Case Report
Benign Foot Drop: Large Cystic Peroneal Schwannoma
Ridvan Alimehmeti1*, Arben Rroji2, Teona Bushati3, Florian Dashi1, Arba Cecia4, Myfit Saraci1
and Arsen Seferi1
1Department of Neurosciences, University of Medicine, Albania
2Department of Radiology, Service of Neuroradiology, University Hospital Center “Mother Theresa”, Tirana, Albania
3Department of Pathology, University of Medicine, Tirana, Albania
4George Mason University, Fairfax, Virginia, Tirana, Albania
*Corresponding author: Ridvan Alimehmeti, Department of Neurosciences, University of Medicine, Tirane, Albania, Kongresi Manastirit, 270, Albania
Published: 25 Oct, 2017
Cite this article as: Alimehmeti R, Rroji A, Bushati T, Dashi
F, Cecia A, Saraci M, et al. Benign
Foot Drop: Large Cystic Peroneal
Schwannoma. Clin Surg. 2017; 2: 1695.
Abstract
We report an exceptional case of large cystic Schwannoma of peroneal nerve causing foot drop. The
clinical, radiological and electrophysiological characteristics are discussed. The cyst was surgically
removed. Careful detachment and decompression of the nerve fibers was followed by gradual
recovery from neurological deficit in six months. Histological examination confirmed the diagnosis
of cystic Schwannoma. There is no local recurrence in more than two years follow-up.
Keywords: Cystic; Schwannoma; Peripheral; Nerve; Foot-drop
Introduction
Schwannoma is benign peripheral nerve tumor that grows slowly and gives rise to neurological deficit due to the compromised function of the fascicle (s) from which it originates, or compression exerted from the growing tumor to the rest of fascicles of the affected nerve contained under the same epineurium. In most of the reported cases peripheral nerve Schwannoma is solid tumor. 5% to 6% of cases are reported to be partly cystic [1]. In such cases the cystic component is mostly heterogenic, sometimes hemorrhagic or necrotic in its content. Only very few cases are reported as purely cystic Schwannoma harboring homogenous liquid compound.
Patients and Methods
A 45-years-old man complained of progressive fatigue in lifting the left leg and especially the
big toe. He had noticed a swelling in the lateral upper part of the left leg. The difficulty in walking
worsened during the last two months before coming to our attention. Physical examination revealed
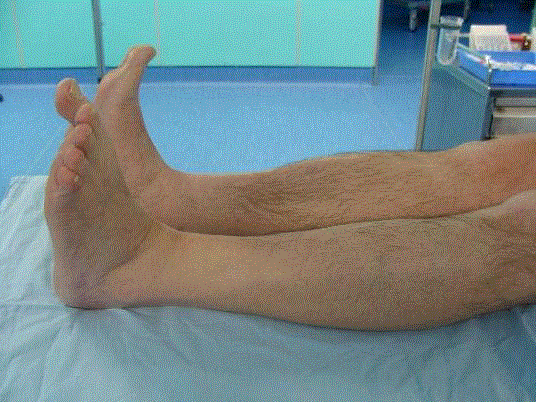
complete deficit of extensor hallucis longus (Figure 1). The patient was incapable to extend the big
toe and there was 3/5 of muscle strength of foot and other toes extension, which caused evident step
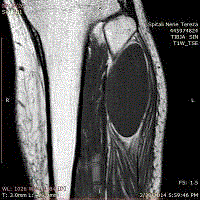
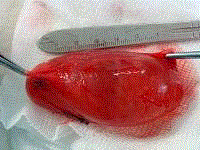
page gait. MRI of the left leg showed large cystic lesion 9 cm x 6 cm x 4 cm (Figure 2) extending from
the head of left fibula downward the peroneal muscles. It had an oval shape and thin capsule with
homogenous liquid content inside. Electrophysiological examination revealed conduction block
of the left common peroneal nerve. Routine laboratory blood work was negative. Blood test for
echinococcosis was negative as well. However, in an endemic area for echinococcosis we considered
needle aspiration caring a potential risk of dissemination and in case of false negative blood tests
for echinococcosis. The possibility of conclusive diagnosis was very small based on the tests over
the liquid inside the cyst, even if we would consider the benign tumor of the nerve as an option.
Hence needle aspiration of the cyst was omitted. We offered the patient the alternative of spinal
epidural anesthesia or general anesthesia. Complete surgical removal of the cyst was achieved under
general anesthesia. A lazy S skin incision over the lump in the lateral proximal part of the left leg was
followed by the opening of the superficial fascia exposed the cyst formation. The first step was the
exposure of the common peroneal nerve (CPN) in its passage around the neck of fibular head. The
CPN was isolated and a small branch of two millimeters wide fascicle was dissected from the rest
of the nerve fascicles. This fascicle gave rise to the cyst which extended down and inward the lateral
part of the leg. Then a longitudinal incision was done on the epineurium over the cyst from proximal
to distal pole. The location of the cyst was in between the peroneal muscles. The cyst extended for
9 cm from proximal to distal pole. The thin transparent layer of tissue over the cyst was handled
very carefully and dissected from the cyst wall in a longitudinal opening and then 360 degrees
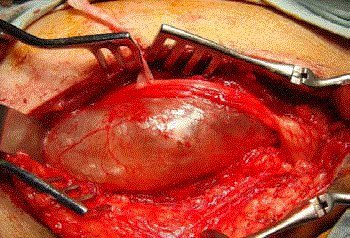
around the cyst. The de-tensioned layer was a bundle of the nerve fascicles compressed by the huge
cyst. They were progressively freed from the cystic wall (Figure 3) until the cyst was thoroughly peeled off from the rest of the nerve fascicles that had become layers superficial to the cyst. The fascicle that gave rise to the cyst in its distal
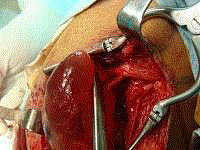
pole was then isolated (Figure 4). Both the fascicle that gave rise to
the cyst was cut 4 mm from the cyst wall respectively (Figure 5) and
the cyst totally removed without being ruptured. The remaining
fascicles of the peroneal nerve were decompressed. The wound was
closed and the patient made an uneventful recovery discharged the
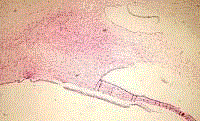
next day of surgery. Hematoxylin eosin stain demonstrated compact
hypercellular proliferation with narrow, elongated cells, with tapered
ends interspersed with collagen fibers. Nuclear palisading around
fibrillary process was seen. Tumor cells have ill-defined cytoplasm,
dense chromatin and there is some degenerative nuclear atypia
(ancient change). Multiple cystic spaces were present. Mitotic figures
are not seen. These characteristics were compatible with the diagnosis
of CS (Figure 6).
The patient made a gradual full recovery of the neurological
deficit. He was walking without difficulty with the left leg six months
from surgery. In more than three years follow up there is no local
recurrence.
Figure 1
Figure 2
Figure 3
Figure 4
Figure 5
Figure 6
Figure 6
Histological view HE x 20. Compact hypercellular proliferation
with narrow, elongated cells, with tapered ends interspersed with collagen
fibers. Nuclear palisading around fibrillary process is seen. Tumor cells have
ill-defined cytoplasm, dense chromatin and there is some degenerative
nuclear atypia (ancient change). Multiple cystic spaces are present. Mitotic
figures are not seen
Discussion
Foot-drop due to huge CS is extremely rare. There are reported
cases of peroneal nerve solid Schwannoma [2]. In front of cystic
formation in the leg causing foot-drop careful differential diagnosis
should be elaborated before tailoring treatment strategies. When the
cyst is in contact with knee and tibiofibular joint the diagnosis of
synovial or ganglion cyst should be taken in account. MRI study of
the intensity of liquid content may reliably distinguish synovial from
muscular, hydatid, abscess, hematoma or seroma.
Schwannoma is a benign tumor of the nerves arising from Schwann
cell of the nerve fibers. It is mostly solid and rarely mixed cystic-solid
tumor [2,3]. There is not enough evidence so far, on the mechanism
of CS formation. There are hypothesis of cystic transformation by degeneration of the Antoni B solid portion of Schwannoma giving
rise to a larger cyst [4]. Furthermore ischemic necrosis due to tumor
growth or intratumoral hemorrhage may result in cyst formation
within the tumor. The cystic fluid is reported to be replaced relatively
fast, considering its recurrence in a few days in reported cases where
needle aspiration has anticipated surgical removal [5]. It seems that
the dimension of the cyst depends on the gradient of pressure in and
outside of it. When the cystic Schwannoma grows in endeavors like
the intermuscular space of a limb as in presented case, or cavities such
as thoracic [1], pelvic [6] or regions like axilla [5], the cyst may grow
to huge dimensions. In presented case the liquid within the cyst was
homogenously light yellow colored, without debris inside.
Large CS of peripheral nerves is very rarely reported [3,7-9]. There
are few reports of CS located inside body cavities: vertebral canal [10],
thoracic cavity arising from intercostal nerves or recurrent laryngeal
nerve with cervical and mediastinal extension [11], abdominal cavity
with extension into the pancreas [12] or inside the orbit [13]. In the
case of cystic Schwannoma, the fine needle aspiration is not helpful
in the cytological diagnosis [14,5]. The differential diagnosis of CS
include: hematoma, ganglion or synovial cyst whenever located in
joint vicinity; abscess, seroma, cystic epidermoid, myxoma, and in
endemic areas like in our case, the intramuscular hydatid cyst. MRI
can highly contribute to differentiate these entities [10,15]. Hydatid
cyst is to be considered in endemic areas like Mediterranean, in order
to avoid seeding through needle aspiration [16]. In this case we did
not proceed with needle aspiration and biopsy considering that our
country is endemic for hydatid disease. On the other hand, a false
malignant diagnosis can lead to unnecessary en bloc resections with
severe postoperative sequelae [17]. MRI characteristics of the present
case were not characteristic for hydatid disease [16], yet could not
exclude it for sure.
The chosen modality of treatment in front of a cystic lesion of soft
tissue is the surgical removal without rupture and to avoid spilling of
the liquid contain within the surgical cavity.
Likewise for solid [2], even CS is cured by complete surgical
removal with sparing of the uninvolved fascicles. The decompression
of the nerve fascicles involved within the same epineurium with the
CS gives rise to gradual sensitive and motor recovery of the footdrop,
as it was observed in the presented case. Schwannoma should
be considered in the differential diagnosis of cystic lesions of the soft
tissues in extremities, especially in the presence of a neurological
deficit of a peripheral nerve.
References
- Paik KY, Choi SH, Heo JS, Choi DW. Solid tumors of the pancreas can put on a mask through cystic change. World J Surg Oncol. 2011;19(9):79.
- Wu JS, Hochman MG. Soft-tissue tumors and tumor like lesions: a systematic imaging approach. Radiology. 2009;253(2):297-316.
- Vena P, Fiorito S. A case of cystic neurinoma of the median popliteal nerve. Clin Ortop. 1974;25(4):270-3.
- Chen WC, Chang YL, Lee YC. A huge cystic cellular schwannoma of the intercostal nerve presenting with dyspnea. Ann Thorac Surg. 2009;87(4):1268-9.
- Akhaddar A, Ajja A, Albouzidi A, Elmostarchid B, Boucetta M. Cystic schwannoma of the cauda equina mimicking hemangioblastoma. Neurochirurgie. 2008;54(2):101-3.
- Rafai MA, El Otmani H, Rafai M, Bouhaajaj FZ, Largab A, Trafeh M, el al. Peroneal nerve schwannoma presenting with a peroneal palsy. Rev Neurol (Paris). 2006;162(8-9):866-8.
- Gueldich M, Hentati A, Chakroun A, Abid H, Kammoun S, M'saad S, et al. Giant cystic schwannoma of the middle mediastinum with cervical extension. Libyan J Med. 2015;10:27409.
- Chen F, Miyahara R, Matsunaga Y, Koyama T. Schwannoma of the brachial plexus presenting as an enlarging cystic mass: report of a case. Ann Thorac Cardiovasc Surg. 2008;14(5):311-3.
- Romner B, Nygaard O, Ingebrigtsen T, Anke IM, Trumpy JH. Large peripheral cystic schwannomas. Two case reports and a review. Scand J Plast Reconstr Surg Hand Surg. 1994;28(3):231-4.
- Karatas A, Is M, Yildirim U, Akyuz F, Gezen F. Thoracic Intradural Cystic Schwannoma: A Case Report. Turk Neurosurg. 2007;17(3):193-6.
- Alimehmeti R, Seferi A, Rroji A, Alimehmeti M. Saphenous neuropathy due to large hydatid cyst within long adductor muscle: case report and literature review. J Infect Dev Ctries. 2012;6(6):531-5.
- Jindal T, Mukherjee S, Kamal MR, Sharma RK, Ghosh N, Mandal SN, et al. Cystic schwannoma of the pelvis. Ann R Coll Surg Engl. 2013;95(1):e1-2
- Bermejo A, Díaz De Bustamante T, Martinez A, Carrera R, Zabía E, Manjón P. MR Imaging in the Evaluation of Cystic-appearing Soft-Tissue Masses of the Extremities. RadioGraphics. 2013;33:833–55.
- Yu GH, Sack MJ, Baloch Z, Gupta PK. Difficulties in the fine needle aspiration (FNA) diagnosis of schwannoma. Cytopathology. 1999;10:186–94.
- Kashyap S, Pushker N, Meel R, Sen S, Bajaj MS, Khuriajam N, et al. Orbital schwannoma with cystic degeneration. Clin Experiment Ophthalmol. 2009;37(3):293-8.
- Tatar IG, Yilmaz KB, Arikok A, Bayar B, Akinci M, Balas S, et al. Cystic schwannoma of the axillary region: imaging findings of a rare disease. Case report. Med Ultrason. 2015;17(1):126-8.
- Somayaji KSG, Rajeshwari A, Gangadhara KS. Schwannoma of the brachial plexus presenting as a cystic swelling. Indian J Otolaryngol Head Neck Surg. 2004;56(3):228-30.