Research Article
Utility of the Enoxaparin and Diclofenac in Prevention of Postoperative Abdominal Adhesions. Experimental Study in Murine Model
Gómez GGV* and Betancourt ET
Department of Experimental Surgery, Mexican Medicine School of La Salle University, Mexico
*Corresponding author: Gilberto Guzmán-Valdivia Gómez, Department of Experimental Surgery, Mexican Medicine School of La Salle University
Published: 24 Jan, 2018
Cite this article as: Gómez GGV, Betancourt ET. Utility
of the Enoxaparin and Diclofenac in
Prevention of Postoperative Abdominal
Adhesions. Experimental Study in
Murine Model. Clin Surg. 2018; 3: 1890.
Abstract
Postoperative abdominal adhesions constitute a condition that occurs in more than 90% of patients undergoing abdominal surgery, and cause major morbidity as intestinal obstruction, secondary infertility and chronic abdominal pain. The purpose of this study was to assess the usefulness of enoxaparin as inhibitor of the coagulation cascade and diclofenac as a non-steroidal anti-inflammatory drug to reduce inflammation, and thus the formation of abdominal adhesions. We used 90 Sprague Dawley rats divided into nine treatment groups of 10 animals each. Surgical adhesions were induced using a controlled brushing of the caecum with sterile cotton gauze and followed by a 3 mm long incision in ileum, repairing with suture. According with the experimental group, Enoxaparine and diclofenac were applied locally or systemic alone or in combination, while all experimental groups were compared against a suitable control group through a midline incision and terminal euthanasia. By far, the greater degree of formation of abdominal adhesions occurs in the non-treated control group I (p< 0.05). In contrast, groups III (enoxaparin SC), IV (diclofenac IP) and VIII (diclofenac IP and enoxaparin SC) induced a lower degree of postoperative abdominal adhesions (p< 0.01), and the combination of enoxaparin SC and diclofenac (IP) showed be better that when administered individually (p< 0.05). Results demonstrated that the combined subcutaneous enoxaparin and intraperitoneal diclofenac reduced the formation of postoperative abdominal adhesions significantly.
Introduction
Abdominal adhesions are vascularized and innervated connective tissue bridges, which form between the peritoneum, bowel and abdominal wall [1]. The intra-abdominal surgery adhesions can cause surgical re intervention, chronic abdominal pain, intestinal obstruction and infertility in women [2-4] has been described that up to 93% of patients who have had one or more previous surgery developed abdominal adhesions [5] 16 and 15% to 20% of female infertility due to postsurgical adhesions [4]. Postoperative Abdominal Adhesions (AAP) result from tissue damage either by incisions, cautery, suture or foreign objects that assaulted the parietal and visceral peritoneum, and this reacts forming scar tissue from the fibrin [6,7]. Fibrin is the end result of the coagulation cascade and it formed deposits, while there is a proliferation of fibroblasts with the objective of forming an Extra Cellular Matrix (ECM) and collagen, which favors the formation of adhesions. COX-2 is an enzyme that regulates the processes of angiogenesis on the development of postsurgical adhesions and inflammatory. The presence of tissue hypoxia and/or adhesion fibroblasts, COX-2 increases its expression [8]. We can summarize that the postoperative abdominal adhesions, are due to stimulation of the coagulation cascade and the imbalance between the fibrinogenesis and fibrinolysis in favor of the former associated with tissue hypoxia secondary to damage mesothelial, to their own response to the increase in the population of adhesion fibroblasts which inhibit the degradation of the extracellular matrix and inflammation. Strategies in search of decrease (AAP) postoperative peritoneal adhesion formation, based on some specific points in the adhesions path physiological origin have been numerous [3,9]. NSAIDs not drugs (NSAIDs) work by inhibiting the COX-1 and COX-2 avoiding the synthesis of prostaglandins and thromboxanes [2]. COX-2 inhibitors NSAIDs have been pharmacological agents that have shown to prevent post-surgical adhesions in various animal models, however, does not exist as such, significant evidence of the specific use of diclofenac(COX-1 and COX-2 inhibitor), medication use and low-cost [10-12]. Heparin is a sulfated glycosaminoglycan acid that stops the formation of fibrin. In a study with rats, was that the ministration of heparin at different doses is effective and safe (62.5 UI, 125 IU and 250 IU) in combination with carboxy methylcellulose, having better results with the highest dose of heparin [13]. The optimal dose for even braking adhesion formation is unknown, so it is necessary to identify the effective dose to prevent the formation of adhesions without increasing the risk of bleeding. In animal models, has been that Heparin Low Molecular Weight (LMWH) decreases the formation of adhesions, given both intraperitoneal and subcutaneous, but is not managed yet and not recommended dose [14,15]. The present work intends to find a mechanism that is effective, simple to implement and low cost in the prevention of the formation of postoperative abdominal adhesions through the reduction of inflammatory responses and coagulation with Diclofenac and the enoxaparin.
Materials and Methods
All experimental manipulations were performed according to
the Mexican Guidelines set forth in the NOM-062-ZOO-1999 and were approve by our local IACUC and the Ethics and Research
Commission of the Facultad Mexicana de Medicina, Universidad La
Salle, A.C, and Mexico.
Animals
Ninety male Sprague Dawley rats, weighing 400-450 g. were
used for this study. All animals were housed singly in conventional
conditions under an environmentally controlled animal facility
(temperature 21°C ± 2; relative humidity 55% ± 10%; and 12:12 hr
dark/light cycle), and were fed ad libitum with Lab Diet 5008® and
fresh water. Fasting for 12 hours prior surgery was established and
all animals were sedated before surgical preparation using 3 mg/kg
of Azaperone (Sural®) Preventative analgesia was implemented by
injecting Buprenorphine (0.05 mg/kg IM; Brospina®, Pisa) IM, and
anesthesia was induced with Tiletamine-zolazepam (3 mg/kg IP;
Zoletil®). All surgical procedures were carried out in a surgical suite
under sterile conditions using prior abdominal disinfection with
iodopovidone 10%.
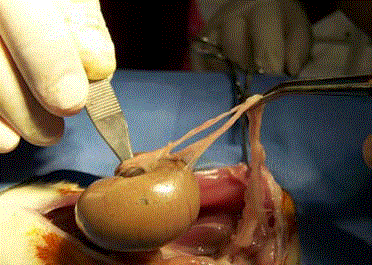
Surgical model for the induction of adherences
All animals were selected randomly to establish nine groups
treated as indicated in (Table 1), with 10 animals per group. A mid-line
incision was done over the muscular and peritoneal layer to expose
the abdominal cavity; once the caecum was located it was subjected
to repeated moderate brushing using a sterile cotton gauze without
injuring the adjacent intestinal loops, until punctuate bleeding
appeared (10-12 ± 1 strokes). The abrasion was always executed by
the same surgeon, followed by a 3 mm long incision located 5 cm
distant to the ileocolic junction and closed with interrupted sutures
5/0 Polypropylene (Atramat®), placed equidistantly over the defect.
The midline incision was closed in all groups using a continuous
suture layer with 3/0 Polypropylene (Atramat®), while the skin was
closed with 3/0 Atramat® Nylon.
Therapeutic procedure
The enoxaparin dose was determined based on the following
criterion: if the average prophylactic dose for a 70-kg human is 20
mg to 40 mg, i.e. 0.28 to 0.56 mg/kg; we opted to use the maximum
level to facilitate dosing, defined as 0.5 mg/kg/day. Consequently,
diclofenac was set at a dose of 2 mg/kg /day IM.
Clinical assessment of adherences
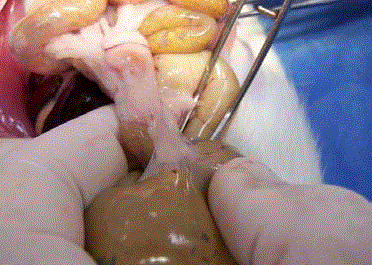
In order to assess adherence formation all rats were euthanized
with CO2 and subjected to laparotomy, from the xiphoid process
to the pubis. All findings were photographically documented using
a Kodak Easy Share digital camera (10X). Blind assessment of the
adherences was executed by two surgeons 14 days after surgery using
the scale of Nair, modified by Guzmán-Valdivia (Figure 1 and Table
2).
Statistical analysis
Statistical analyses were performed using the software package
Graph Pad Instat 3.1. The data is grouped as mean ± deviation
standard. Means were compared by ANOVA. The non-parametric
Kruskal-Wallis test was employed to define differences among
the groups, and depending on the results the multiple comparison
test of Dunn was applied to determine if significant differences
existed among the treated groups. For the comparison among two
independent groups the Mann-Whitney U Test was used. Results are
expressed with a confidence interval of 95%. Probability values below
0.05 were considered statistically significant. In order to determine the inter observer concordance used the Kappa Coefficient (κ).
Figure 1A
Figure 1B
Figure 2
Table 1
Table 1
Arrangement and Treatment of Nine Experimental Groups. IP:
Intraperitoneal, SC: Subcutaneous.
Table 2
Results
A total of 90 rats were sacrificed on postoperative day 14 to
evaluate macroscopically adhesion formation. They were compared
the experimental groups with the control group and between groups
with treatment. During the investigation, died 5 animal experiments,
which were replaced, all they underwent autopsy. The causes of death
were: two cases with anesthetic complications, and evisceration,
intra-abdominal and abdominal sepsis, hemorrhage, a case of each.
The results reveal that the control group significantly formed greater
degree of adhesions in comparison to groups with treatment (Table 3).
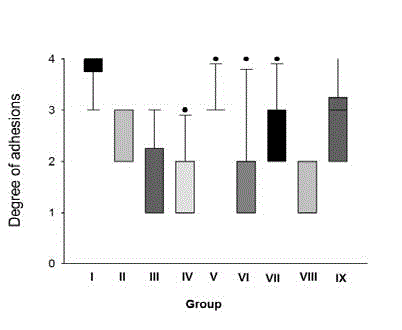
The comparison between the groups with treatment (Figure 2) shows
that groups III (SC enoxaparin), Group IV (IP diclofenac), Group VI
(IP enoxaparin and IP diclofenac) and the Group VIII (SC enoxaparin
and IP diclofenac) demonstrated a lower degree of postoperative
abdominal adhesions. (Table 4) summarizes the comparison among
the averages of each group exhibiting less adhesion formation, hence
observing:
• Subcutaneous administration of enoxaparin was superior
that the intraperitoneal route.
• Intraperitoneal administration of diclofenac was superior
to the intramuscular route.
• There was no difference between subcutaneous applications
of enoxaparin versus the intraperitoneal diclofenac route.
• The expected synergistic action of the combination of
subcutaneous enoxaparin and intraperitoneal diclofenac failed;
therefore, it was not superior to the independent administration of
each drug tested.
• The kappa coefficient (k) was 0.81 (very good), which
means that the evaluation of adhesions between observers is the same.
Discussion
Abdominal postoperative adhesions are a significant clinical
problem; they can be the cause of multiple surgeries and escalate with
repetitive formation constituting a greater risk of frequent hospital
readmissions for surgical treatment, including intestinal resections,
while standing as an important cause of infertility in woman and
chronic abdominal pain. Such events have a significant economic
impact for the patient and the institutional health care systems [1-
3]. According to the pathophysiology of the PPA formation, factors
involved include the activation of the inflammation process of the
coagulation cascade to form fibrin bridges, under a poor process
of fibrinolysis, amid a tissular hypoxic environment concomitant
to prior damage to the mesothelium or peritoneum. In short,
it can then be asserted that these events concomitantly led to a
diminished activity of the activator of plasminogen, to cytological and
biochemical alterations of the extracellular matrix being formed, an
increase of fibrogenesis and angiogenesis and a reduction of apoptosis
[16,17]. Numerous strategies have been attempted to prevent the
formation of PPA, including mechanical barriers, chemicals and
various pharmacologic drugs with no definite results for humans.
In this work, we attempted to validate the efficacy of a LMWH
(enoxaparin) to prevent the formation of PPA, by interfering with
the coagulation cascade to form fibrin bridges, hence differentiating
its efficacy among its systemic and local administration. In addition,
we aimed to determine the effectiveness of a commonly used NSAID
(diclofenac), a COX-2 inhibitor, and its ability to limit the peritoneal
inflammatory response and differentiate it´s effects either when
administered systemically or locally. Finally, we wanted to find
out if the suggested synergistic combination of both drugs had any
beneficial effect to limit the formation of PPA. On the other hand,
as reported for Opitz [18], Watson [19] and Jansen [20] in their
studies for the prevention of PPA with heparin, they resulted most
controversial given that different results were observed, when in their
experiences failed to show human efficacy; in contrast, the research
of Kement [21] and Sharifi [16] found a decrease in the formation
of postoperative adhesions associated to the use of a physical barrier
of carboxymethyl cellulose and acid hyaluronic. Under the concept
that by diminishing the formation of fibrin or limiting its activation
during the coagulation cascade, is then that various experiments have been devised and executed, using heparins of low molecular
weight, predominantly Nadroparin by the intraperitoneal route [22]
and enoxaparin, using a twofold dose with respect of the estimated
prophylactic dose, as was reported by Arikan, et al. [23] who employed
1 mg/kg, injected subcutaneously for 7 days.
In our experiment, we used enoxaparin at a dose of 0.5 mg/
kg for being assessed as having prophylactic effects and being less
risky to induce internal bleeding. In this case, the drug was applied
both, intraperitoneal (locally) and subcutaneously (systemic) for
seven days, finding its best prophylactic effect against the formation
of PAA when injected systemically (subcutaneously). In the search
of new treatments to decrease adhesion formation by inhibiting
the inflammatory process, through the use of Non Steroidal Anti-
Inflammatory Drugs (NSAIDs), specifically COX-2 inhibitors, as
seen in the cases of Rofecoxib [24] administered by the oral route
and Celecoxib compared by Greene AK, et al. [25] against other
anti-inflammatory drugs such as aspirin, naproxen, ibuprofen,
indomethacin and Rofecoxib (oral), he concluded that Celecoxib
resulted the best anti-inflammatory drug able to reduce the formation
of PAA. In our study we wanted to assess the efficacy of diclofenac,
a commonly used anti-inflammatory COX-1 and COX-2 inhibitor,
easy to acquire and inexpensive, which according to our knowledge it
has never been studied before in any experimental animal model. In
this case, diclofenac was applied using the therapeutic dose according
to animal-weight, and further injected locally and systemically,
finding its best effect when used by the intraperitoneal route and
administered as a single dose per day. Although there are studies
combining physical barriers primarily based on sodium hyaluronate
and methylcellulose with LMWH [26], there is not prior experience
reporting the combined effects of NSAIDs and LMWH, expecting
that the synergistic action of both drugs would led to a decrease in
the formation of PAA by inhibiting two different pathways: fibrin
formation, when the coagulation cascade is activated, and limiting the
active factors of inflammation. In our experiment, we found that the
combination of systemic Enoxaparin with diclofenac intraperitoneal
have a synergist action, demonstrating lower incidence of
postoperative adhesions that applying the drugs alone. With these
results we are performing the experiment in a porcine model using the
same drugs on routes of greater efficiency, trying to assess the results
in an animal model with more complex and more like the human
inflammatory response and measure the response of inflammation
and fibrosis by microscopic evaluation and biochemical markers.
Table 3
Table 4
Acknowledgment
The authors thank the Universidad La Salle for the financial support through the project "New surgical procedures in diseases of the digestive system and abdominal wall".
References
- Arung W, Meurisse M, Detry O. Pathophysiology and prevention of postoperative peritoneal adhesions. World J Gastroenterol. 2011;17:4545-53.
- Ergul E, Korukluoglu B. Peritoneal adhesions: facing the enemy. Int J Surg. 2008;6(3):253-60.
- Brüggmann D, Tchartchian G, Wallwiener M, Münstedt K, Tinneberg HR, Hackethal A. Intra-abdominal adhesions: definition, origin, significance in surgical practice, and treatment options. Dtsch Arztebl Int. 2010;107(44):769-75.
- Liakakos T, Thomakos N, Fine P, Dervenis Ch, Young L. Peritoneal adhesions: etiology, pathophysiology and clinical significance. Recent advances in prevention and management. Dig Surg. 2001;18(4):260-63.
- Menzies D, Ellies H. Intestinal obstruction from adhesion: how big is the problem?. Ann R Coll Surg Engl. 1990;72(1):60-3.
- Maciver A, McCall M, Shaphiro J. Intra-abdominal adhesion: cellular mechanisms and strategies for prevention. Int J Surg. 2011;9(8):589-94.
- Werner M, Galecio J, Bustamante H. Adherencias abdominales postquirúrgicas en equinos: patofisiología, prevención y tratamiento. Arc Med Vet. 2009;41:1-15.
- Butureanu SA, Butureanu TA. Pathophysiology of adhesions. Chirugia. 2014;109(3):293-8.
- Ward BC, Panitch A. Abdominal Adhesions: Current and Novel Therapies. J Surg Res. 2011;165(1):91-111.
- Yong IK. Comparative Study for Preventive Effects of Intra-Abdominal Adhesion Using Cyclo-Oxygenase-2 Enzyme (COX-2) Inhibitor, Low Molecular Weight Heparin (LMWH), and Synthetic Barrier. Yonsei Med J. 2013;54(6):14971-7.
- Wei G, Chen X, Wang G, Jia P, Xu Q, Ping G, et al. Inhibition of ciclooxigenase-2 prevents intra-abdominal adhesions by decreasing activity of peritoneal fibroblasts. Drug Des DevelTher. 2015:9:3083-98.
- Shahzmani P, Ashrafzadeh M, Jahanshahi Am Sotoudeh A. Effects of dexamethasone, piroxicam and sterile aloe vera extracts on prevention o postoperative peritoneal adhesion formation in rat. Advances in Environmental Biology. 2012;6(11):2851-65.
- Gutt CN, Oniu T, Schemmer P, Mehrabi A, Büchler MW. Fewer adhesions induced by laparoscopic surgery?. Surg Endosc. 2004;18(6):898-906.
- Kement M, Censur Z, Oncel M, Buyukokuroglu M, Gezen F. Heparin for adhesion prevention: comparison of three different dosage with separafilm in a murine model. Int J Surg. 2011;9(3):225-8.
- Sharifi S, Derakshanfar A, Pourjafar M, Mohamadnia A, Charlang K. Effect of heparin in prevention of experimental abdominal adhesions in rat. Iranian J Vet Surg. 2007;2(3):24-31.
- Saed GM, Diamond MP. Molecular characterization of postoperative adhesions: The adhesion phenotype. J Am Assoc Gynecol Laparosc. 2004;11(3):307-14.
- Hobbs MS, Mai Q, Knuiman MW, Fletcher DR, Ridout SC. Surgeon experience and trends in intraoperative complications in laparoscopic cholecystectomy. Br J Surg. 2006;93(7):844-53.
- Opitz I, van der Veen HC, Braumann C, Ablassmaier B, Führer K, Jacobi CA. The influence of adhesion prophylactic substances and taurolidine/heparin on local recurrence and intraperitoneal tumor growth after laparoscopic-assisted bowel resection of colon carcinoma in a rat model. Surg Endosc. 2003;17(7):1098-104.
- Watson A, Vandekerckhove P, Lilford R. Liquid and fluid agents for preventing adhesions after surgery for subfertility. Cochrane Database Syst Rev. 2000;2:CD001298.
- Jansen RP. Failure of peritoneal irrigation with heparin during pelvic operations upon young women to reduce adhesions. Surg Gynecol Obstet. 1988;166(2):154-60.
- Kement M, Censur Z, Oncel M, Buyukokuroglu M, Gezen F. Heparin for adhesion prevention: comparison of three different dosage with separafilm in a murine model. Int J Surg. 2011;9(3):225-8.
- Kutlay J1, Ozer Y, Isik B, Kargici H. Comparative effectiveness of several agents for preventing postoperative adhesions. World J Surg. 2004;28(7):662-5.
- Arikan S, Adas G, Barut G, Toklu AS, Kocakusak A, Uzun H, et al. An evaluation of low molecular weight heparin and hyperbaric oxygen treatment in the prevention of intra-abdominal adhesions and wound healing. Am J Surg. 2005;189(2):155-60.
- Aldemir M, Oztürk H, Erten C, Büyükbayram H. The preventive effect of Rofecoxib in postoperative intraperitoneal adhesions. Acta Chir Belg. 2004;104(1):97-100.
- Green AK, Alwayn IP, Nose V, Flynn E, Sampson D, Zurakowski D, et al. Prevention of Intra-abdominal adhesion using the antiangiogenic COX-2 inhibitor celecoxib. Ann Surg. 2005;242(1):140-6.
- Kaptanoglu L, Kucuk HF, Yegenoglu A, Kurt N. Effects of Seprafilm and heparin in combination on intra-abdominal adhesions. Eur Surg Res. 2008;41(2):203-7.