Case Report
Inferior Vana Cava Occlusion Revascularization: Case Report
Kintur Sanghvi*
Department of Interventional Cardiology, Deborah Heart and Lung Center, Philadelphia School of Osteopathic
Medicine, USA
*Corresponding author: Kintur Sanghvi, Department of Interventional Cardiology, Deborah Heart and Lung Center, Philadelphia School of Osteopathic Medicine, USA
Published: 16 Nov, 2017
Cite this article as: Sanghvi K. Inferior Vana Cava
Occlusion Revascularization: Case
Report. Clin Surg. 2017; 2: 1729.
Abstract
This case illustrates management of a patient with disabling symptoms from Inferior Vena Cava (IVC) filter occlusion. Inferior vena cava obstruction from organized thrombus and fibrosis can lead to severe bilateral lower extremity edema despite chronic anticoagulation therapy. Complete Endovascular revascularization with self-expanding stents is a feasible and attractive treatment option with remarkable clinical response.
Introduction
Chronic occlusion of the Inferior Vena Cava (IVC) is an uncommon condition that could be caused by variety of underlying diseases. Patients can present with a myriad of signs and symptoms, depending on collateral venous drainage. We present a patient with chronic, post-thrombotic occlusion of IVC filter with intractable bilateral lower extremity swelling. Patients with similar clinical conditions are often misdiagnosed to have lymphedema and not offered revascularization [1-3]. The patient was treated with progressive balloon dilation and self-expanding stents in a cardiac catheterization lab resulting in remarkable clinical improvement.
Case Presentation
An 85-year-old-female with past medical history of right lower extremity Deep Vein Thrombosis
(DVT) underwent non-retrievable IVC filter implantation in 2004. She initially presented to us
in 2013 with progressive worsening of bilateral lower extremity pitting edema and past medical
history of Lupus antibody, Coronary artery disease with previous coronary stents, and peripheral
artery disease with previous bilateral iliac stenting. She was on chronic anticoagulation therapy with
warfarin since 2004. Her ultrasound evaluation then showed bilateral femoral vein occlusion from
chronic DVT. Because of interactive progression of the bilateral swelling despite anticoagulation
therapy, an endovascular procedure was performed via bilateral popliteal vein access. Balloon
venoplasty was performed after 30 hr of ultrasound enhanced thrombolytic infusion with EKOS
catheter (EKOS Corporation, Bothell, WA) in the occluded IVC filter and bilateral iliac vein. Despite
recanalization and re-establishment of outflow in the IVC and transient improvement of the lower
extremity painful swelling, on subsequent follow-up after 6 months she started having even worst
progression of bilateral lower extremity swelling.
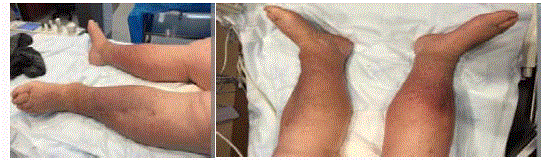
Over the year of follow-up after the initial endovascular venous intervention patient continued
to have worsening bilateral lower extremity swelling, pain, and disabling discomfort with chronic
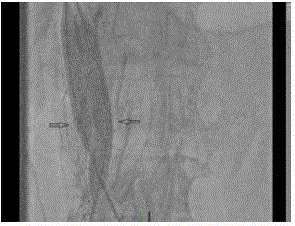
venous stasis changes (Figure 1A and 1B). She was brought back to the cath-lab. Ultrasound guided
bilateral common femoral vein access was achieved using a 6 French sheath. Bilateral femoral
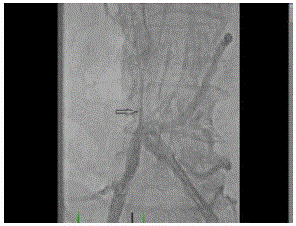
venography showed partially obstructive thrombotic disease of bilateral iliac veins. The IVC was
occluded through the IVC filter (Figure 2). A collateral tributary (superficial epigastric vein) drained
the blood to IVC cranial to the occlusion in-side the IVC filter. Weight based heparin (90 unit/kg)
was given IV to achieve therapeutic activated clotting time.
A very aggressive wire escalation technique used along with crossing catheter was not successful.
Multiple different wires including a stiff angle glide (Terumo Corporation, summerset NJ) wire
were unsuccessful in crossing. Ultimately, distal end (back end) of the stiff angled glide wire and
0.035 Quick-Cross catheters (Spectranetics Corporation) were used with contralateral fluoroscopy
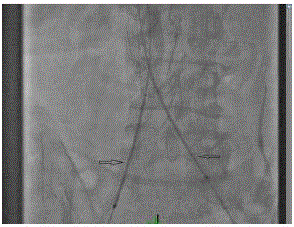
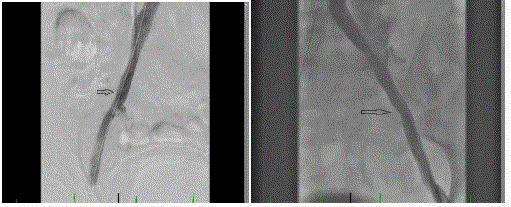
guidance to remain inside the frame of the filter but cross through the occlusion successfully. A
0.035” J-wire was inserted in to the SVC through the Quick-cross catheter. (Figure 3) Balloon
venoplasty was performed using a 10 mm balloon for allowing crossing the IVC filter occlusion from the left femoral vein approach also. Sheaths were exchanged for a 9 French sheath in each femoral vein. After pre-dilating on both
sides with 10 mm balloon, 14 mm x 10 mm balloons were advanced
simultaneously into the IVC via both access sites and simultaneous
kissing balloon inflations were performed. Post-venoplasty a
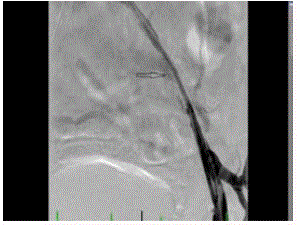
significant flow through the IVC was re-established. Two 14 mm x
80 mm self-expanding Protégé stents were deployed in simultaneous
kissing fashion extending superior and inferior to the IVC filter
(Figure 4). Subsequently, two 14 mm x 60 mm Protégé stents were
deployed again in simultaneous kissing fashion overlapping with
the first stent and extending into the ipsilateral iliac veins. The left
external iliac vein still had severe stenosis (Figure 5) which was
treated with 12 mm x 80 mm Protégé self-expanding stent that was
post dilated with a 10 mm balloon. Stents were post-dilated with 14
mm x 60 mm balloons and simultaneous kissing balloon inflations
were performed (Figure 6). At the end of the procedure, robust blood
flow was observed through bilateral iliac veins (Figure 7A and 7B)
& IVC (Figure 8). Bilateral venous access was removed with manual
compression for hemostasis. Patient was discharged home the next
morning with continued treatment with Coumadin and Aspirin.
In 24 hr post-intervention patient had 4100 ml urine output with
positive balance of 2800 ml. Patient continued to have improvement
in swelling and pain. During the one-month, follow-up patient had
venous ultrasound showing patency of the venous flow through
bilateral iliac veins.
Figure 1A and B
Figure 2
Figure3
Figure 4
Figure 4
Protégé stents were deployed in simultaneous kissing fashion
extending superior and inferior to the IVC filter.
Figure 5
Figure 6
Figure 7A and 7B
Figure 8
Discussion
The IVC filter is a commonly used device for the prevention of
Pulmonary Embolism (PE). Increased use of IVC filters over past
few decades has led to an increasing frequency of long-term filterrelated
complications. These complications include, but are not
limited to local thrombosis and increased risk of recurrent lower
extremity Deep Vein Thrombosis (DVT) [1-3]. The incidence of DVT and IVC thrombosis after filter placement varies widely (6%
to 37%) in different patient populations [4,5]. Although previous
recommendations and guidelines discussed endovascular treatment
options in de novo DVT in non-filter-bearing IVC and iliofemoral
thrombosis, there are more recent reports describing the safety and
outcome of endovascular treatment for filter-bearing IVC thrombosis
[6-8]. If the patient has, a thrombosed filter-bearing IVC with total
venous occlusion, significant lower extremity edema and pain are
often present and severe complications such as sensory deficits,
venous stasis, or ulceration may also be present [9]. For patients that
are good anticoagulation candidates and caval filtration is not needed,
stenting of the IVC is a choice. A recent retrospective review [10] of
708 patients with chronic non-malignant post-thrombotic iliocaval
outflow obstruction included 25 patients with thrombosed IVC with
occluded IVC filters. This study revealed that stenting across the filter
may be safely performed with no apparent tear of the IVC, clinically
manifesting bleeding, or PE. There was no mortality and only minimal
morbidity associated with filter baring IVC stenting. The patency was
related to the severity of post-thrombotic disease. A comparison of
occlusions with and without IVC filters in same study showed no
significant difference in cumulative primary and secondary patency
rates at 56 months indicating that the presence of filters did not affect
the outcome.
We are reporting this case with three primary discussion
objectives. (1) Although multiple reports of feasibility, and success of
endovascular treatment of an IVC occlusion have been published in
vascular surgery & interventional radiology literature; interventional
cardiologist with the endovascular skills of crossing chronic total
occlusion using wide array of wires, crossing catheters and hybrid
access approaches are likely to have similar or higher success in
treating such complex venous occlusive disease. The knowledge
of the pharmacokinetics and pharmacodynamics of anticoagulant
and thrombolytic agents may further enhance success rate of
interventional cardiologists. Because of very low risk of requirement
of surgery to treat any complications, such procedure can be
performed in cardiac catheterization lab. (2) This case example could
be used to discuss wiring technique for IVC occlusion. A 0.014”
wire does not have required tip load and body to cross through
tough fibrosis from organized thrombus in IVC. A 0.018” wire
can be used along with Crossing Catheter (CC) for initial attempts
to cross the proximal fibrous cap. A wire with one-piece core and
spring coil design can be used along with a 0.018” CC or Over the
Wire (OTW) balloon. Shaping the wire with a 450 band 2 mm - 3
mm from the tip is useful to cross Chronic Total Occlusion (CTO).
The CC is advance over the wire to the proximal fibrous cap. The
wire is advance through the CC or OTW balloon and spin in one
direction very rapidly with forward pressure to give a drilling effect.
The examples of such wires are V 18 Control wire (Boston Scientific)
or Treasure 12 (Asahi Corporation). For précised rapid spinning of
the wire in one direction a device, SPINR (Merit Medical), can be
used to adjust the speed from 0-2500 Rounds per Minute (RPM).
The tip load against the lesion normally changes according to the
length of wire extended from the tip of CC. For the CTO wires like
Treasure or Astato wire the tip load dramatically increases with only
short wire extending out of the CC. The tip load can increase by up to
10 times with such maneuvers [9]. If the initial wire is unsuccessful
in crossing the next 0.018” wire for escalation is Astato 30 (Asahi
Corporation). Astato 30 is a high-penetration wire with tapered tip
and 30 gram tip load that can break through very tough fibrous tissue.
The effective tip load of an Astato wire depending on the distance out
of CC can be much higher and that along with tapered tip requires
careful control and vigilance. The other wiring technique is to create
a “knuckle” using a 0.035” soft tip and stiff shaft wires such as stiff
angled glide wire (Terumo Corporation) along with a 0.035”CC.
When the tip of wire engages the proximal cap body of the stiff
wire is pushed forcefully to create a loop which in turn causes blunt
dissection and less likely to cause perforation. The wire and CC is
advance with force simultaneously under the fluoroscopic guidance.
This technique is very useful technique with high success rate to cross
fibrous IVC occlusion. A wire puncture through the wall of fibrosis of
occluded IVC, as long as the CC is not advanced through, is unlikely
to cause significant bleeding. Simultaneous antegrade and retrograde
approach can be used if only antegrade approach from the femoral
vein access fails. Basilic vein access in the right arm or jugular vein
access is the choices for retrograde wiring of IVC occlusion. Similar
escalation as described above can be used simultaneously from both
sides in a hybrid approach. When above described wiring techniques
fail careful consideration of alternate technique should be weighed
for risk benefit analysis. We previously reported a technique of
using Brockenbrough (septal puncture) needle to cross fibrotic IVC
occlusion [10]. Frequent changes of image intensifier in contralateral
angles are crucial while crossing through chronic total occlusion. In
this case in particular contralateral angles were used to stay within
the frame of IVC filter while advancing the back-end of stiff angled
glide wire along with a 0.035” CC. Backend of wire is very stiff and
can penetrate through resilient fibrous tissue such as in this case.
With every cm advancement of the wire the CC was advanced
fixing the wire. Frequent change of fluoroscopy angle confirmed the
wire’s progress within the IVC filter fraWhile the number of IVC
filter placement is increasing steadily, the optional filter accounts
for more than half of all filters. A large proportion of patients have
permanent IVC filters placed while patient’s clinical condition only
requiring retrievable IVC filter. For many patients the retrievable
filters are left in-situ and not retrieved after clinical need is over. Such
practice trends demand for societies to come up with an evidencebased
guideline for appropriate use criteria similar to management of
coronary artery disease and peripheral artery disease. These guidelines
should also advice the importance of clinical and imaging follow-up
after IVC filter placement.
References
- Hammond CJ, Bakshi DR, Currie RJ, Patel JV, Kinsella D, McWilliams RG, et al. Audit of the use of IVC filters in the UK: Experience from three centers over 12 years. Clin Radiol. 2009;64:502-10.
- Spencer FA, Bates SM, Goldberg RJ, Lessard D, Emery C, Glushchenko A, et al. A population-based study of inferior vena cava filters in patients with acute venous thromboembolism. Arch Intern Med. 2010;170(16):1456-62.
- Streiff MB. Vena caval filters: a comprehensive review. Blood. 2000;95(12):3669-77.
- Given MF, McDonald BC, Brookfield P, Niggemeyer L, Kossmann T, Varma DK, et al. Retrievable Gunther Tulip inferior vena cava filter: Experience in 317 patients. J Med Imaging Radiat Oncol. 2008;52(5):452-7.
- Kalva SP, Athanasoulis CA, Fan CM, Curvelo M, Geller SC, Greenfield AJ, et al. “Recovery” vena cava filter: Experience in 96 patients. Cardiovasc Intervent Radiol. 2006;29(4):559-64.
- Arabi M, Krishnamurthy V, Cwikiel W, Ranjith V, Thomas WW, John R, et al. “Endovascular treatment of thrombosed inferior vena cava filters: Techniques and short-term outcomes.” Indian J Radiol Imaging. 2015;25(3):233-8.
- Neglén P, Oglesbee M, Olivier J, Raju S. Stenting of chronically obstructed inferior vena cava filters. J Vasc Surg. 2011;54(1):153-61.
- Robbins MR, Assi Z, Comerota AJ. Endovascular stenting to treat chronic long-segment inferior vena cava occlusion. J Vasc Surg. 2005;41(1):136-40.
- Waksman R, Saito S. Chronic total occlusion: a guide to revascularization. Wiley-Blackwell. 2013;149.
- Patel T, Sanghvi K, Shah S, Fonseca K. Successful stenting of a complex inferior vena cava stenosis using a modified sharp recannulization technique. Catheter Cardiovascular Interv. 2001;52(4):492-5.