Research Article
Magnetic Resonance Imaging of Immediate Breast Reconstruction with Silicone Implant and Latissimus Dorsi Flap
Aleksandra Pirjavec Mahić1, Damir Grebić2*, Barbara Čandrlić3, Ivan Sebaher1 and Harry Grbas4
1Department of Plastic and Reconstructive Surgery, Clinical Hospital Center Rijeka, Croatia
2Department of General and Oncological Surgery, Clinical Hospital Center Rijeka, Croatia
3Department of Radiology, Clinical Hospital Center Rijeka, Croatia
4Department of Digestive Surgery, Clinical Hospital Center Rijeka, Croatia
*Corresponding author: Damir Grebić, Department of General and Oncological Surgery, Clinical Hospital Center Rijeka, Krešimirova 42, 51000 Rijeka, Croatia
Published: 12 Feb, 2018
Cite this article as: Mahić AP, Grebić D, Čandrlić D,
Sebaher I, Grbas H. Magnetic
Resonance Imaging of Immediate
Breast Reconstruction with Silicone
Implant and Latissimus Dorsi Flap. Clin
Surg. 2018; 3: 1907.
Abstract
Purpose: Skin-sparing mastectomy followed by immediate breast reconstruction is one of the latest
revolutions of surgical treatment for breast cancer.
Purpose was to analyse implants using magnetic resonance imaging after immediate breast
reconstruction with a combined silicone implant and latissimus dorsi muscle flap technique and to
compare the results with clinical findings using the Baker scale.
Methods: From May 2011 to May 2015, 22 patients with breast cancer (T1 and T2N0M0) underwent
skin-sparing mastectomy and immediate breast reconstruction procedures using the latissimus
dorsi muscle flap in combination with anatomical silicone breast implants. Magnetic resonance
imaging was used to detect signs of capsular contracture, intracapsular or extracapsular rupture.
Results: Analysing magnetic resonance, 9 patients (41%) (6 of which underwent radiotherapy)
presented signs of capsular contracture, manifested with an anteroposterior to mediolateral ratio
of less than 2. This correlates with 6 patients who presented clinical findings of Baker grades II and
III contracture.
The mean fibrous capsule thickness in patients with radiotherapy was 1.6 mm and without
radiotherapy 0.9 mm. The mean value of maximum length of free fluid around the implant was 3.8
mm in patients with radiotherapy versus 2.9 mm in patients without radiotherapy. The mean value
of reconstructed muscle thickness was 5 mm in patients with radiotherapy and 5.8 mm in patients
without radiotherapy.
Conclusion: We consider immediate breast reconstruction to have better results if the operation
is performed without additional radiotherapy. Magnetic resonance imaging can be used to analyse
reconstructed breasts, especially for the detection of postoperative complications.
Keywords: Breast cancer; Capsular contracture; Immediate breast reconstruction; Magnetic
resonance imaging; Skin-sparing mastectomy
Introduction
Skin-Sparing Mastectomy (SSM) is one of the latest revolutions of surgical treatment for breast
cancer. SSM followed by Immediate Breast Reconstruction (IBR) using autologous tissue and/
or silicone implants has been advocated as an effective treatment option for patients with early
stage breast cancer [1,2]. The greatest asset of SSM is the tremendous advantage that it provides for
the reconstructive surgeon, as it preserves the intact skin envelope of the breast, thereby reducing
the size of the mastectomy scar [3]. The breast can be reconstructed using prosthesis alone, using
autologous tissue alone, or by combining these two methods (using the latissimus dorsi muscle
flap plus implant). The cosmetic appearance of the reconstructed breast is dependent upon the
quantity of remaining breast skin and the location of the skin incision. Utilising the native skin
envelope, it is possible to optimise the final contour of the reconstructed breast, and to achieve
an excellent cosmetic result [4-7]. SSM should not be used in patients with inflammatory breast
cancer or an extensive tumour that involves the skin [8]. Evaluation of capsular contracture is
more accurate using Magnetic Resonance Imaging (MRI) compared
to mammography or sonography, or clinical signs (Baker scale).
Implant rupture can be a complication in patients with implants.
The most reliable signs for intracapsular rupture are considered to be
the linguine and keyhole signs [9]. Macroscopic extrusion of silicone
through the fibrous capsule into the parenchyma, muscles, or lymph
nodes is a characteristic of extracapsular rupture.
MRI is the most accurate method for evaluating implant
integrity, but it requires understanding the numerous variations in
implant construction that are encountered clinically. Indications for
breast MRI include patients with silicone implants after mastectomy
or augmentation mammoplasty, as well as for the detection of
recurrence, prosthesis rupture, or capsular contracture [9]. The aim
of this study was to compare the results of MRI analysis of breast
implants obtained by combining silicone implants and latissimus
dorsi muscle flaps in patients with and without radiotherapy, and to
compare the MRI results with clinical findings using the Baker scale.
Figure 1
Figure 1
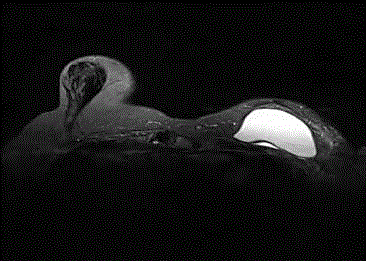
Axial turbo inversion recovery magnitude water suppressed
silicone image shows left breast silicone implant with an anteroposterior to
mediolateral ratio less than 2, approximately 1.7. Fibrous capsule thickness
is approximately 1.6 mm.
Figure 2
Figure 2
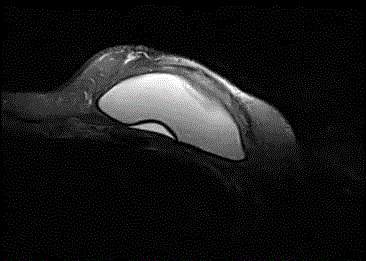
Axial T1 flash 3D non-fat-sat image shows a lot of radial folds.
Fibrous capsule thickness is approximately 1.8 mm.
Materials and Methods
The retrospective analysis was performed at the Department
of Plastic and Reconstructive Surgery, and at the Department of
Radiology, during a period of 4 years from May 2011 to May 2015.
Informed consent was obtained from each patient. The procedure
was performed on 22 patients, mean age 50 (range 28 - 68). All 22
patients with early breast cancer underwent SSM and IBR procedures
using latissimus dorsi muscle flaps in combination with anatomical
silicone breast implants. The SSM was always performed by the
same team of breast surgeons, and the IBR was always carried out
by the same plastic surgeon. The criteria for choosing patients were
primary breast cancer stages T1N0M0 and T2N0M0, according to
the TNM classification. Patients with inflammatory breast cancers,
disseminated malignant disease, exulcerated breast cancers, or with
mesenchymal breast tumours or tumours with two components
(mesenchymal and epithelial, phyllodes tumours), and those with
systemic collagenosis (such as scleroderma), were excluded from
this study. We also excluded women younger than 20 and older
than 70. Contour Profile gel breast implants feature an anatomic
shape that closely resembles the outline of a natural breast. We
considered the age, histological findings, and adjuvant radiotherapy
and/or chemotherapy. One year after treatment completion, all
patients underwent clinical examination using the Baker scale for
capsular contracture, and MRI examination of the implant. MRI was
performed by using a 1.5T system with a dedicated breast surface.
Breast implants were examined using axial sagittal T2- and T1-
weighted turbo spin-echo images, axial turbo inversion recovery
magnitude, and T1 Dixon W and T1 Dixon F images. We evaluated
the following MRI parameters: anteroposterior to mediolateral
implant ratio, fibrous capsule thickness (mm), circumferential fluid
collection presence, latissimus dorsi muscle flap thickness, implant
contour regularity (anteroposterior or mediolateral folds), lumen
homogeneity or dishomogeneity, shell interruption with silicone
leakage, and free silicone presence in the breast or axilla.
We monitored for the presence of signs for capsular contracture,
intracapsular rupture, or extracapsular rupture. Capsular contracture
was considered when the silicone implant was asymmetrical in
shape, manifested as an anteroposterior to mediolateral ratio
less than 2, as decided by the authors. Patients were divided into
two groups (16 patients who did not receive radiotherapy and 6
patients who underwent radiotherapy), and MRI parameters were
compared. The results of MRI findings were analysed by the same
qualified radiologist. Indication for radiotherapy was determined by
an oncologist, based on pathohistological findings. We calculated
the mean patient's age using descriptive statistics. The differences
between values of various parameters in patients who underwent
radiotherapy and those who did not were determined by the nonparametric
Student's t-test. P-values less than 0.05 were considered
statistically significant. Statistical analyses were performed using a
statistical software package for Windows (new version 10 by Statsoft,
Inc.).
Results
There were 10 patients (45%) with stage T1N0M0 breast cancer
and 12 patients (55%) with stage T2N0M0 breast cancer according
to TNM classification. Intraoperative diagnosis of breast carcinoma
was performed in 4 patients (18%) by pathohistological examination
of the excised tumour, while the preoperative diagnosis of carcinoma
was confirmed by frozen section examination in 18 patients (82%).
Histological analysis showed that among 22 patients included in this
study, 10 of them (45%) presented ductal carcinoma in situ, while 11
patients (50%) had invasive ductal carcinoma. There were no patients
with lobular carcinoma in situ, but 1 patient (4,5%) presented an
invasive type of lobular carcinoma. All patients with invasive cancer
components underwent a sentinel lymph node biopsy, and all patients
had an intraoperatively negative imprint cytology examination
result. Clinically, 4 (18%) of the evaluated patients presented the
complication of Baker grade II capsular contracture, and 2 (9%)
patients Baker grade III capsular contracture. There were no patients
with Baker grades I or IV capsular contracture. None of the evaluated
patients presented contracture to such a degree that would have
required substitution of the implant, nor was any protrusion observed.
No patients presented flap necrosis, implant loss, wound infection,
haematoma, or seroma requiring surgical evacuation. There were no
signs of extracapsular rupture or intracapsular rupture. The mean
postoperative hospital stay was 5 days (range 3 - 7 days). During the
follow-up period, with a median of 2 years, there was no evidence of
local or distant disease relapse. Seven patients (32%) received adjuvant
chemotherapy and 6 patients (27%) received adjuvant radiotherapy.
Capsule formation requiring capsulotomy was also not observed.
Based on MRI examination, 9 patients (41%) presented signs of
capsular contracture manifested as an anteroposterior to mediolateral
ratio less than 2 (Figure 1). This correlates with 6 patients (27%) who
presented clinical findings of Baker grade II and III contracture. Only
one patient clinically considered to be Baker II did not present an
anteroposterior to mediolateral ratio less than 2, but the patient did
present a fibrous capsule thickness of 1.8 mm, and a lot of radial folds,
as well as a significant length of peri-implant fluid (more than 4 mm)
(Figure 2). All patients who underwent radiotherapy (6 patients) had
an anteroposterior to mediolateral ratio less than 2 and manifested
capsular contracture clinically (Baker grade II and III).
Student’s t-test detected a significant difference between mean
fibrous capsule thickness around the implant between patients that
underwent radiotherapy and those who did not (1.60 ± 0.37 mm
versus 0.89 ± 0.17 mm, t =5.44; p < 0.05). There was also a significant
difference between the mean value of maximum length of free fluid
around the implant according to Student’s t-test (3.78 ± 0.62 mm
in the group that underwent radiotherapy versus 2.93 ± 0.32 mm
in the group that did not, t =3.84; p < 0.05, Figure 3). Patients with
radiotherapy presented a mean value of latissimus dorsi muscle
flap thickness of 5 mm, while in those without radiotherapy 5.8
mm. However, the difference between mean thickness values of the
reconstructed muscle between tested groups was not statistically
significant (data not shown). No patients had signs of capsular
rupture or significant implant dishomogeneity.
Figure 3
Figure 3
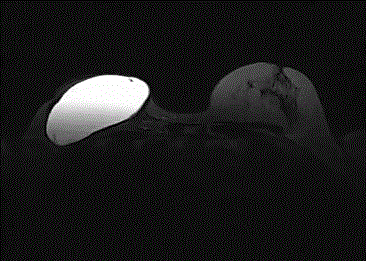
Axial T2-weighted turbo spin-echo fat suppressed image shows left
breast single lumen implant with length of free fluid approximately 6.5 mm in
a patient who underwent radiotherapy.
Figure 4
Figure 4
Axial TIRM water suppressed silicone image shows normal right
single lumen implant. The capsule is seen a dark ring around the implant.
Discussion
Breast conserving therapy is avoided in cases of primary breast cancer based on the size or the distribution of the tumour, or if breast conserving therapy would have a poor cosmetic outcome or local recurrence [10]. Some studies suggest that autologous tissue reconstruction provides better cosmetic results when compared to implant use, especially if it is deemed necessary to use radiotherapy [11,12]. The use of implants has advantages such as simplicity, shorter operation time and hospital stay, no extra scars, and no donor site morbidity. The main disadvantages of breast reconstruction with prosthesis include the risk of failure due to infection, rupture, extrusion, capsular contracture, the inability to withstand radiotherapy, and limited cosmetic results in comparison to autologous flaps [11]. The latissimus dorsi musculocutaneous flap is a popular method for breast reconstruction along with the transverse rectus abdominis musculocutaneous flap [13]. In selected patients, the latissimus dorsi myocutaneous flap provides adequate coverage of the prosthesis, and the skin island of the latissimus muscle flap can be incorporated into the defect after nipple-areola complex removal. This creates a tension-free muscular pocket, and provides adequate tissue coverage for the silicone implant [2,14,15]. The combined technique for reconstruction used in this study allowed for immediate breast volume replacement in patients who were not good candidates for more complex procedures and allowed for final adjustments during the postoperative period with minimal morbidity. The muscular flap provides a stable soft-tissue cover for the silicone implant, and protection against implant exposure in wound healing complications. In addition, the pocket exhibited little tension on the underlying silicone implant, allowing natural expansion without tension, and avoiding secondary upper pole fullness [1-3]. Another advantage of this combined method of breast reconstruction is also seen in this study, as none of the patients had signs of extracapsular or intracapsular implant rupture. The results of this study demonstrate that breast reconstruction with silicone implant and latissimus dorsi myocutaneous flap is a simple and consistent technique that has an important place among the main breast reconstruction procedures available at the moment. The success of the procedure depends on patient selection that is coordinated with the oncology surgeon, and on careful intraoperative and postoperative management. SSM and IBR for operable breast cancer is associated with a high level of patient satisfaction and low morbidity [4,5]. Evaluation of complications and cosmetic outcome is complex because of the significant variation in the sequencing of radiotherapy, the administration of systemic therapy, the duration of follow-up, and the techniques of radiation delivery [6]. For this purpose, a large clinical trial and longer follow-up periods are necessary for conclusions to be drawn that have adequate statistical power. The most common complication of SSM is skin flap necrosis because the skin envelope blood supply can become compromised during dissection [6], but none of our patients experienced this complication. IBR using prosthesis offers the best aesthetic results if postmastectomy radiotherapy is not needed. If radiotherapy is indicated, then delayed reconstruction is preferable to avoid potential aesthetic and radiation delivery problems [12]. Postmastectomy radiotherapy causes fibrosis and increases the incidence of capsular contracture, as seen in this study. MRI findings of capsular contracture according to our study do not correlate well with clinical findings using the Baker scale. MRI has been reported as a sensitive and specific method in the detection of capsular contracture. As can be seen in this study, MRI revealed incipient capsular contracture in three patients that was not seen clinically using the Baker scale. MRI depicts the capsule as a dark ring-like structure where thickness may vary (Figure 4). A formed capsule will contract the implant in cases where capsular fibrosis occurs. As a result of pressure, the sensation of tension will begin. The implant can assume an abnormal shape [9]. The MRI examinations confirm our conclusion that patients who undergo radiotherapy are more likely to develop capsular fibrosis and contracture compared to patients that do not undergo radiotherapy. These results can be explained considering an increased chronic inflammatory response during radiotherapy. Our further study, related to the follow-up of patients with breast cancer reconstruction, along with standard MRI techniques, will encompass silicone selective sequences as well. An outlined procedure should produce more accurate and objective information related to the development of capsular fibrosis and contracture.
Conclusion
This study shows that patients with IBR who undergo radiotherapy have more undesirable parameters. We consider that immediate reconstruction has better results if the operation is performed without additional radiotherapy. The Baker scale is not sufficient in determining the appearance of capsular contracture after SSM and IBR. Therefore, we emphasise that MRI can be a method for the analysis of breast reconstruction, especially for the detection of postoperative complications (such as capsular contracture), and when clinical signs are not evident (Baker scale I). From a radiological point of view, the challenge remains to expand our study further, investigate a larger number of patients in order to precisely define the parameters that describe each phase of contracture. Such knowledge will enable us to more accurately advise surgeons on their plans for further treatment of capsular contracture.
References
- Abdalla HM, Shalaan MA, Fouad FA, Elsayed AA. Immediate breast reconstruction with expander assisted latissimus dorsi flap after skin sparing mastectomy. J Egypt Natl Canc Inst. 2006;18(2):134-40.
- de la Torre JI, Fix RJ, Gardner PM, Vasconez LO. Reconstruction with the latissimus dorsi flap after skin-sparing mastectomy. Ann Plast Surg. 2001;46(3):229-33.
- Munhoz AM, Aldrighi C, Montag E, Arruda EG, Aldrighi JM, Filassi JR, et al. Periareolar skin-sparing mastectomy and latissimus dorsi flap with biodimensional expander implant reconstruction: surgical planning, outcome and complications. Plast Reconstr Surg. 2007;119(6):1637-49.
- Patani N, Devalia H, Anderson A, Mokbel K. Oncological safety and patient satisfaction with skin-sparing mastectomy and immediate breast reconstruction. Surg Oncol. 2008;17(2):97-105.
- Salhab M, Al Sarakbi W, Joseph A, Sheards S, Travers J, Mokbel K. Skin-sparing mastectomy and immediate breast reconstruction: patient satisfaction and clinical outcome. Int J Clin Oncol. 2006;11(1):51-4.
- Ueda S, Tamaki Y, Yano K, Okishiro N, Yanagisawa T, Imasato M, et al. Cosmetic outcome and patient satisfaction after skin-sparing mastectomy for breast cancer with immediate reconstruction of the breast. Surgery. 2008;143(3):414-25.
- Hardwicke JT, Prinsloo DJ. An analysis of 277 consecutive latissimus dorsi breast reconstructions: a focus on capsular contracture. Plast Reconstr Surg. 2011;128(1):63-70.
- Ho CM, Mak CK, Lau Y, Cheung WY, Chan MC, Hung WK. Skin involvement in invasive breast carcinoma: safety of skin-sparing mastectomy. Ann Surg Oncol. 2003;10(2):102-7.
- Vestito A, Mongieri FF, Ancona A, Minervini C, Perchinunno V, Rinaldi S. Study of breast implant rupture: MRI versus surgicall findings. Radiol Med. 2012;117(6):1004-18.
- Fajdic J, Djurovic D, Gotovac N, Hrgovic Z. Criteria and procedures for breast conserving surgery. Acta Inform Med. 2013;21(1):16-9.
- Kroll SS. Why autologous tissue? Clin Plast Surg. 1998;25(2):135-43.
- Pepe N, Pepe P, Carně D. [Breast reconstruction: a possible balance between oncological therapy, reconstructive methods and the patients]. Minerva Chir. 1999;54(3):143-56.
- Zhong T, Novak CB, Bagher S, Maass SW, Zhang J, Arad U, et al. Using propensity score analysis to compare major complications between DIEP and free muscle-sparing TRAM flap breast reconstructions. Plast Reconstr Surg. 2014;133(4):774-82.
- Hodgson EL, Malata CM. Implant-based breast reconstruction following mastectomy. Breast Dis. 2002;16:47-63.
- Nicholson RM, Leinster S, Sassoon EM. A comparison of the cosmetic and psychological outcome of breast reconstruction, breast conserving surgery and mastectomy without reconstruction. Breast. 2007;16(4):396-410.