream of a roller pump, there is potential for circuit rupture, however, with the after load sensitive centrifugal pump, a tubing rupture is unlikely [27]. In addition, centrifugal pumps do not rely on gravity to drive drainage and are more compact than roller pumps, facilitating ambulatory ECMO.
Heparin Coating
The modern ECMO circuit components can be heparin coated, reducing the anticoagulation dosage required by the patient, potentially reducing the risk of bleeding complications and transfusion requirements [28]. ECMO circuit components interact with all blood components initiating a blood/surface interaction that can either activate or consume the reactive elements [29]. Subsequently, thrombotic complications, or a consumptive coagulopathy, can result. While heparin coated components decrease activation and the risk for these complications, an anticoagulation strategy must be maintained. Heparin coated oxygenators can be used safely for several weeks before irregular flow and clotting require the oxygenator to be exchanged [30].
ECMO Configurations
The four modern ECMO configurations include Veno- Venous (VV), Veno-Arterial (VA), Veno-Arterial-Venous (VAV), and Arterio-Venous (AV). The ECMO configuration for lung transplantation candidates is based on the type of cardiopulmonary failure exhibited by the patient. VV ECMO is used for patients presenting with respiratory failure but do not need circulatory support, while VA ECMO is used for patients that need both respiratory and circulatory support. If the patient presents with severe right heart dysfunction or develops Harlequin syndrome on VA ECMO, VAV ECMO is used. AV ECMO is used when the patient presents with hypercarbic respiratory failure but does not need circulatory support. Extracorporeal Carbon Dioxide Removal (ECCOR) is a low flow VV or AV extracorporeal circuit, designed for carbon dioxide removal only [31].
Veno-Venous (VV) ECMO
Veno-Venous (VV) ECMO provides respiratory support, but
does not provide circulatory support. Blood is drained from the
central venous system, either from the superior or inferior vena
cava, oxygenated, and reintroduced into the right atrium. From
the right atrium, the oxygenated blood flows to the right ventricle
and is pumped by the native heart through the pulmonary artery,
dysfunctional lungs, and pulmonary vein. Blood flow continues to
the left atrium and left ventricle where it is pumped through systemic
circulation.
The VV ECMO configuration can be accomplished through
a variety of cannulation techniques. The cannulation site(s) is/are
chosen according to the needs of the patient. Although a single lumen
or double lumen cannulation can be used, the Avalon Elite DLC is
recommended to maximize ambulation and minimize recirculation
of oxygenated blood through the ECMO circuit before reaching
systemic circulation. The Avalon Elite DLC has been shown to
have only 2% recirculation [20]. If the VV ECMO patient develops
worsening cardiac function the Avalon Elite DLC can be converted
to VAECMO by placing a cannula in the right subclavian artery to
facilitate ambulation or femoral artery if urgent access is required
[32]. Low flow VV ECMO is known as ECCOR and is used when
only CO2 removal is required.
Veno-Arterial (VA) ECMO
Veno-Arterial (VA) ECMO provides respiratory and circulatory
support. Blood is drained from a central vein, oxygenated, and
returned to arterial circulation. For emergent cases, venous blood
is drained from the femoral vein and oxygenated blood is returned
via the femoral artery. This configuration delivers oxygenated blood
to the coronary arteries and cerebral circulation via retrograde flow
to the aortic arch. If cardiac function improves, the increased left
ventricular ejection competes with the retrograde ECMO flow. In
this case, deoxygenated blood from the failing lungs ejected from
the left ventricle perfuses the ascending aorta and only partially and
unpredictably mixes with the retrograde perfused oxygenated ECMO
blood. Consequently, the coronary arteries and cerebral circulation
are supplied variably deoxygenated blood.
When the ascending circulation is desaturated and symptomatic, a
complication of femoral cannulation VA ECMO known as Harlequin
syndrome, North-South syndrome, or Watershed phenomenon
results [33]. VA ECMO patients should be vigilantly monitored
for Harlequin syndrome by gas sampling of the right radial artery
and pulse oximitery of the right upper extremity. In a study of 85
VAECMO patients, 10.6% developed Harlequin syndrome [34]. If a
patient develops Harlequin syndrome, the VA ECMO configuration
can be converted to subclavian artery reinfusion or VAV ECMO
(see below) [35,36]. Another complication of VA ECMO is lower
extremity ischemia occurring when the large femoral artery cannula
inhibits oxygenated blood flow to the lower limb. Many programs
now routinely cannulate and perfuse the distal femoral artery.
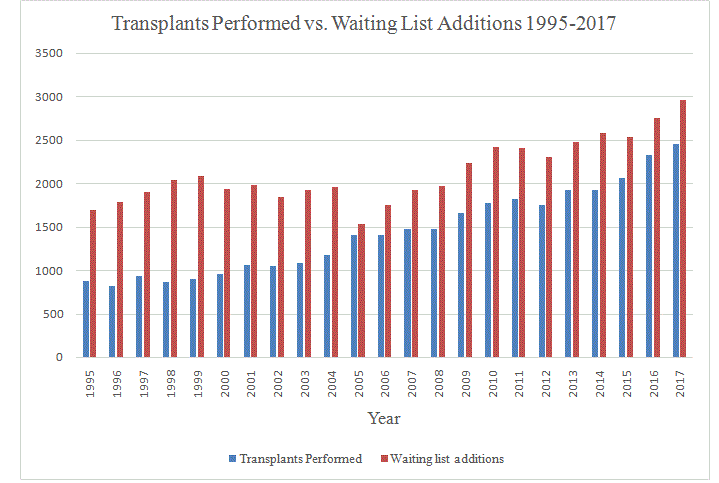
Figure 2
Figure 2
Annual lung transplants performed in the United States versus annual additions to the waiting list according to the Organ Procurement and Transplantation Network. Between 2007 and 2017 waiting list additions exceeded lung transplantation procedures by an average of 538 ± 76 patients.
Veno-Arterial-Venous (VAV) ECMO
Veno-Arterial-Venous (VAV) ECMO provides respiratory and circulatory support. VAV ECMO is used in cases of severe right heart dysfunction. Blood is drained from venous circulation, oxygenated, and returned to the right atrium (VV) and a central artery (VA). The “sport model” developed by Bacchetta and colleagues is the optimal cannulation technique for VAV ECMO. The “sport model” uses a double lumen cannula VV ECMO configuration with an additional oxygenated blood return cannula in the right subclavian artery [36]. For example, an Avalon Elite DLC is inserted into the right internal jugular vein where deoxygenated blood is drained from the inferior vena cava and superior vena cava through the drainage lumen of the DLC. The oxygenated blood is then directed toward the tricuspid valve through the reinfusion lumen of the DLC. This cannulation alone is a VV ECMO configuration. An additional cannula is placed in the right subclavian artery to supply oxygenated blood to the aortic arch. The VAV technique allows cardiac and pulmonary resuscitation with total gas exchange of perfused oxygenated blood to the proximal and distal aortic arch. As the patient improves, VAV allows weaning from VA to VV ECMO and ambulation for pre-transplantation rehabilitation [35-38]. Due to these options and advantages, we encourage VAV when practical.
Artereo-Venous (AV) ECMO (AVCO2R)
Arterio-Venous (AV) ECMO in lung transplantation candidates is used for carbon dioxide removal in hypercarbic respiratory failure. This may occur in patients with Chronic Obstructive Pulmonary Disease (COPD) awaiting lung transplantation. This form of AV ECMO is known as Arterio-Venous Carbon Dioxide Removal (AVCO2R). In AVCO2R, oxygenated blood is drained from a systemic artery (usually femoral), flows through an interventional lung assistant, iLA® or Novalung®, or other low resistance oxygenator such as Quadrox D (Maquet, Rastatt, Germany), and is returned to venous circulation via a central vein. AVCO2R paired with continuous positive airway pressure offers an alternative to intubation for COPD patients [39]. Low flow VV ECMO techniques such as ECCOR, discussed above, have largely replaced AVCO2R to avoid femoral artery cannulation.
ECMO as a Bridge to Lung Transplantation
Increased demands for donor lungs and increased time on the
waiting list have prompted the use of ECMO as a bridge to lung
transplantation [40]. According to data from the Organ Procurement
and Transplantation Registry, every year for the past 10 years,
annual additions to the waiting list for a donor lung in the United
States have exceeded lung transplantation procedures by an average
of 538 patients. This discrepancy between donors and recipients has
remained unacceptably high, with waiting list additions exceeding
lung transplantation procedures by at least 400 patients annually
since 2007 and climbing as high as 658 patients in 2014 (Figure 2).
Recent changes in donor lung allocations strategies have not relieved
donor organ shortages [41].
One-year survival of patients bridged to lung transplantation on
ECMO has been reported from 50% to 90%, with retrospective, singlecenter,
observational study designs accounting for the wide reported
range [4]. ECMO is traditionally used as a salvage therapy for patients
who fail to reach adequate gas exchange on Mechanical Ventilation
(MV) [42]. Patient deconditioning from prolonged sedation on MV
is associated with poor outcomes [43]. Advancements in cannulation
techniques and miniaturization of circuit components have
eliminated the need for sedation-necessitating intubation on ECMO
[44]. Awake ECMO patients are spontaneously breathing and able
to participate in pre-transplantation rehabilitation. Awake ECMO is
currently the preferred support to bridge to lung transplantation, and
is associated with a 30% 6-month post-operative survival benefit to
MV [43,45]. Improvement while on awake ECMO, allows weaning
and decannulation with transition to low pressure MV. Further
improvement results in extubation [46].
Intraoperative ECMO
CMO is gaining acceptance as an alternative to CPB for intraoperative cardiopulmonary support [47]. Likewise, patients bridged to lung transplantation on ECMO should continue ECMO support intraoperatively [48,49]. Advantages of intraoperative ECMO include ease of transition (since VV ECMO can be converted to VAECMO during or after lung transplantation for deteriorating cardiac function) and potentially fewer days in the Intensive Care Unit (ICU) [32,50,51]. While reports of long term survival are conflicting, a recent meta-analysis by Hoechter et al. [52] found that intraoperative ECMO had better 3-month and 1-year survival outcomes compared to CPB, however the benefit failed to reach significance.
Weaning from ECMO Post-Transplantation
Patients maintained on ECMO intraoperatively should be weaned from ECMO post lung transplantation as soon as gas exchange allows. Extracorporeal support should be gradually decreased until extracorporeal support has been lowered to 30%. The ability of the lungs and/or heart to function on their own must be tested by conducting a “trial off.” A trial off for VV ECMO consists of adjusting MV to post-ECMO settings, capping the oxygenator, and halting the sweep gas. If lungs function for one or more hours, ECMO support can be removed. Likewise, a VA ECMO trial off evaluates lung function and perfusion by mimicking the post-ECMO state (protective ventilator settings and clamped circuit flow). Decannulation should take place 30 min to one hour after heparin delivery has been halted [3]. The patient should be placed on protective mechanical ventilation throughout the weaning process and extubated as early as possible. The goal of protective mechanical ventilation, which includes low tidal volume (VT less than or equal to 6 ml/kg) and Positive End Expiratory Pressure (PEEP) targeted towards alveolar opening pressures, should be balanced with the need to maintain adequate gas exchange and perfusion to the newly transplanted lung [53,54].
ECMO Post Lung Transplantation
ECMO initiation post lung transplantation is indicated for patients who are not responding to conventional treatment, requiring ventilator peak inspiratory pressures greater than 35 cmH2O or FiO2 greater than 60% [55]. Timing of ECMO initiation is influential on patient outcomes. Initiation of ECMO within 48 hours of transplantation improved outcomes for patients suffering from Primary Graft Dysfunction (PGD) post-transplantation [56- 58]. Additionally, the perfusion control provided by ECMO along with non-aggressive ventilation has been shown to benefit the transplanted lung [59]. If ECMO is initiated post transplantation, the right heart must receive an adequate supply of oxygenated blood so the transplanted lungs receive adequate perfusion and oxygenation. For this reason, VAV ECMO is the recommended configuration in this scenario.
Complications
The most frequent complication of adult ECMO is hemorrhage associated with cannula insertion. Bleeding complications arise from the systemic anticoagulation required to maintain circuit patency. Perforation of the vessel during cannula placement can lead to retroperitoneal, intrathoracic, or abdominal hemorrhage [60]. In a single center study of 2000 ECMO cases (pediatric and adult) spanning 40 years (1972-2010), Gray and colleagues reported bleeding complications in 39% of patients [61]. Surprisingly, 8% of patients experienced intracranial bleeding, more than double the average rate according Extracorporeal Life Support Organization (ELSO) registry data for adults on ECMO support from 1991-2015 [60,62]. The second most frequent complication was renal dysfunction, reported in 31% of patients [60]. Primary kidney injury is usually associated with inadequate perfusion to central organs, with an inflammatory response related to the ECMO circuit thought to contribute [63]. Air embolus is also a potential complication especially during cannula placement or removal in a spontaneously breathing patient [52].
Patient Selection
Appropriate patient selection is critical in order to minimize risks and maximize benefits of ECMO support. While there are no absolute contraindications to ECMO, individual cases must be evaluated by an interdisciplinary team before initiating ECMO for a lung transplant patient. Relative contraindications to ECMO in adult respiratory failure include mechanical ventilation at high settings (FiO2 >0.9, PPl at >30) for 7 or more days, immunosuppression (absolute neutrophil count less than 400/mm3), recent or expanding CNS hemorrhage, non-recoverable comorbidity such as severe CNS damage or terminal malignancy, and older age [52]. Some centers consider additional relative contraindications including active bloodstream infection, renal failure requiring dialysis, coagulation disorders, and additional end organ failure [64]. A healthcare team consisting of lung transplantation and ECMO specialists should evaluate the clinical status of a patient before ECMO is initiated. Complications such as sepsis or other organ failure may disqualify a patient for transplantation and such risks should be evaluated by the healthcare team and discussed with the patient and family.
Important Considerations
There is no standardized process for the decision to withdraw ECMO as life support as this is highly individualized; however, adequate pain control and sedation should be implemented. The possibility that the patient may need to pursue comfort measures only before the time of lung transplantation should be clearly communicated with the patient and family in order to set appropriate expectations. This discussion should include establishment of clear and reasonable timelines for recovery. Although these timelines vary by center, ECMO support of a patient with lung failure who is not a transplant candidate may be time limited to about 8 weeks of ECMO, if no lung function [52]. Some centers, however, continue indefinitely depending on patient co-morbidities, complications, and estimated potential for recovery.
Editorial
The use of ECMO in lung transplantation has been established as an effective alternative for patients that are not responsive to conventional treatment. Over the past 15 years, the use of ECMO as a rescue for acute deterioration, support for respiratory failure, intraoperative cardiopulmonary support, post-operative PGD, and acute deterioration has been shown to improve outcomes in lung transplantation and save patient lives. Advances in ECMO configurations, such as the dual lumen cannula and “awake” ECMO (spontaneous breathing and no sedation) configurations such as the “sport model” have also improved outcomes for lung transplantation patients. Patient selection, ECMO circuit configuration, and choice of cannulation site should be carefully managed by the healthcare team. Cannulation site selection should facilitate ambulation; with VV ECMO using a dual lumen cannulaifonly respiratory support is needed. If both respiratory and circulatory supports are needed, the “sport model” of VAV ECMO should be implemented. Awareness of common complications is critical for patient success and appropriate stewardship of scarce donor lungs. If the patient is maintained on VA ECMO, monitoring for Harlequin syndrome should be implemented. Bleeding remains the most common complication and this risk must be balanced with the need for anti-coagulation. With these considerations in mind, ECMO has been increasingly recognized for its role in providing lifesaving support for lung transplantation patients.
References
- Squiers JJ, Lima B, DiMaio JM. Contemporary extracorporeal membrane oxygenation therapy in adults: Fundamental principles and systematic review of the evidence. J ThoracCardiovasc Surg. 2016;152:20-32.
- Zwischenberger JB, Bartlett RH. An introduction to extracorporeal life support. In: Zwischenberger JB, Bartlett RH, editors. ECMO Extracorporeal Cardiopulmonary Support in Critical Care. Ann Arbor, MI: Extracorporeal Life Support Organization (ELSO); 1995;11-3.
- Extracorporeal Life Support Organization (ELSO). General guidelines for all ECLS cases. 2017.
- Abrams D, Brodie D, Bacchetta M, Keshavjee S. ECMO as bridge to lung transplantation. In: Brogan T, Lequier L, Lorusso R, MacLaren G, Peek G, editors. Extracorporeal Life Support: The ELSO Red Book. 5th ed. Ann Arbor, MI: Extracorporeal Life Support Organization (ELSO). 2017;639-48.
- Hill JD, O'Brien TG, Murray JJ, Dontigny L, Bramson ML, Osborn JJ, et al. Prolonged extracorporeal oxygenation for acute post-traumatic respiratory failure (shock-lung syndrome). Use of the Bramson membrane lung. N Engl J Med. 1972;286:629-34.
- Veith FJ. Lung transplantation. Transplant Proc. 1977;9(1):203-8.
- [No authors listed]. Sequential bilateral lung transplantation for paraquat poisoning. A case report. The Toronto Lung Transplant group. J Thorac Cardiovasc Surg. 1985;89(5):734-42.
- Zapol WM, Snider MT, Hill JD, Fallat RJ, Bartlett RH, Edmunds LH. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA. 1979;242:2193-6.
- Morris AH, Wallace CJ, Menlove RL, Clemmer TP, Orme JF Jr, Weaver LK. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J RespirCrit Care Med. 1994;149:295-305.
- Mason DP, Thuita L, Nowicki ER, Murthy SC, Pettersson GB, Blackstone EH. Should lung transplantation be performed for patients on mechanical respiratory support? The US experience. J Thorac Cardiovasc Surg. 2010;139(3):765-73.
- Jurmann MJ, Haverich A, Demertzis S, Schaefers HJ, Wagner TO, Borst HG. Extracorporeal membrane oxygenation as a bridge to lung transplantation. Eur J Cardiothorac Surg. 1991;5(2):94-7.
- Peek GJ, Mugford M, Tiruvoipati R, Wilson A, Allen E, Thalanany MM, et al. CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentrerandomised controlled trial. Lancet. 2009;374:1351-63.
- Zwischenberger JB, Lynch JE. Will CESAR answer the adult ECMO debate? Lancet. 2009;374(9698):1307-8.
- Noah MA, Peek GJ, Finney SJ, Griffiths MJ, Harrison DA, Grieve R, et al. Referral to an extracorporeal membrane oxygenation center and mortality among patients with severe 2009 Influenza A(H1N1). JAMA. 2011;306:1659-68.
- Sarkar M, Prabhu V. Basics of cardiopulmonary bypass. Indian J Anaesth. 2017;61(9):760-767.
- Lafç G, Budak AB, Yener AÜ, Cicek OF. Use of extracorporeal membrane oxygenation in adults. Heart Lung Circ. 2014;23(1):10-23.
- Salna M, Bacchetta M. Extracorporeal lung support. Curr Opin Anesthesiol. 2016;29:1-8.
- Peek GJ, Killer HM, Reeves R, Sosnowski AW, Firmin RK. Early experience with a polymethyl pentene oxygenator for adult extracorporeal life support. ASAIO J. 2002;48:480-2.
- Lehle K, Philipp A, Gleich O, Holzamer A, Müller T, Bein T, et al. Efficiency in extracorporeal membrane oxygenation-cellular deposits on polymethylpentene membranes increase resistance to blood flow and reduce gas exchange capacity. ASAIO J. 2008;54:612-7.
- Bartosik W, Egan JJ, Wood AE. The Novalung interventional lung assist as bridge to lung transplantation for self-ventilating patients - initial experience. Interact CardiovascThorac Surg. 2011;13:198-200.
- Vuylsteke A, Brodie D, Combes A, Fowles J-anne, Peek G. Cannulation and decannulation. In: ECMO in the Adult Patient. Core Critical Care. Cambridge: Cambridge University Press; 2017:96-118.
- Kohler K1, Valchanov K, Nias G, Vuylsteke A. ECMO cannula review. Perfusion. 2013;28(2):114-24.
- Lehle K, Philipp A, Müller T, Schettler F, Bein T, Schmid C, et al. Flow dynamics of different adult ECMO systems: a clinical evaluation. Artif Organs. 2014;38(5):391-8.
- Wang D, Zhou X, Liu X, Sidor B, Lynch J, Zwischenberger JB. Wang-Zwische double lumen cannula-toward a percutaneous and ambulatory paracorporeal artificial lung. ASAIO J. 2008;54:606-11.
- Javidfar J, Wang D, Zwischenberger JB, Mongero L, Sonett J, Bacchetta M, et al. Insertion of bicaval dual lumen extracorporeal membrane oxygenation catheter with image guidance. ASAIO J. 2011;57:203-5.
- Peek GJ, Thompson A, Killer HM, Firmin RK. Spallation performance of extracorporeal membrane oxygenation tubing. Perfusion. 2000;15:457-66.
- Shade BC, Schiavo K, Rosenthal T, Connelly JT, Melchior RW. A single center’s conversion from roller pump to centrifugal pump technology in extracorporeal membrane oxygenation. Perfusion. 2016;31:662-7.
- Johnson G, Curry B, Cahalan L, Prater R, Biggerstaff J, Hussain A, et al. Effects of surface-bound and intravenously administered heparin on cell-surface interactions: inflammation and coagulation. Perfusion. 2013;28(3):263-71.
- Wilm J, Philipp A, Müller T, Bredthauer A, Gleich O, Schmid C, et al. Leukocyte Adhesion as an Indicator of Oxygenator Thrombosis During Extracorporeal Membrane Oxygenation Therapy? ASAIO J. 2018;64:24-30.
- Cho HJ, Kim DW, Kim GS, Jeong S. Anticoagulation therapy during extracorporeal membrane oxygenator support in pediatric patients. Chonnam Med J. 2017;53:110-7.
- Pettenuzzo T, Fan E, Del Sorbo L. Extracorporeal carbon dioxide removal in acute exacerbations of chronic obstructive pulmonary disease. Ann Transl Med. 2018;6:31.
- Bacchetta M, Javidfar J, Sonett J, Kim H, Zwischenberger J, Wang D. Ease of conversion from venovenous extracorporeal membrane oxygenation to cardiopulmonary bypass and venoarterial extracorporeal membrane oxygenation with a bicaval dual lumen catheter. ASAIO J. 2011;57:283-5.
- Chung M, Shiloh AL, Carlese A. Monitoring of the adult patient on venoarterial extracorporeal membrane oxygenation. Scientific World J. 2014;2014:393258.
- Cakici M, Gumus F, Ozcinar E, Baran C, Bermede O, Inan MB, et al. Controlled flow diversion in hybrid venoarterial-venous extracorporeal membrane oxygenation. Interact Cardiovasc Thorac Surg. 2018;26(1):112-8.
- Biscotti M, Bacchetta M. The "sport model": extracorporeal membrane oxygenation using the subclavian artery. Ann Thorac Surg. 2014;98:1487-9.
- Biscotti M, Lee A, Basner RC, Agerstrand C, Abrams D, Brodie D, et al. Hybrid configurations via percutaneous access for extracorporeal membrane oxygenation: a single-center experience. ASAIO J. 2014;60:635-42.
- Werner NL, Coughlin M, Cooley E, Haft JW, Hirschl RB, Bartlett RH, et al. The University of Michigan Experience with Veno-Venoarterial Hybrid Mode of Extracorporeal Membrane Oxygenation. ASAIO J. 2016;62:578-83.
- Zhao J, Wang D, Ballard-Croft C, Wang J, Hsu PL, Bacchetta M, et al. Hybrid Extracorporeal Membrane Oxygenation Using Avalon Elite Double Lumen Cannula Ensures Adequate Heart/Brain Oxygen Supply. Ann Thorac Surg. 2017;104:847-53.
- Hayes D, Zwischenberger JB, Zhou X, Liu X, Lynch JE, Ballard-Croft C, et al. Complete respiratory support with AVCO2R and CPAP-mimic ventilation for total gas exchange in sheep. ASAIO J. 2012;58:155-9.
- Gulack BC, Hirji SA, Hartwig MG. Bridge to lung transplantation and rescue post-transplant: the expanding role of extracorporeal membrane oxygenation. J Thorac Dis. 2014;6(8):1070-9.
- Chen H, Shiboski SC, Golden JA, Gould MK, Hays SR, Hoopes CW, et al. Impact of the lung allocation score on lung transplantation for pulmonary arterial hypertension. Am J Respir Crit Care Med. 2009;180(5):468-74.
- Strueber M. Extracorporeal support as a bridge to lung transplantation. Curr Opin Crit Care. 2010;16(1):69-73.
- Fuehner T, Kuehn C, Hadem J, Wiesner O, Gottlieb J, Tudorache I, et al. Extracorporeal membrane oxygenation in awake patients as bridge to lung transplantation. Am J Respir Crit Care Med. 2012;185(7):763-8.
- Mohite PN, Sabashnikov A, Reed A, Saez DG, Patil NP, Popov AF, et al. Extracorporeal Life Support in "Awake" Patients as a Bridge to Lung Transplant. Thorac Cardiovasc Surg. 2015;63(8):699-705.
- Biscotti M, Gannon WD, Agerstrand C, Abrams D, Sonett J, Brodie D, et al. Awake Extracorporeal Membrane Oxygenation as Bridge to Lung Transplantation: A 9-Year Experience. Ann Thorac Surg. 2017;104(2):412-9.
- Biscotti M, Sonett J, Bacchetta M. ECMO as bridge to lung transplant. Thorac Surg Clin. 2015;25(1):17-25.
- Biscotti M, Yang J, Sonett J, Bacchetta M. Comparison of extracorporeal membrane oxygenation versus cardiopulmonary bypass for lung transplantation. J ThoracCardiovasc Surg. 2014;148:2410-5.
- Ius F, Kuehn C, Tudorache I, Sommer W, Avsar M, Boethig D, et al. Lung transplantation on cardiopulmonary support: venoarterial extracorporeal membrane oxygenation outperformed cardiopulmonary bypass. J ThoracCardiovascSurg. 2012;144:1510-6.
- Yu WS, Paik HC, Haam SJ, Lee CY, Nam KS, Jung HS, et al. Transition to routine use of venoarterial extracorporeal oxygenation during lung transplantation could improve early outcomes. J Thorac Dis. 2016;8:1712-20.
- Hoechter DJ, von Dossow V, Winter H, Müller HH, Meiser B, Neurohr C, et al. The Munich Lung Transplant Group: Intraoperative Extracorporeal Circulation in Lung Transplantation. Thorac Cardiovasc Surg. 2015;63:706-14.
- Machuca TN, Collaud S, Mercier O, Cheung M, Cunningham V, Kim SJ, et al. Outcomes of intraoperative extracorporeal membrane oxygenation versus cardiopulmonary bypass for lung transplantation. J Thorac Cardiovasc Surg. 2015;149:1152-7.
- Hoechter DJ, Shen YM, Kammerer T, Günther S, Weig T, Schramm R, et al. Extracorporeal Circulation During Lung Transplantation Procedures: A Meta-Analysis. ASAIO J. 2017;63:551-61.
- Extracorporeal Life Support Organization (ELSO). Guidelines for adult respiratory failure. 2017.
- Acute Respiratory Distress Syndrome Network; Brower RG, Matthay MA, Morris A, Schoenfeld D, Thompson BT, Wheeler A. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301-8.
- Castleberry AW, Hartwig MG, Whitson BA. Extracorporeal membrane oxygenation post lung transplantation. Curr Opin Organ Transplant. 2013;18(5):524-30.
- Hartwig MG, Walczak R, Lin SS, Davis RD. Improved survival but marginal allograft function in patients treated with extracorporeal membrane oxygenation after lung transplantation. Ann ThoracSurg. 2012;93:366-71.
- Diaz-Guzman E, Davenport DL, Zwischenberger JB, Hoopes CW. Lung function and ECMO after lung transplantation. Ann Thorac Surg. 2012;94(2):686-7.
- Oto T, Rosenfeldt F, Rowland M, Pick A, Rabinov M, Preovolos A, et al. Extracorporeal membrane oxygenation after lung transplantation: Evolving technique improves outcomes. Ann Thorac Surg. 2004;78:1230-5.
- Pereszlenyi A, Lang G, Steltzer H, Hetz H, Kocher A, Neuhauser P, et al. Bilateral lung transplantation with intra- and postoperatively prolonged ECMO support in patients with pulmonary hypertension. Eur J Cardiothorac Surg. 2002;21:858-63.
- Makdisi G, Wang I. Extra Corporeal Membrane Oxygenation (ECMO) review of a lifesaving technology. J Thorac Dis. 2015;7:E166-76.
- Gray BW, Haft JW, Hirsch JC, Annich GM, Hirschl RB, Bartlett RH. Extracorporeal life support: experience with 2,000 patients. ASAIO J. 2015;61(1):2-7.
- Camboni D, Schmid C. Neurologic and pulmonary complications in ECLS. In: Brogan T, Lequier L, Lorusso R, MacLaren G, Peek G, editors. Extracorporeal Life Support: The ELSO Red Book. 5th ed. Ann Arbor, MI: Extracorporeal Life Support Organization (ELSO). 2017;575-81.
- Villa G, Katz N, Ronco C. Extracorporeal Membrane Oxygenation and the Kidney. Cardiorenal Med. 2015;6(1):50-60.
- Diaz-Guzman E, Hoopes CW, Zwischenberger JB. The evolution of extracorporeal life support as a bridge to lung transplantation. ASAIO J. 2013;59(1):3-10.