Research Article
Risk Factors for Recurrence after Endovascular Treatment of Saccular Cerebral Aneurysms
Yasuhiro Kawabata1,2*, Takuya Nakazawa3, Naohiro Yonemoto4, Shunichi Fukuda1, Tetsuya Tsukahara1
1Department of Neurosurgery, National Hospital Organization Kyoto Medical Center, Kyoto, Japan
2Department of Neurosurgery, Kyoto Social Welfare Foundation Kyoto Katsura Hospital, Kyoto, Japan
3Department of Neurosurgery, Shiga University of Medical Science Hospital, Shiga, Japan
4Department of biostatistics, Kyoto University School of Public Health, Kyoto, Japan
*Corresponding author: Yasuhiro Kawabata, Department of Neurosurgery, Kyoto Social Welfare Foundation, Kyoto Katsura Hospital, 17 Yamada Hirao-cho, Nishikyo-ku, Kyoto, 615-8256, Japan
Published: 12 Jul, 2018
Cite this article as: Kawabata Y, Nakazawa T, Yonemoto N,
Fukuda S, Tsukahara T. Risk Factors
for Recurrence after Endovascular
Treatment of Saccular Cerebral
Aneurysms. Clin Surg. 2018; 3: 2008.
Abstract
Background and Purpose: Endovascular coiling is the established standard treatment for managing
both unruptured and ruptured cerebral aneurysms worldwide, but long-term durability remains
inferior to surgical clipping. We investigated the risk factors for recurrence after endovascular
treatment of cerebral aneurysms.
Materials and Methods: Between April 2012 and Aug 2016, we treated 92 patients with 95 saccular
cerebral aneurysms. Aneurysms were categorized as side-wall or bifurcation type based on their
morphology.
Results: We included 72 patients; 31 aneurysms were classified as side-wall aneurysms, and two had
an axis parallel to the parent artery. Forty-one bifurcation aneurysms had an axis either perpendicular
(n=23) or parallel (n=18) to the parent artery. Twelve recurrences (16.7%) were observed during
follow-up, and nine underwent retreatment after 3–25 months. All retreatments were performed
endovascularly without subsequent neurological complications. In univariate analysis, aneurysm
dome size, neck width, cerebral aneurysms with an axis parallel to the parent artery, and incomplete
occlusion showed statistically significant associations with recurrence. In multivariate Cox regression
analysis, dome size (hazard ratio: 1.38; 95% CI: 1.06–1.97; p=0.013); branch incorporation (hazard
ratio: 5.76; 95% CI: 1.05–35.66; p=0.042); and axis parallel to the parent artery (hazard ratio: 33.04;
95% CI: 4.47–430.36; p=0.0004) remained risk factors for recurrence.
Conclusion: Dome size, cerebral aneurysms with an axis parallel to the parent artery, and branch
incorporation were significant factors for recurrence after endovascular coiling. More meticulous
embolization and follow-up or alternative methods of changing blood flow into the aneurysm are
necessary in this subgroup of patients.
Abbreviations
CI = Confidence Interval
Introduction
Since the publication of the International Subarachnoid Aneurysm Trial [1], endovascular coiling has been the accepted standard treatment to manage both ruptured and unruptured cerebral aneurysms. Advancements in endovascular technology and techniques have increased the safety of endovascular treatment for the majority of cerebral aneurysms [2,3]. However, the major drawback of endovascular treatment compared with surgical clipping is that long-term durability is inferior especially for large aneurysms [4-6]. A meta-analysis by Ferns et al. [6] showed that aneurysms recurred in 20.8% of patients, and retreatment was performed in 10.3%. In contrast, the reported recurrence rate after surgical clipping was less than 5% [7]. Both coil compaction and aneurysm sac growth are considered the principal mechanisms of recanalization after coil embolization [8]. We prospectively collected data for all patients treated by endovascular treatment between 2012 and 2016, at Kyoto Medical Center. We investigated the incidence of recurrence in clinical practice and identified the risk factors significantly associated with angiographic recurrence after endovascular treatment of cerebral aneurysms.
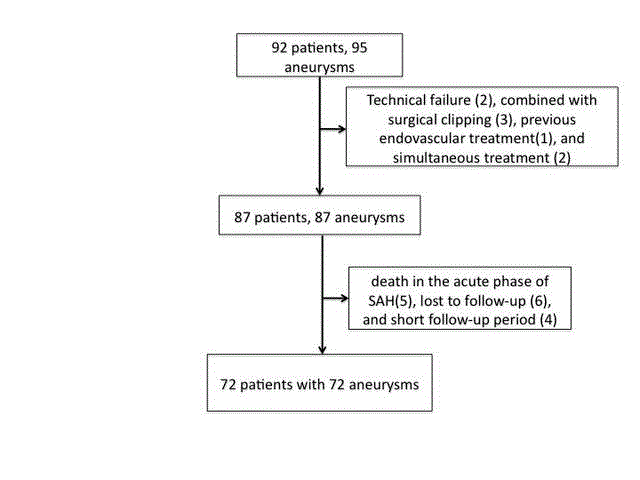
Figure 1
Figure 1
Flowchart shows the selection protocol for patients with cerebral
aneurysms treated by endovascular coiling.
Methods
Patients
Ninety-two patients with 95 saccular cerebral aneurysms were
endovascularly treated at the National Hospital Organization Kyoto
Medical Center between April 2012 and Nov 2016. We retrospectively
included patients who were followed-up for at least six months with
available follow-up head magnetic resonance angiography or cerebral
angiography results. We excluded patients with dissecting or blood
blister-like aneurysms, those who were treated with both coiling
and surgical clipping and patients who underwent endovascular
retreatment. After exclusions, 72 patients met the study criteria
and constituted our study population. Eight patients had multiple
aneurysms, and three of these patients had two aneurysms treated
endovascularly. In two multiple aneurysm patients, because two
aneurysms were in the same location and treated simultaneously,
only one aneurysm was included in the analysis. In one patient with
ruptured anterior communicating aneurysm and an unruptured
basilar artery aneurysm, only the basilar artery aneurysm was included
in the analysis because the anterior communicating aneurysm was
bilobed and treated with coiling combined with surgical clipping.
We excluded patients from the study because of treatment failure
(n=2), death in the acute phase of subarachnoid hemorrhage (n=5),
treatment with coiling combined with surgical clipping (n=2), patient
lost to follow-up (n=6), and short follow-up period (n=5) (Figure 1).
Aneurysms were categorized as side-wall or bifurcation type, based
on their location. Thirty-one aneurysms were classified as side-wall
aneurysms, and two of these had an axis parallel to the parent artery,
as described previously [9]. Forty-one bifurcation aneurysms had
an axis either perpendicular (n=23) or parallel (n=18) to the parent
artery.
Endovascular treatment
The median size of treated aneurysms was 5 mm (range, 2–23
mm). Dual antiplatelet premedication (100 mg acetylsalicylic acid
plus 75 mg clopidogrel or 200 mg cilostazol) was administered to
all patients with unruptured aneurysms. Antiplatelet premedication
(40 mg ozagrel sodium and 200 mg cilostazol with or without 200
mg acetylsalicylic acid and 300 mg clopidogrel) was administered to
three patients with ruptured aneurysms who were treated by stentassisted
coiling. Antiplatelet premedication (100–200 mg cilostazol
with or without 200 mg acetylsalicylic acid or 40 mg ozagrel sodium)
was given to eight other patients with ruptured aneurysms. Almost
all of the patients with ruptured aneurysms were treated under
general anesthesia, whereas almost all of the patients with unruptured
aneurysms were treated using local anesthesia. Anticoagulation
was initiated by an intravenous injection of a bolus of 2000–5000
IU heparin just after introducer sheath placement to increase the
activated clotting time by 2- to 2.5 times above baseline. In ruptured
cases, the activated clotting time was gradually increased during the
procedure. In 44/72 procedures, endovascular coiling was performed
using the single-catheter technique with various types of coils. In
the other procedures, balloon-remodeling (n=15), stent-assisted
(n=8), and double-catheter (n=8) techniques were used based on
the aneurysm geometry. In three procedures, the double-catheter
technique was combined with stent-assisted or balloon-remodeling
techniques. The immediate angiographic outcomes were classified
into three categories, as previously published by Roy et al. [10]. All
patients with major recurrences were retreated endovascularly.
Clinical and angiographic follow-up
Patients were clinically assessed by neurosurgeons before and
after treatment, just prior to discharge, and during clinical followup.
Functional outcomes were evaluated using the modified Rankin
scale score. Follow-up magnetic resonance angiography was used
to evaluate recurrence for most cases at 6, 12, and 24 months after
the procedures. When recurrence was suspected based on followup
magnetic resonance angiography, catheter angiography was also
performed. Recurrence was defined as any increase in the size of the
remnant, and follow-up angiographic results were classified into
three categories: stable or improved occlusion, minor recurrence
requiring no retreatment, or major recurrence requiring retreatment.
The median follow-up period was 15 months (range, 6–44 months).
Statistical analysis
All statistics were evaluated (or performed) by a statistician
(N. Y.).We described frequency and percentage for binary data.
Distribution of continuous variables was described by means and
standard deviation. Univariate Cox regression analysis was performed
to assess the association between a possible risk factor and recurrence,
and we performed multivariate Cox regression with four variables
that are well-known risk factors for recurrence [4-6,10-12] based on
Raymond-Roy classification (class 3 vs. 1, 2), dome size, neck width,
location (internal carotid-posterior communicating vs. other), and
the two variables, branch incorporation and cerebral aneurysm axis
to the parent artery. Variable selection in the model was performed
by backward selection [13]. All P-values are two-sided, and the level
of statistical significance was defined as p<0.05. All statistical analyses
were performed using JMP software for Mac, version 11 (SAS
software, Cary, NC).
Results
Study population
Our study included 24 men and 48 women, with ages ranging from
20–86 years (median, 66 years). Thirty-one patients had unruptured
aneurysms, and 41 patients presented with subarachnoid hemorrhage,
including one who experienced subarachnoid hemorrhage several
years earlier and was initially treated conservatively.
Procedure outcomes
Patients’ demographic and clinical characteristics and the
treatment results are summarized in Table 1. Aneurysms were
located on the internal carotid artery in 34 (47%) patients, middle
cerebral artery in 8 (11%), anterior cerebral artery in 19 (26%), and
vertebrobasilar system in 11 (15%). Postembolization angiography
revealed complete occlusion in 31 (43%) aneurysms, neck remnant
in 32 (44%), and incomplete occlusion in 8 (13%). No patients with
unruptured aneurysms had a change in modified Rankin scale score
after treatment. Thirty-four of 41 (83%) patients with ruptured
aneurysms had favorable outcomes. Overall, 64 patients (89%) had
favorable outcomes (modified Rankin scale, 0–2) at 90 days after
treatment. However, one patient died after 6 months because of
multiple organ failure secondary to sepsis unrelated to the procedure
or the cerebral aneurysm.
Recurrences
Recurrences occurred in 12 (15.7%) of the treated aneurysms,
as shown in Table 2. Recurrent aneurysms were located on the
internal carotid-posterior communicating artery in seven cases,
middle cerebral artery in one, anterior communicating artery in
two, and basilar artery in two. Nine recurrent patients were retreated
endovascularly with no neurological sequelae. The timing of
retreatment ranged from 3–25 months after the first procedure. In 5/9
repeat procedures, embolization was performed using a stent-assisted
technique as shown in Figure 2. One of these patients experienced
a second and third recurrence after retreatment with coils because
of enlargement of the aneurysmal wall, and was eventually treated
using a stent-assisted technique at 19 months. In one patient with a
major recurrence, retreatment was suspended because of the patient’s
advanced age and concomitant chronic kidney disease. No patient
experienced a bleeding episode during follow-up.
Factors associated with recurrences
The associations between potential clinical risk factors and
recurrence after embolization are summarized in Table 2. In the
Cox regression analysis, the variables aneurysm dome size (hazard
ratio: 1.47; 95% confidence interval: 1.21–1.87; p<0.0001), neck width
(hazard ratio: 2.09; 95% confidence interval: 1.30–3.29; p=0.035),
cerebral aneurysm with an axis parallel to the parent artery (hazard
ratio: 3.73; 95% confidence interval: 1.16–12.04; p=0.029), and
incomplete occlusion (hazard ratio: 5.56; 95% confidence interval:
1.47–17.96; p=0.035) showed statistical significance (Table 3).
Multivariate analysis revealed that cerebral aneurysm with an axis
parallel to the parent artery (hazard ratio: 33.04; 95% confidence
interval: 4.47–430.36; p=0.0004), dome size (hazard ratio: 1.38; 95%
confidence interval: 1.06–1.97; p=0.013), and branch incorporation
(hazard ratio: 5.76; 95% confidence interval: 1.05–35.66; p=0.042)
remained statistically significant risk factors (Table 4).
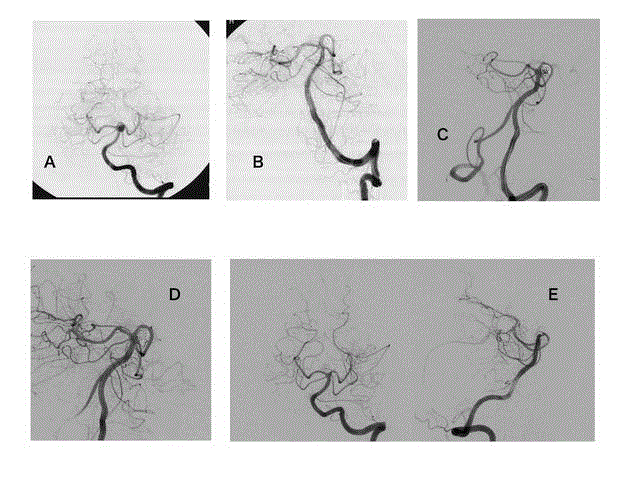
Figure 2
Figure 2
Images from a 63-year-old man with an unruptured aneurysm at the left basilar artery-superior cerebellar artery. A, anteroposterior view of a left
vertebral angiogram shows a saccular aneurysm at the left basilar artery-superior cerebellar artery. B, Postprocedural angiogram shows adequate occlusion of the
aneurysm while preserving the superior cerebellar artery using a balloon-remodeling technique. C, Follow-up angiogram at 12 months after treatment shows major
recanalization of the aneurysm. D, Retreatment was performed using stent-assisted coil embolization. An Enterprise VRD 4.5 mm × 22 mm stent was deployed
through the basilar artery to the left posterior cerebral artery, and the curved basilar artery was linearized. E, Biplanar image of the last follow-up angiograms at 25
months shows stable occlusion of the aneurysm and linearization of the basilar artery.
Table 1
Table 2
Table 3
Table 3
Association between potential risk factors and recanalization after coil embolization of cerebral aneurysms were assessed using univariate Cox regression analysis.
Discussion
Endovascular coiling is the established standard treatment
in managing both unruptured and ruptured cerebral aneurysms
worldwide. However, most authors agree that the durability of
endovascular treatment is inferior to that of surgical clipping [7,14].
The reported incidence of recanalization in a large prospective study
was 5.5% to 33% [7,14-17]. Large aneurysmal size, posterior circulation
aneurysms, wide neck, incomplete occlusion, low packing density,
and ruptured aneurysms are reported risk factors for recurrence
[4,6,12,18-20]. Although some clinical studies have included
advanced technologies to gain more stability or packing density,
including stent-assisted embolization [21,22], hydrogel-coated coils
[23], and large-diameter coils [24], less durability remains the major
drawback of endovascular coiling. In our series, the recanalization
rate after coil embolization of ruptured and unruptured aneurysms
was 16%, which was compatible with previous reports. In our
univariate analysis, larger size, wider neck, and incomplete occlusion
were associated with recanalization, as in previous studies.
Branch incorporation
Because of remarkable advances in endovascular techniques
and technology, aneurysms with a branch incorporated into the
aneurysmal wall can now be embolized without occluding the
incorporated branch [25], which was considered difficult in the
past. However, although it is now easier to preserve an incorporated
branch, it remains difficult to balance the patency of the incorporated
branch and treatment durability to prevent bleeding. Also, few data
are available regarding long-term durability of endovascular coiling
in this subgroup of cerebral aneurysms. To our knowledge, branch
incorporation has not been investigated previously as a factor in
recanalization after endovascular coiling.
Axis to the parent artery
It is well known that cerebral aneurysms at the tip of the basilar
artery are likely to recur after embolization [26,27]. Basilar tip
aneurysms are classified as bifurcation aneurysms and usually have
an axis parallel to the parent artery. In our series, an axis parallel to
the parent artery was the most significant predictor of recanalization,
possibly because of a jet-pattern inflow for these aneurysms. Szikora
et al. [9] reported that aneurysms with a main axis parallel to the
parent artery have a tendency to have a jet-flow pattern and uneven
distribution of unsteady pressure. We are currently conducting a
clinical study to clarify the association between disturbed flow in the
aneurysm and recurrence after embolization using computational
flow-dynamic techniques [28]. We hypothesize that high-magnitude
wall-shear stress and strong disturbed flow may be involved in
aneurysm recurrence. However, our findings and hypothesis require
confirmation in larger future studies.
Size
In our series, univariate analysis showed that aneurysm size was
highly associated with recurrence, similar to previous studies [6,26].
Recurrence was observed in 3/6 (50%) aneurysms larger than 10 mm,
which was compatible with previous studies reporting an incidence
of recanalization after embolization of large or giant aneurysms as
high as 39% [29]. Therefore, although a flow-diverting stent can be a
promising option [30,31], further study is needed to establish the best
method to treat large and giant aneurysms.
Table 4
Limitations
There are certain limitations in our study. First, because of the retrospective nature, selection bias may be present. However, this bias may have been minimized by prospectively recording the data for aneurysms treated endovascularly into a database. A second limitation is the limited number of cases; our findings should be confirmed in a larger prospective study. A third limitation is that the follow-up period was relatively short; the median follow-up period was 15 months. However, all of the patients in this series had follow-up magnetic resonance imaging at 6–12 months, and all recurrences were observed within 12 months. Reopening of cerebral aneurysms is rare when adequate occlusion is confirmed at 6 months after embolization [32]. We found that the risks of recurrence associated with axis to the parent artery and branch incorporation were significant, but a slight data bias may be present [33] because these aneurysms had large hazard ratios and/or very wide confidence intervals on multivariable Cox regression.
Conclusion
In our cohort, the recanalization rate after coil embolization of ruptured and unruptured aneurysms was 16%. In addition to dome size, cerebral aneurysms with an axis parallel to the parent artery and branch incorporation were significant predictors of recurrence after endovascular coiling. More meticulous embolization and followup or alternative methods of changing flow into the aneurysm are necessary in this subgroup of patients.
Acknowledgment
We thank Jane Charbonneau, DVM, from Edanz Group (www. edanzediting.com/ac) for editing a draft of this manuscript.
References
- Molyneux A, Kerr R, Stratton I, Sandercock P, Clarke M, Shrimpton J, et al. International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet. 2002;360:1267-74.
- Brinjikji W, Rabinstein AA, Lanzino G, Kallmes DF, Cloft HJ. Patient outcomes are better for unruptured cerebral aneurysms treated at centers that preferentially treat with endovascular coiling: a study of the national inpatient sample 2001-2007. AJNR Am J Neuroradiol. 2011;32:1065-70.
- Brinjikji W, Rabinstein AA, Nasr DM, Lanzino G, Kallmes DF, Cloft HJ. Better outcomes with treatment by coiling relative to clipping of unruptured intracranial aneurysms in the United States, 2001-2008. AJNR Am J Neuroradiol. 2011;32:1071-75.
- Campi A, Ramzi N, Molyneux AJ, Summers PE, Kerr RS, Sneade M, et al. Retreatment of ruptured cerebral aneurysms in patients randomized by coiling or clipping in the International Subarachnoid Aneurysm Trial (ISAT). Stroke. 2007;38:1538-44.
- van Rooij WJ, Sprengers ME, Sluzewski M, Beute GN. Intracranial aneurysms that repeatedly reopen over time after coiling: imaging characteristics and treatment outcome. Neuroradiology. 2007;49:343-9.
- Ferns SP, Sprengers ME, van Rooij WJ, Rinkel GJ, van Rijn JC, Bipat S, et al. Coiling of intracranial aneurysms: a systematic review on initial occlusion and reopening and retreatment rates. Stroke. 2009;40:523-9.
- Spetzler RF, McDougall CG, Zabramski JM, Albuquerque FC, Hills NK. The Barrow Ruptured Aneurysm Trial: 6-year results. J Neurosurg. 2015;123(3):609-17.
- Hasan DM, Nadareyshvili AI, Hoppe AL, Mahaney KB, Kung DK, Raghavan ML. Cerebral aneurysm sac growth as the etiology of recurrence after successful coil embolization. Stroke. 2012;43:866-8.
- Szikora I, Paal G, Ugron A, Nasztanovics F, Marosfoi M, Berentei Z, et al. Impact of aneurysmal geometry on intraaneurysmal flow: a computerized flow simulation study. Neuroradiology. 2008;50(5):411-21.
- Roy D, Milot G, Raymond J. Endovascular treatment of unruptured aneurysms. Stroke. 2001;32(9):1998-2004.
- Wehman JC, Hanel RA, Levy EI, Hopkins LN. Giant cerebral aneurysms: endovascular challenges. Neurosurgery. 2006;59:S125-38.
- Pierot L, Cognard C, Anxionnat R, Ricolfi F, Clarity Investigators. Endovascular treatment of ruptured intracranial aneurysms: factors affecting midterm quality anatomic results: analysis in a prospective, multicenter series of patients (CLARITY). AJNR Am J Neuroradiol. 2012;33:1475-80.
- Robins JM, Greenland S. The role of model selection in causal inference from nonexperimental data. Am J Epidemiol. 1986;123(3):392-402.
- Molyneux AJ, Birks J, Clarke A, Sneade M, Kerr RS. The durability of endovascular coiling versus neurosurgical clipping of ruptured cerebral aneurysms: 18 year follow-up of the UK cohort of the International Subarachnoid Aneurysm Trial (ISAT). Lancet. 2015;385:691-7.
- White PM, Lewis SC, Gholkar A, Sellar RJ, Nahser H, Cognard C, et al. Hydrogel-coated coils versus bare platinum coils for the endovascular treatment of intracranial aneurysms (HELPS): a randomised controlled trial. Lancet. 2011;377:1655-62.
- Coley S, Sneade M, Clarke A, Mehta Z, Kallmes D, Cekirge S, et al. Cerecyte coil trial: procedural safety and clinical outcomes in patients with ruptured and unruptured intracranial aneurysms. AJNR Am J Neuroradiol. 2012;33:474-80.
- McDougall CG, Johnston SC, Gholkar A, Barnwell SL, Vazquez Suarez JC, Masso Romero J, et al. Bioactive versus bare platinum coils in the treatment of intracranial aneurysms: the MAPS (Matrix and Platinum Science) trial. AJNR Am J Neuroradiol. 2014;35:935-42.
- Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, Lambert A, et al. Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke. 2003;34:1398-403.
- Ogilvy CS, Chua MH, Fusco MR, Griessenauer CJ, Harrigan MR, Sonig A, et al. Validation of a System to Predict Recanalization After Endovascular Treatment of Intracranial Aneurysms. Neurosurgery, 2015;77:168-73.
- Sluzewski M, van Rooij WJ, Slob MJ, Bescos JO, Slump CH, Wijnalda D. Relation between aneurysm volume, packing, and compaction in 145 cerebral aneurysms treated with coils. Radiology. 2004;231:653-8.
- Geyik S, Yavuz K, Yurttutan N, Saatci I, Cekirge HS. Stent-assisted coiling in endovascular treatment of 500 consecutive cerebral aneurysms with long-term follow-up. AJNR Am J Neuroradiol. 2013;34(11):2157-62.
- Nishido H, Piotin M, Bartolini B, Pistocchi S, Redjem H, Blanc R. Analysis of complications and recurrences of aneurysm coiling with special emphasis on the stent-assisted technique. AJNR Am J Neuroradiol. 2014;35:339-44.
- Brinjikji W, White PM, Nahser H, Wardlaw J, Sellar R, Cloft HJ, et al. HydroCoils reduce recurrence rates in recently ruptured medium-sized intracranial aneurysms: a subgroup analysis of the HELPS trial. AJNR Am J Neuroradiol. 2015;36:1136-41.
- Milburn J, Pansara AL, Vidal G, Martinez RC. Initial experience using the Penumbra coil 400: comparison of aneurysm packing, cost effectiveness, and coil efficiency. J Neurointerv Surg. 2014;6(2):121-4.
- Kim BM, Park SI, Kim DJ, Kim DI, Suh SH, Kwon TH, et al. Endovascular coil embolization of aneurysms with a branch incorporated into the sac. AJNR Am J Neuroradiol. 2010;31(1):145-51.
- Ferns SP, Sprengers ME, van Rooij WJ, van Zwam WH, de Kort GA, Velthuis BK, et al. Late reopening of adequately coiled intracranial aneurysms: frequency and risk factors in 400 patients with 440 aneurysms. Stroke. 2011;42:1331-7.
- van Eijck M, Bechan RS, Sluzewski M, Peluso JP, Roks G, van Rooij WJ. Clinical and Imaging Follow-Up of Patients with Coiled Basilar Tip Aneurysms Up to 20 Years. AJNR Am J Neuroradiol. 2015;36(11):2108-13.
- Shimogonya Y, Fukuda S. Computational and experimental studies into the hemodynamics of cerebral aneurysms. J Biomechanical Science and Engineering. 2016;11;15-488.
- Chalouhi N, Tjoumakaris S, Gonzalez LF, Dumont AS, Starke RM, Hasan D, et al. Coiling of large and giant aneurysms: complications and long-term results of 334 cases. AJNR Am J Neuroradiol. 2014;35(3):546-52.
- Becske T, Kallmes DF, Saatci I, McDougall CG, Szikora I, Lanzino G, et al. Pipeline for uncoilable or failed aneurysms: results from a multicenter clinical trial. Radiology. 2013;267(3):858-68.
- Szikora I, Berentei Z, Kulcsar Z, Marosfoi M, Vajda ZS, Lee W, et al. Treatment of intracranial aneurysms by functional reconstruction of the parent artery: the Budapest experience with the pipeline embolization device. AJNR Am J Neuroradiol. 2010;31:1139-47.
- Sprengers ME, Schaafsma J, van Rooij WJ, Sluzewski M, Rinkel GJ, Velthuis BK, et al. Stability of intracranial aneurysms adequately occluded 6 months after coiling: a 3T MR angiography multicenter long-term follow-up study. AJNR Am J Neuroradiol. 2008;29:1768-74.
- Greenland S, Mansournia MA, Altman DG. Sparse data bias: a problem hiding in plain sight. BMJ. 2016;352:i1981.