Research Article
Unrequited Returns in Asymptomatic Colorectal Cancer Detection
Williamson B Strum*
Scripps Clinic Medical Group, CA, USA
*Corresponding author: Williamson B Strum, Scripps Clinic Medical Group, CA, USA
Published: 28 Jun, 2018
Cite this article as: Strum WB. Unrequited Returns in
Asymptomatic Colorectal Cancer
Detection. Clin Surg. 2018; 3: 1995.
Abstract
Aim: Colonoscopy has become the favored screening test for colorectal cancer. Measures have been
identified since 2000 to enhance the quality of colonoscopy and to increase screening for at-risk
persons. This study was designed to determine if changes in the rates of detection of colorectal
cancer in asymptomatic persons are occurring. Methods: this is an observational study of a database
of 887 patients taken from the colorectal cancer registry between 2006 and 2015. Participants were
between 50 and 89 years of age and had initial detection of colorectal neoplasia by screen-detection
or symptom-detection, and the clinical characteristics were compared. Rates of detection of
colorectal cancer in asymptomatic and symptomatic persons were evaluated over five consecutive,
two-year segments.
Results: 417 persons met the inclusion criteria. Screen-detected colorectal cancer occurred in 66
(16%) and symptom-detected in 351 persons and indicated the following: average age was 62.7 years
vs. 71.3 years (p=0.001), men represented 66.2% vs. 49.6% (p=0.014), and early stage of disease was
found in 69.7% vs. 43.9% (p=0.001). Race, family history, and location of disease were not different.
Evaluation of the five two-year segments for changes in the rates of detection of screen-detected
colorectal cancer did not demonstrate a significant change (p=0.803).
Conclusion: Persons with colorectal cancer who were detected by screening were significantly
younger, more often men, and had lower stages of disease. Rates of screen-detected colorectal cancer
remained stagnant over the decade of study, implicating a rejection by at-risk persons to undergo
screening or a lack of referral for screening.
Introduction
Colorectal cancer mortality in the United States has been declining since 1975, yet in 2017 135,000 persons will be discovered to have colorectal cancer and 50,000 will die [1]. The decline in mortality from colorectal cancer since 1975 is attributed to 3 factors: 1) screening, accounting for 53% of the decline, 2) improved life style, accounting for 35%, and improved treatment, accounting for 12% [1,2]. Most of the benefit from screening is attributed to increased application of optical colonoscopy [1-3]. Colonoscopy has the advantage of being preventive by removing precancerous adenomas as well as by detecting early-stage cancer [4-7]. However, less than 30% of colorectal cancer cases are reported as being diagnosed before having symptoms [8-10], indicating that screen-detection approaches have not been reaching most persons with early-stage colorectal cancer. Increased public health emphasis on screening, including fecal occult blood testing, and improvements in the quality of colonoscopy [4-7,11], began over a decade ago as screening goals for all parameters were increasing from 25% in 2005 to 70% in 2015 [1,11]. The success of these efforts in increasing screen-detected colorectal cancer has not been demonstrated. This study was designed to evaluate the impact these measures had on the number of screen-detected cases of colorectal cancer between 2006 and 2015 as they relate to a large medical group practice in southwestern United States.
Methods
This is an Internal Review Board-approved, observational study of patients with colorectal cancer taken from the colorectal cancer registry of the Scripps Green Hospital in La Jolla, CA. Participants diagnosed between January 1, 2006 and December 31, 2015 with ages between 50 and 89 years were evaluated for initial detection of colorectal cancer based on screening measures versus diagnosis by symptom detection. Colorectal cancer was defined as adenocarcinoma between the cecum and the dentate line. The age of inclusion began with the age at which average-risk persons are first advised to have a screening examination for colorectal cancer and ended at 3 years after the last age at which screening is recommended in select persons [1,3,4]. All persons were diagnosed and treated at the Scripps Green Hospital and satellite facilities. The electronic medical record for each person was reviewed by the author to verify the primary results. Persons were excluded who were diagnosed at an outside institution and transferred to Scripps Green Hospital, as were persons with a history of colorectal cancer, colon adenomas, prior colonoscopy, inflammatory bowel disease, non-adenomatous colon cancer, Lynch and other familial colorectal cancer syndromes, or an incomplete record. Stage of disease was divided into early or late at the inflection point between Stage ll-A and Stage ll-B, which separates those not recommended for adjuvant therapy from those who are [12]. Symptomatic persons were evaluated for the dominant sign or symptom, which was included in 1 of 5 categories; rectal bleeding, iron deficiency anemia, abdominal pain, persistent change in bowel habit, or miscellaneous, the latter encompassing weight loss, ascites, prolonged fatigue or fever. The subjects underwent bowel preparation with a minimum of 24-hr, clear liquid diet and standard laxation with sodium phosphate or polyethylene glycol (Suprep, Go-LYTELY, NULYTLY, Braintree Laboratories; Moviprep, Salix Pharmaceuticals). All colonoscopy examinations were performed by one of sixteen experienced, board-certified gastroenterologists or gastroenterology fellows-in-training under direct supervision in either an accredited hospital or an outpatient endoscopy room in 30 min to 45 min time slots. Each staff examiner had a minimum experience of 500 to over 5000 colonoscopies at the outset of the study and met benchmark rates for adenoma detection, cecal intubation, duration of withdrawal time and low rate of complications. Each examiner performed an average of 600-1000 endoscopic examinations per year. All examiners were fully aware of the need for examination withdrawal time of at least 6 or more minutes, the importance of reaching the cecum in all cases, meeting or exceeding minimum adenoma detection rates, the utility of retroflexed evaluation of the proximal colon and rectum, and the necessity of a well-prepared colon for a quality examination. Any cases not meeting these requirements were to have a repeat evaluation in the immediate future. Compliance was determined during periodic assessments by the Quality Assurance Team of Scripps Green Hospital and the satellite facilities. The examination rooms were equipped with high-definition Olympus colonoscopes with narrow band imaging beginning in 2005 and reaching comparable equipment with Olympus 180 colonoscopes in all rooms by 2013. All pathology readings were by experienced board-certified pathologists who held daily conferences for group review of complicated cases. Fecal occult blood tests used the guaiac method from 2006-2009 and fecal immunochemistry test (Polymedco) from 2010 through 2015. The participants were divided by two primary end-points: 1) persons who were asymptomatic and deemed screen-detected by colonoscopy, with or without a current positive screening fecal occult blood test; and 2) persons diagnosed based on symptoms and deemed symptom-detected. These two categories were compared for age, sex, race, positive family history of colorectal cancer in at least one first degree relative, location of disease, and early versus late stage of disease. The location of disease was divided between proximal colon (cecum to splenic flexure) and distal colon (descending colon to rectum). Demographics and clinical characteristics of the study population were summarized using a mean and standard deviation for the continuous variable (age) and frequencies for the categorical variables (sex, race, family history, cancer location, and stage of disease). An independent sample t-test and chi-square tests were used to assess differences in demographics and characteristics between screen-detected and symptom-detected persons. Chi-square tests were used to compare the overall ratio of screen-detected vs. symptom-detected persons across all years, as well as between each of the five consecutive, two-year segments. SPSS version 13.0 was used for all analyses (SSPS Inc, Chicago, Illinois). Statistical significance was set at p<0.05.
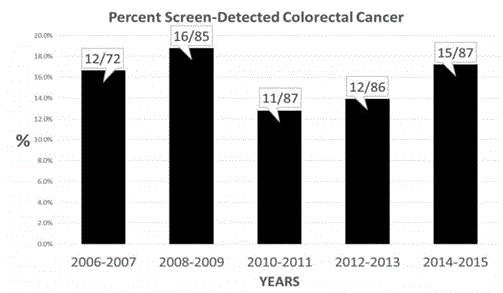
Figure 1
Figure 1
Time trend of percent of persons with screen-detected colorectal
cancer compared to total study persons with colorectal cancer by two-year
intervals.
Results
The Scripps Green Hospital colorectal cancer registry contained
887 entrants between January 1, 2006 and December 31, 2015, of
which 417 met the inclusion criteria. Of the 470 persons excluded
from the analysis, 154 had had prior colonoscopy evaluation, 115
were non analytic cases originally evaluated at other institutions, 102
were too young, 34 were too old, 23 had neuroendocrine cancers, 13
had synchronous colorectal cancer, 11 had an incomplete record,
7 had squamous cell cancers, 7 had Lynch syndrome, and 4 had
inflammatory bowel disease. Persons with screen-detected colorectal
cancer numbered 66 out of 417 persons (16%). Within the two years
preceding diagnosis of colorectal cancer, 14 of the 66 screen-detected
persons had a positive screening fecal occult blood test (21%),
which initiated the pre-symptomatic colonoscopy examination, 10
had a negative fecal occult blood test (15%), and 42 were not tested
(64%). The 66 diagnoses by screening colonoscopy were randomly
shared among the 16 examiners with a range of 1-9 diagnoses per
examiner, which generally corresponded to the number of screening
examinations performed. The dominant sign or symptom leading to
symptom-detected colorectal cancer in 351 persons was rectal bleeding
in 125 (36%), iron-deficiency anemia in 115 (33%), abdominal
pain in 75 (21%), persistent change in bowel habit in 21 (6%), and
miscellaneous in 15 (3%). An abdominal computerized Tomographic
scan was abnormal in 74 of the 351 symptomatic persons prior to
the diagnosis of colorectal cancer. A comparison of the age, sex,
race, family history, location of disease, and stage of disease of the 66
screen-detected persons to the 351 symptom-detected persons with
CRC led, respectively, to the following: the average age was 62.7 +
9.6 years vs. 71.3 + 11.0 years (p<0.001), men represented 66.2% vs.
49.6% (p<0.014), race was predominantly Caucasian at 92.3% and
91.4% (p<0.805), positive family history for at least one first degree
relative was present in 13.3% vs. 18.9% (p<0.303), the cancers were
distributed equally between the proximal and distal colon, and early
stage of disease was found in 69.7% vs. 43.9% (p<0.001) (Table 1).
After dividing the cases into five consecutive 2-year partitions,
the data indicated no significant change within the partitions for
screen-detected cases, which were evenly distributed over the 5
biennial segments (p=0.803) and the ratio did not exceed 18%. There
was no significant difference between the results within the 5-year
segments (all p values >0.05), and the number of persons having
screen-detected colorectal cancer remained relatively consistent over
the decade of study, never exceeding 20% of the total (Figure 1).
Table 1
Table 1
Demographics and characteristics of screen-detected and symptomdetected
persons with colorectal cancer.
Discussion
Colonoscopy is recognized as the only screening technique
which offers early detection of asymptomatic colorectal cancer as
well as prevention of colorectal cancer by removal of precancerous
adenomas [4-7]. The application of colonoscopy for colorectal cancer
screening proliferated dramatically over the past 17 years, increasing
from 34% in 2000 to 63% in 2015 [1]. These results were propelled by
two main factors, the clinical demonstration of benefit and the cost
coverage of screening colonoscopy by private insurance and Medicare
[1,2]. Randomized studies of fecal occult blood testing and flexible
sigmoidoscopy demonstrate significant, albeit modest, reductions
in the incidence and mortality of colorectal cancer [13-15], while
colonoscopy takes its mantle from observational, case-control and
prospective cohort studies which estimate reductions in colorectal
cancer incidence and mortality to lie between 20% and 50% [1,15,16].
The numeric extent of the benefits for colonoscopy in randomized,
controlled trials remains to be quantified, as several studies are
underway evaluating variations of annual fecal immunochemistry
test or colonoscopy vs. no screening and fecal immunochemistry test
vs. one-time colonoscopy on incidence and mortality [18-20].
This analysis had one primary objective: to determine if enhanced
colonoscopy techniques, made apparent over the past 12 years, in
conjunction with intensified advocacy of colonoscopy, fecal occult
blood tests, and any other screening for at-risk persons were leading
to increased numbers of persons with screen-detected colorectal
cancer as opposed to symptom-detected colorectal cancer. The results
indicated no demonstrable rise in the number of screen-detected
colorectal cancer between 2006 and 2015. At no point in the study did
the needle move to indicate screen-detected cases even reaching 20%.
Consequently, the remaining 80% to 85% of persons with colorectal
cancer had established symptoms before diagnosis. These results
overlap and extend data from the Massachusetts General Hospital
which evaluated 1071 persons with colon cancer, (not including
rectal cancer), between 2004 and 2011and reported 217/ 1071 persons
(20.3%) having screen-detected colon cancer, (having excluded 1.9%
of persons with a positive Hemoccult test for blood as a screening
examination) [8]. The combined reports document the stagnant
nature of the 20% to 30% ceiling for screen-detected colorectal cancer
with the current study extending data another 4 years to 2015. One
might have predicted a gradual rise in screen-detected colorectal
cancer over the decade of evaluation due to improved colonoscopy
technique. A reciprocal fall in interval cancers is associated with
endoscopists who demonstrate high or improved adenoma
detection rates over time [21,22] or have a longer withdrawal time
[23]. However, reports from one of these centers that has employed
systematic quality assessments of adenoma detection rates since 2000
also noted only a modest rise in screen-detected cases from 0.8 to
0.9% between 2000-2004 and 2004-2008 [22,24]. No rise occurred in
the present study.
The dismal results found in the current study implicate the ratelimiting
step for a timely diagnosis as the failure of patients to receive
an initial deep screening test at a pre-symptomatic stage that might
have been accomplished by a fecal occult blood test or direct referral
for colonoscopy. The fault may lie at several areas, including a lapse
in appropriate referral, reluctance, procrastination or rejection by
the patient, or extensive co-morbidities [4-7]. Inadequate quality
of endoscopy, unavailability of resources, and undue costs did not
appear applicable to this population. When taking into account
the attendance rates in several large, no-cost screening studies of
persons being invited to have either fecal immunochemistry test or
colonoscopy that show uptakes of about 40% and 25% [18,26,27], the
results suggest that most of the failure lies with the persons-at-risk not
agreeing to participate, simply procrastinating, or not being referred.
Over the decade of this study the average age of the unscreened,
symptom-detected persons was 71.3 +11.0 years, representing 8.6
years more than the average age of the screen-detected persons. A
concern might be the appropriateness of screening for the upper range
of this group that has reached an older age without screening and
is now facing increased risks for other fatal diseases. However, this
study and others confirm intuitive expectations that early detection
of colorectal cancer leads to an earlier stage of disease [8-11], and an
early stage of disease is associated with a concomitant reduction in
morbidity and mortality with longer survival, reduced requirements
for treatment, less suffering, and lower costs [8-11,28,29]. Leading
experts have recommended that unscreened persons in this age
category who are physically fit be carefully considered for screening,
with colonoscopy being the preferred test [4-6,25]. This analysis has
several strengths as well as limitations. The strengths include the
use of primary data representing quantitative information obtained
from the electronic medical records from a single institution that
was collected for scientific purposes. Each case was evaluated
individually by the author, and the clinical management represented
a standard of care that was stable over the decade of evaluation. The
colonoscopy examinations were performed by a consistent team of
experienced endoscopists each of whom averaged between 600 and
1000 endoscopic examinations per year. Although the denominator
for the total number of patients at risk for colorectal cancer for this
institution was not available, the comparative number of total cases
in each of the five 2-year partitions documented remarkable stability
during this decade, and the total number of colorectal cancer cases
per year was almost identical to that in a report from previous years
[30]. Limitations include that the examinations were not individually
assessed for quality nor were all video photos reviewed leaving the
possibility that some examinations were incomplete, although
this would not likely have changed the primary results. A remote
possibility remains that cases in this southwestern United States area
had differences in life-style, occupations, and education that make it
unaccountable to a broader population. Likewise, confounding biases
may have been present which were unrecognized.
The outcomes from this and related analyses suggest that the
causes for reduction in the incidence and mortality of colorectal
cancer are less related to screening than previously thought and
that efforts toward increased screening have an appreciable margin
for improvement [1,31-33]. Although colonoscopy remains an
established asset for its ability to detect and remove precancerous
adenomas, the full value of early detection of colorectal cancer has
been muted, and the expectations for screening colonoscopy have
not been requited. This study indicates the apparent sources of the
defaults, thus identifying targets for improvement.
Acknowledgement
I thank Julie McCauley for statistical assistance, Alina So and Rajiv Pande for assistance with preparation of the data, Karen Wheeler, Kylie Newman, and Clarence Sandbakken for research assistance.
References
- American Cancer Society. Colorectal Cancer Facts & Figures. 2017-2019. Atlanta: American Cancer Society; 2017.
- Murphy CC, Sandler RS, Sanoff HK, Yang YC, Lund JL, Baron JA. Decrease in incidence of colorectal cancer among individuals 50 years or older after recommendations for population-based screening. Clin Gastroenterol Hepatol. 2017;15(6):903-09.
- Welch HG, Robertson DJ. Colorectal cancer on the decline- Why screening can’t explain it all. N Engl J Med. 2016;374:1605-07.
- Inadomi JM. Screening for colorectal neoplasia. N Engl J Med. 2017;376:149-56.
- Bibbins-Domingo K, Grossman DC, Curry SJ, Davidson KW, Epling JW Jr, García FAR, et al. Screening for colorectal cancer; US Preventive Services Task Force Recommendation Statement. JAMA 2016;315(23):2564-75.
- Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, et al. Colorectal cancer screening: Recommendations for physicians and patients from the US Mult-Society Task Force on Colorectal Cancer. Am J Gastroenterol. 2017;112:1016-30.
- Strum WB. Colorectal adenomas. N Engl J Med. 2016;374:1065-75.
- Amri R, Bordelanou LG, Sylla P, Berger DL. Impact of screening colonoscopy on outcomes of colon cancer surgery. JAMA. 2013;148(8):747-54.
- Kubisch CH, Crispin A, Mansmann U, Goke B, Kolligs FT. Screening for colorectal cancer is associated with lower disease stage: a population-based study. Clin Gastroenterol Hepatol. 2016;14(11):1612-18.
- Friedrich K, Gruter L, Gotthardt D, Christoph Eisenbach, Wolfgang Stremmel, Sabine G. Scholl, et al. Survival in patients with colorectal diagnosed by screening colonoscopy. Gastrointestinal Endoscopy. 2015;82(1):133-7.
- Rex DK, Schoenfeld PS, Cohen J, Pike IM, Adler DG, Fennerty MB, et al. Quality indicators for colonoscopy. Am J Gastroenterol. 2015;110(1):72-90.
- Redston M, Driman DK. Epithelial neoplasms of the large intestine. In: Odze RD, Goldblum JR, editors. Surgical pathology of the GI tract, liver, biliary tract, and pancreas. 3rd ed. Philadelphia: Elsevier/Saunders, 2015:621- 42.
- Shaukat A, Mongin SJ, Geisser MS, Frank A. Lederle, John H. Bond, Jack S. Mandel, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013;369:1106-14.
- Robertson DJ, Lee JK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: A consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;152(5):1217-37.
- Schoen RE, Pinsky PF, Weissfeld JL, Yokochi LA, Church T, Laiyemo AO, et al. Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med. 2012;366(25):2345-57.
- Nishihara R, Wu K, Lochhead P, Morikawa T, Liao X, Qian ZR, et al. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med. 2013;369:1095-105.
- Zauber AG, Winawer SJ, O’Brien MJ, Vogelaar IL, Ballegooijen MV, Hankey BF, et al. Colonoscopicpolypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366(8):687-96.
- Quintero E, Castells A, Bujanda L, Cubiella J, Salas D, Lanaset A, et al. Colonoscopy versus fecal immunochemical testing in colorectal-cancer screening. N Engl J Med. 2012;366:697-706.
- Dominitz JA, Robertson DJ, Ahnen DJ, Allison JE, Antonelli M, Boardman KD, et al. Colonoscopy vs Fecal Immunochemical Test in Reducing Mortality from Colorectal Cancer (CONFIRM): Rationale for Study Design. Am J Gastroenterol. 2017;112(11):1736-46.
- SCREESCO TRIAL. Screening of Swedish colons; Sponsor: Uppsala University Hospital. 2014.
- Corley DA, Jensen CD, Marks AR, Zhao WK, Lee JK, Doubeni CA, et al. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med. 2014;370:1298-306.
- Kaminski MF, Wieszczy P, Rupinski M, Wojciechowska U, Didkowska J, Kraszewska E, et al. Increased rates of adenoma detection with reduced risk of colorectal cancer and death. Gastroenterology. 2017;153(1):98-105.
- Shaukat, A, Church TR, Lederle FA, Kim AS, Rank JM, Allen JI, et al. Longer Withdrawal Time Is Associated With a Reduced Incidence of Interval Cancer After Screening Colonoscopy. Gastroenterology. 2015;149(4):952-57.
- Kaminski MF, Regula J, Kraszewska E, Polkowski M, Wojciechowska U, Didkowska J, et al. Quality indicators for colonoscopy and the risk of interval cancer. N Engl J Med. 2010;362:1795-803.
- Lieberman DA. Screening for colorectal cancer. N Engl J Med. 2009;361:1179- 87.
- Segnan N, Sinore C, Andreoni B, Azzoni A, Bisanti L, Cardelli A , et al. Comparing attendance and detection rate of colonoscopy with sigmoidoscopy and FIT for colorectal cancer screening. Gastroenterology. 2007;132(7):2304-12.
- Gupta S, Halm EA, Rockey DC, Hammons M, Koch M, Carter E, et al. Comparative effectiveness of fecal immunochemical test outreach, gastroenterology research, and usual care for boosting colorectal cancer screening for the underserved: a randomized clinical trial. JAMA Intern Med. 2013;173:1725-32.
- vanHees F, Habbema DJF, Meester RG, Lansdorp-Vogelaar I, van Ballegooijen M, Zauber AG. Should colorectal cancer screening be considered in elderly persons without previous screening? A cost-effectiveness analysis. Ann Intern Med. 2014;160(11):750-9.
- Meester RG, Doubeni CA, Lansdorp-Vogelaar, Jensen CD, van der Meulen MP, Levin TR, et al. Variation in Adenoma Detection Rate and the Lifetime Benefits and Cost of Colorectal Cancer Screening: A Microsimulation Model. JAMA. 2015;313(23):2349-58.
- Strum WB. Impact of a family history of colorectal cancer on age at diagnosis, anatomic location, and clinical characteristics of colorectal cancer. Int J Gastrointest Cancer. 2005;35(2):121-6.
- Le V, Syed S, Vega KJ, Sharma T, Madhoun MF, Srinivasan N, et al. Patient prompting of their physician resulted in increased colon cancer screening referrals. World J Gastroenterology Oncol. 2014;6(7):257-262.
- Alberti LR, Garcia DPC, Coelho DL, Alves De Lima Dc, Petroianu A. How to improve colon cancer screening rates. World J Gastroenterology Oncol. 2015;7(12):484-91.
- Levin TR. Colorectal cancer screening: Money isn’t everything… but it helps. Gastroenterology. 2017;153(5):1181-183.