Review Article
Surgical Approaches to the Internal Carotid Artery at the Extracranial Middle Skull Base
Desai M1, Rolls A1, Girish G2 and Baker DM1 *
1Department of Vascular Surgery, Royal Free London NHS Foundation Trust, London, UK
2Consultant General and Vascular Surgeon, West Hertfordshire Hospitals NHS Trust, Watford, UK
*Corresponding author: Daryll M. Baker, Consultant General and Vascular Surgeon, Royal Free Hospital, Pond Street, London NW3 2QG, UK
Published: 11 Jul 2017
Cite this article as: Desai M, Rolls A, Girish G, Baker DM.
Surgical Approaches to the Internal
Carotid Artery at the Extracranial Middle
Skull Base. Clin Surg. 2017; 2: 1551.
Abstract
The aims of this review are to critically review the surgical approaches to the internal carotid artery
(ICA) at the skull base as reported in the literature. We discuss the relevant anatomy and the
common indications and approaches to the internal carotid artery at the skull base are outlined.
We conclude that open surgical approaches to the ICA at the skull base are feasible with careful
anatomic dissection and can be performed with minimal morbidity in most cases. The optimum
approach continues to be debated.
Keywords: Surgical approaches; Carotid artery; Skull base
Introduction
Operative exposure of the high parapharyngeal and intrapetrous segments of the internal carotid artery (ICA) presents a challenge to the skull base surgeon. Historically, lesions in the portion of the artery above the level of a line drawn between the angle of the mandible and the tip of the mastoid process (Blaisdell line) had been considered inaccessible by standard surgical techniques [1]. Adequate exposure through a standard carotid incision is often not possible. The pathway must be carefully designed to protect the multiple critical neurovascular structures immediately adjacent to the ICA in this area [2]. Over the past decades, with advances in technologies, endovascular therapy has enhanced accessibility of the distal ICA and offers a minimally invasive alternative to open surgery. However, some lesions may not be amenable and long-term results are lacking. So open surgical approaches remain an important part of treatment strategies for lesions in this area. The aims of this review are to discuss the anatomy of the ICA and the skull base and critically review the surgical approaches to the ICA at the skull base reported in the literature.
Course and Relations
In considering the course and relations of the ICA, it may be divided into four portions: cervical,
petrous, cavernous, and cerebral (Figures 1,2 and 3). Through its course, it forms several bands and
gives off branches as shown in Figure 1.
Cervical
This portion of the internal carotid begins at the bifurcation of the common carotid, opposite the
upper border of the thyroid cartilage, and runs perpendicularly upward below the skull, where it has
an area of fibrous ring making mobilisation difficult (Figure 2). The relationship with nasopharynx,
vertebral artery, condyle of atlas and occipital condyle is shown in Figure 2. It passes in front of the
transverse processes of the upper three cervical vertebræ, to enter the carotid canal in the petrous
portion of the temporal bone. It is comparatively superficial at its commencement, where it is
contained in the carotid triangle, and lies behind and lateral to the external carotid, overlapped
by the Sternocleidomastoideus, and covered by the deep fascia, Platysma, and integument: it then
passes beneath the parotid gland, being crossed by the hypoglossal nerve, the Digastricus and
Stylohyoideus, and the occipital and posterior auricular arteries. Higher up, it is separated from
the external carotid by the Styloglossus and Stylopharyngeus, the tip of the styloid process and the
stylohyoid ligament, the glossopharyngeal nerve and the pharyngeal branch of the vagus. It is in
relation, behind, with the Longus capitis, the superior cervical ganglion of the sympathetic trunk,
and the superior laryngeal nerve; laterally, with the internal jugular vein and vagus nerve, the nerve
lying on a plane posterior to the artery; medially, with the pharynx, superior laryngeal nerve, and
ascending pharyngeal artery. At the base of the skull the glossopharyngeal, vagus, accessory, and
hypoglossal nerves lie between the artery and the internal jugular vein.
Petrous
The cervical ICA becomes the petrous segment of the artery as it
enters the petrous temporal bone at the base of the skull anterior to
the internal jugular vein and medial to the styloid process.
When the internal carotid artery enters the canal in the petrous
portion of the temporal bone, it first ascends a short distance, then
curves forward and medial ward, and again ascends as it leaves the
canal to enter the cavity of the skull between the lingula and petrosal
process of the sphenoid. It emerges through foramen lacerum and
passes vertically upwards. The artery is separated from the bony wall of
the carotid canal by a prolongation of dura mater, and is surrounded
by a number of small veins and by filaments of the carotid plexus,
derived from the ascending branch of the superior cervical ganglion
of the sympathetic trunk.
Cavernous
In this part of its course, the artery is situated between the layers
of the dura mater forming the cavernous sinus, but covered by the
lining membrane of the sinus. It at first ascends toward the posterior
clinoid process, then passes forward by the side of the body of the
sphenoid bone, and again curves upward on the medial side of the
anterior clinoid process, and perforates the dura mater forming the
roof of the cavernous sinus. This portion of the artery is surrounded
by filaments of the sympathetic nerve, and on its lateral side is the
abducent nerve.
Cerebral
Having perforated the dura mater on the medial side of the
anterior clinoid process, the ICA passes between the optic and
oculomotor nerves to the anterior perforated substance at the medial
extremity of the lateral cerebral fissure, where it gives off its terminal
or cerebral branches.
Branches
The cervical portion of the internal carotid gives off no branches.
Those from the other portions are as shown in Figures 1-3. The figures
also show relationship of the branches with important nerves and
other anatomical landmarks.
1. From the Petrous Portion
a. Caroticotympanic branch to middle ear
b. Artery of the Pterygoid Canal
2. From the Cavernous Portion
a. Cavernous
b. Inferior hypophyseal
c. Semilunar
d. Anterior Meningeal
e. Ophthalmic
3. From the Cerebral Portion
a. Anterior Cerebral
b. Middle Cerebral
c. Posterior Communicating
d. Superior hypophyseal
e. Anterior Choroidal
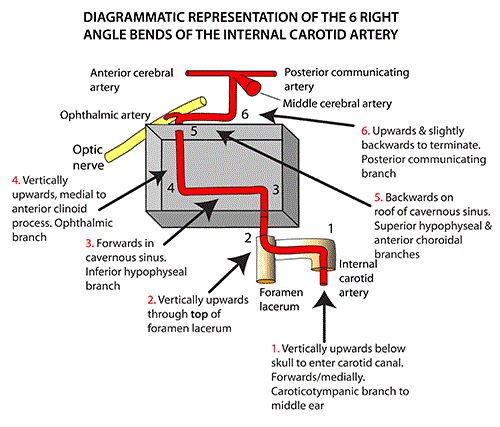
Figure 1
Figure 1
Internal Carotid Artery in skull (we thank Instantanatomy.
net for permission to use this image). This figure is a diagrammatic
representation of the 6 bends of the internal carotid artery as it courses
after entering the carotid canal. Branches coming off at each level are also
included.
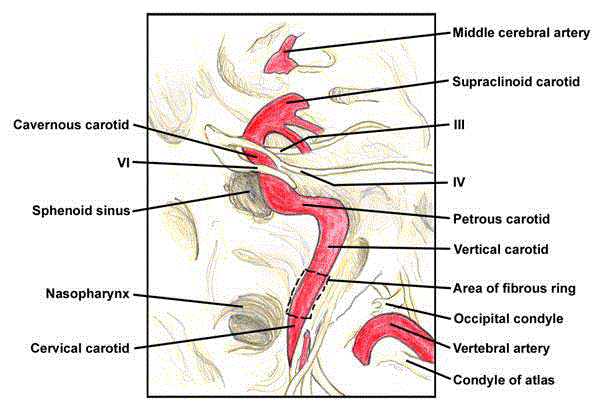
Figure 2
Figure 2
ICA segments (with permission from emedicine.medscape.
com). This figure shows the intracranial course of internal carotid artery.
Cervical, petrous, cavernous, and supraclinoid (cerebral) segments are
shown including its relations with cranial nerves III, IV and VI, nasopharynx,
sphenoid sinus and vertebral artery posteriorly as it merges from the
transverse process of C2 (axis), and sweeps laterally to pass through the
transverse foramen of C1 (atlas).
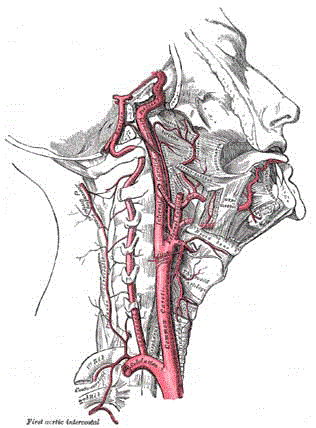
Figure3
Figure 3
Anatomy of the ICA (with permission from radiopaedia.org.
This diagram shows the origin of right common carotid artery from the
brachiocephalic artery, its cervical course, relations with trachea and thyroid
cartilage and division into the internal and external carotid arteries. The
cervical course of internal carotid artery is visible with its relationship with
the vertebral artery running posteriorly following its origin from the subclavianartery
in addition to the relative position of the hyoid bone and main
muscles of the face and the neck. More distally, the petrous and cavernous
segments of the internal carotid artery are visible with right-angled bends
along its courseas it enters the petrous temporal bone and proceeds towards
anterior clinoid process at the base of the skull.
Skull Base Anatomy
The skull base forms the floor of the cranial cavity and separates
the brain from other facial structures. The 5 bones that make up the
skull base are the ethmoid, sphenoid, occipital, paired frontal, and
paired parietal bones. The skull base can be subdivided into 3 regions:
the anterior, middle, and posterior cranial fossae.
Anterior skull base
The anterior limit of the anterior skull base is the posterior wall
of the frontal sinus. The anterior clinoid processes and the planum
sphenoidale, which forms the roof of the sphenoid sinus, mark the
posterior limit. The frontal bone forms the lateral boundaries. The
major structures in this area are the olfactory bulb and tract.
Middle skull base
The greater wing of the sphenoid helps form the anterior limit
of the middle skull base. The posterior limit is the clivus, which is
formed from the sphenoid and occipital bones. The greater wing
of the sphenoid forms the lateral limit as it extends laterally and
upward from the sphenoid body to meet the squamous portion of the
temporal bone and the anteroinferior portion of the parietal bone.
The greater wing of the sphenoid forms the anterior floor of the fossa.
The anterior aspect of the petrous temporal bone forms the posterior
floor of the middle cranial fossa.
Posterior skull base
The posterior skull base consists of primarily the occipital bone,
with contributions from the sphenoid and temporal bones. The
midbrain, the pons, the medulla, and the cerebral and cerebellar
hemispheres lie in the posterior fossa.
Indications
The common indications for surgical approach to the ICA at the
skull base are outlined below:
Difficult carotid endarterectomy
Management of carotid bifurcation atherosclerotic stenosis is a
cornerstone of stroke prevention. The standard approach for carotid
endarterectomy (CEA) provides excellent access to the cervical carotid
artery, but lesions that extend outside this zone can be difficult to treat
surgically. The high location of the carotid bifurcation at or above the
level of the C2 cervical vertebra and a very distal extension of internal
carotid atherosclerotic disease may challenge vascular surgeons
performing CEA by increasing technical difficulty and possibly the
incidence of cranial nerve injury. Division of the digastric muscle,
mandibular manipulations such as anterior translational movement
when the mandible moves forward with teeth, condyles and rami all
moving in the same direction and to the same degree or subluxation
can help in accessing lesions of the distal cervical ICA. In addition,
by division of tethering artery and vein to sternocleidomastoid, the
descending hypoglossal branch of ansa cervicalis or the occipital
artery can help in mobilisation of the hypoglossal nerve and exposure
of more distal internal carotid artery. Although rarely required, these
high carotid exposures may be associated with increased difficulty
in directly visualising the end point of the endarterectomy and with
increased incidence of cranial nerve injury, particularly cranial
nerve IX [3,4]. Nasotracheal intubation has also been suggested in
some early reports, but the recent evidence from a cadaveric study
suggests nasotracheal intubation does not improve access to a high
carotid artery bifurcation as compared with orotracheal intubation
[5]. Carotid artery stenting (CAS) is an alternative in these patients,
however anatomic factors that may complicate this process include
difficult access with aortoiliac tortuosity, a sharply angulated aortic
arch (type III), or a carotid lesion with more than two 90° bends
within a short distance of the target lesion [6]. Significant distal ICA
tortuosity may also complicate the placement and stabilisation of a
distal embolic protection device [7]. Pending more evidence from
ongoing clinical trials, CEA will remain the mainstay of treatment
and vascular surgeons will need to develop strategies to effectively
manage anatomically challenging lesions.
Carotid body paragangliomas
Carotid body tumours (CBTs) belong to the classification
of paragangliomas because they originate from paraganglia in
chromaffin-negative glomus cells derived from the embryonic
neural crest, functioning as part of the sympathetic nervous system.
These cells normally act as special chemoreceptors located along
blood vessels, particularly in the carotid bodies (at the bifurcation
of common carotid artery in the neck) and in the aortic bodies
(near the aortic arch) [8]. Most of these lesions are benign; however
some can show malignant behavior with few reports of histological
confirmation of malignant CBTs [9]. There are no clear histological
features for diagnosis of malignant carotid body paragangliomas to
differentiate them from benign tumours. Paragangliomas tend to
occur in sites where basement membrane penetration, the hallmark
of malignancy in many epithelial tumours, cannot be assessed.
Histologic features such as nuclear pleomorphism, necrosis, mitotic
rate, and local invasion may be seen in benign paragangliomas and
are not diagnostic of malignancy. According to 2004 World Health
Organisation criteria, the diagnosis is reserved for tumours with local,
regional and distant metastasis [10]. The treatment modalities for
CBTs are surgical excision and/or radiotherapy. Surgical removal is
the treatment of choice as it provides immediate and complete tumour
removal. CBT surgery remains a challenge for surgeons because of
tumour’s location in the vicinity of critical blood vessels and cranial
nerves. In addition to its location, additional difficulty is its high
vascularity, as its blood supply is the richest per gram of tissue of any
tumour [11]. CBTs are usually classified by the criteria described by
Shamblin et al.[12] (Table 1) which is used to assess invasiveness.
Complete resection of Shamblin class III CBTs is very challenging
and often requires temporary interruption of cerebral circulation for
vascular reconstruction with significant risk of permanent vascular
and neural defects [13].
The strategies utilised to aid in surgical resection include
preoperative embolisation and use of intraoperative shunting. The
routine use of preoperative embolisation is controversial because of
the potential neurologic complication associated with the accidental
reflux of particulate matter into the ophthalmic or cerebral circulation.
Some authors advocate its use for larger tumours as it may decrease
the tumour vascularity and subsequent intraoperative blood loss. The apparent benefit of embolisation should be weighed against the risk
of stroke.
An intraoperative shunt can also be used in the following
circumstances to aid in CBT resection and shorten surgical time [14]:
To avoid the injury of cranial nerves: when the large size of the
tumour and the narrow operative space make it difficult to excise the
tumour and easy to injure the cranial nerves, under the guidance of
the shunt, the direction of the ICA is more distinct, and cranial nerves
are more clear, which helps tumour dissection.
To decrease the size of the tumour: with the use of the shunt, the
blood supply to the tumour decreases, thereby decreasing the size of
the tumour.
Even with the use of these adjuncts, complete surgical resection
may not be possible through a transcervical approach alone and more
radical exposure is warranted.
Internal carotid artery aneurysms
Internal carotid artery aneurysms (ICAA) are rare. Surgical
ICAA repair accounts for less than 1% of all aneurysm repairs [15].
Aneurysms of the ICA developed at the base of the skull in the intratemporal
fossa are even rarer [16]. The aetiology of ICAA is multiple
including atherosclerosis, fibromuscular dysplasia, post-traumatic
and infectious lesions [17]. Prevention of thrombo-embolic
complications is the main indication for treatment; however surgical
approach to these lesions faces anatomical difficulties due to the
complexity of the region and the close relation between the ICA and
the cranial nerves, mainly the facial nerve [18]. Endovascular stentgrafting
is particularly attractive in this situation and has been used
but its role in still in its infancy with arterial dissection, embolism
during deployment, stent fracture, intimal hyperplasia, and longterm
occlusion as potential risks associated with it and uncertain
long-term results. The alternatives to surgical repair are ligation of the
ICA and ligation of the ICA combined with external carotid/internal
carotid bypass [19]. However, these options are less preferred due to
the high incidence of ischaemic complications and stroke [17,18]. As
a consequence, the direct surgical repair seems to be the best solution,
leaving the challenging problem of the approach to the ICA at the
base of the skull.
In addition to the above relatively common indications, ICA
blunt trauma causing pseudo-aneurysm, dissection or stenosis and
other tumours may mandate surgery at this level.
Table 1
Table 2
Approaches
Historical perspective
Several techniques have been proposed as outlined in Table 2.
However, they were mostly associated with more or less perturbation
of the facial nerve, temporal bone and mandible causing significant
functional morbidity. We reviewed the contemporary literature
to evaluate and critically review the approaches in current use as
discussed below.
Transcervical approaches
Adequate exposure can be occasionally achieved especially for
lesions located in the lower parapharyngeal space with adjuncts
including resection of the posterior belly of digastric muscle with
identification and preservation of facial nerve. This procedure can
also be combined with parotidectomy to gain access to tumours
and other lesions in the middle parapharyngeal space. This is
based on compartmentalisation of the parapharyngeal space as
described by Shahinian et al. [20]. The authors suggest a cervical
submandibular approach for tumours in the inferior parapharyngeal space (hypopharynx) extending inferiorly into the cervical area. For
tumours in the midparapharyngeal space (mesopharynx) a parotidcervical
approach can be used with anterior and inferior retraction
of the mandible. For smaller tumours of the superior parapharyngeal
space (epipharynx) extending to the skull base an infratemporal
fossa approach with a preauricular incision and a plane of dissection
anterior to the middle ear, petrous horizontal internal carotid artery,
and the eustachian tube is recommended sparing the middle ear,
temporomandibular joint, and the cranial nerve V3. For massive
tumours of the entire parapharyngeal spaces that extend to or through
the skull base superiorly or encase the petrous portion of the internal
carotid artery an infratemporal fossa type approach is required with
blind closure of external auditory canal, a mandatory conductive
hearing loss, removal of the temporomandibular articular disc, and
sacrifice of cranial nerve V3.
Lateral infratemporal approaches
The transcervical incision can be extended if more exposure
is needed with a preauricular incision laterally into the temporal
area. The temporal branch of facial nerve should be preserved. The
temporomandibular joint (TMJ) is freed, intact from the temporal
fossa and distracted anterior to the articular eminence with resection
of the mastoid tip often needed. The Eustachian tube is identified and
removed and the middle meningeal artery is usually ligated to allow
greater access [2]. Bone is removed over the proximal ascending and
horizontal portions of the petrous carotid until sufficient exposure is
gained. At the conclusion, TMJ is replaced in the glenoid fossa and
posterior joint capsule is sutured back in its place. A similar approach,
with some modifications has also been described by Malikov et al.
[18].
• Prasad et al. [21] have recently described three lateral skull
base infratemporal fossa approaches for upper parapharyngeal space
tumours.Type A with permanent anterior transposition of the facial
nerve to provide optimum exposure of the jugular foramen and to
allow control over the distal parapharyngeal ICA up to the vertical
petrous portion.
• Type B for tumours with antero-medial extension with
respect to ICA. This approach provides access to the vertical and
horizontal portions of the petrous ICA.
• Type D for tumours with antero-lateral extension with
respect to ICA. This approach consisted of a preauricular incision
with dissection anterior to the horizontal petrous ICA and the
Eustachian tube.
Midline mandibulotomy
The ICA runs in the parapharyngeal space and can therefore
be approached from medial direction using combined midline
mandibulotomy and an extended cervical incision. Vikatmaa et al.
[22] reported their experience in five cases. Lip split and intraoral
mucosal incision are performed with medial mobilisation of the
tongue and lateral rotation of the mandible. Injury to the marginal
mandibular branch of the facial nerve is avoided by identifying it
1 cm anteriorly and inferiorly to the mandibular angle or ligating
and elevating the facial vessels to protect it [23]. The tympanic bone
and the carotid canal can be reached and distal ICA control can be
obtained. The bony structures of the skull base do not need routine
resection.
A new exposure technique with application of double mandibular
osteotomy has been described by Ktenidis et al. [24] in the treatment
of giant ICAA. An osteotomy was made at the base of the condylar
process to increase mandibular mobility and to allow rotation of
the body and ramus of the mandible when a second mandibular
osteotomy was carried out anterior to the mental foramen. The
coronoidectomy improved the exposure of the parapharyngeal space,
increased mandibular segment mobility, and prevented postoperative
trismus.
Other approaches
Transnasal endoscopic approaches to the skull base are an
alternative to more traditional open approaches in selected cases.
What is crucial in these approaches is the anatomy of the ICA, which
takes a complicated, tortuous course through this area [25]. The
lateral pterygoid plate and posterior border of mandibular ramus are
important anatomic landmarks during the endoscopic approach to
the infratemporal fossa. Endoscopic transvestibular approach has
also been described by Chan et al. although the risk of this approach
is the tunnel-like exposure, surrounded by vital structures [26].
Hybrid approach with combined open and endovascular treatment
for saccular ICA aneurysm with redundant ICA loop has also been
reported [27]. Preoperative endovascular stenting has been proposed
in patients having high risk of ICA injury. The main purpose is to
reinforce the arterial wall allowing a safe subadventitial dissection
during tumour removal. Anterior exposure of the infratemporal
fossa and distal control of the carotid artery at the level of the carotid
canal has been recently described through a transcervical approach,
performing double mandibular osteotomies with superior reflection
of the middle mandibular section [28]. In addition, exposure and
mobilisation of the intrapetrous carotid artery using a retrosigmoid
approach is feasible and could represent a viable option for the
possibility of total resection of selected skull base tumours, even when
involvement of the carotid canal is present [29].
Discussion
Exposure of the ICA near the base of the skull is complex and
associated with substantial morbidity. Recent progress in endovascular
technology has raised the possibility of treating some of these lesions
without radical surgical exposure but there have been only sporadic
reports and further development and long-term results are awaited.
Surgery continues to be the mainstay especially in the management
of carotid body and other parapharyngeal space tumours. The main
aims are selecting a technique with adequate intraoperative visibility
for radical resection and minimal functional and cosmetic sequelae.
Several different historic techniques have been reported in the
literature in the last four decades but most of them are associated with
significant and unacceptable morbidity. More recently, acceptable
results have been reported with lateral infratemporal approaches and
midline mandibulotomy but the optimum technique has not been
defined. Proponents of the lateral infratemporal approaches suggest
there is low immediate morbidity associated with this procedure with
excellent long-term results [18]. Although, the petrous ICA can also
be exposed by the posterior approach, this is less preferred to prevent
profound conductive hearing loss associated with this technique
[30]. With the lateral approach, drilling free both the vertical and
horizontal portions of the petrous carotid artery allows the removal
of bone around 270° of the artery. This allows excellent mobility of
the artery for bypass [31]. The morbidity from this type of resection
results primarily from cranial nerve dysfunction, Eustachian tube dysfunction with possible need for myringotomy and tube placement
and loss of chorda tympany nerve, however permanent cranial nerve
injuries are uncommon [30]. Patients should also be advised of
possible TMJ discomfort and taste disturbances.
Other group of surgeons prefers the midline and paramidline
mandibulotomies with lateral luxation of the ipsilateral mandible,
which gives an excellent exposure to the oral cavity and oropharynx
and has been widely used for resection of tumours. With these
techniques, they claim the skull base can be widely exposed and the
distal ICA runs lateral to the nasopharyngeal cavity superficially
under the mucous membrane [22]. Although the sterility is obviously
compromised, the risk of major infection problems is low. They
suggest the lateral approach can lead to injury to the glossopharyngeal
and vagus nerves lying just medial to the styloid process. In a
cadaveric anatomic study by Beretta et al, the last 10 mm of skull
base remained unexposed and the operative field remained narrow
(range 5°-49°) making a distal anastomosis impossible or hazardous
to perform [32]. However, the argument against mandibulotomy is
facial scarring from lip splitting mandibulotomies, and malocclusion,
loss of mental nerve, paralysis of the mandibular branch of facial
nerve, tracheostomy in 6% to 27% of the cases and delayed wound
healing with other mandibulotomies [33-35]. Even the more recent
single subcutaneous midline mandibulotomy has the drawback of
tooth morbidity and in some cases the application of tension band or
segmental arch bar that is required to prevent rotation of the alveolus
[21,36,37].
Conclusion
Open surgical approaches to the ICA at the skull base are feasible with careful anatomic dissection and can be performed with minimal morbidity in most cases. The optimum approach continues to be debated and until long-term results are available, skull base surgeons will continue with their preferential technique. There are insufficient data to draw any definite conclusions on which method to choose in the management of high ICA lesions. The role of endovascular therapy will continue to expand but further research is warranted before their widespread adaptation.
References
- BlaisdellWF, Clauss RH, Galbraith JG, Imparato AM, Wylie EJ. Joint study of extracranialarterial occlusion. IV. A review of surgical considerations. JAMA.1969;209(12):1889-95.
- LangermanA, Naslund TC, Netterville JL. Skull base approach to carotid artery lesions:technique, indications and outcomes. J Neurol Surg B Skull Base.2012;73(3):163-7.
- JaspersGW, Witjes MJ, van den Dungen JJ, Reintsema H, Zeebregts CJ. Mandibularsubluxation for distal internal carotid artery exposure in edentulous patients.J Vasc Surg. 2009;50(6):1519-22.
- Simonian GT, Pappas PJ, Padberg FT Jr, Samit A, Silva MB Jr,Jamil Z, et al. Mandibular subluxation for distal internal carotid exposure:technical considerations. J Vasc Surg. 1999;30(6):1116-20.
- ForemanPM, Harrigan MR, Griessenauer CJ, Loukas M, Tubbs RS. Access to the carotidartery bifurcation: Cadaveric study with application to nasotracheal intubationas a technique to improve access to a high carotid artery bifurcation. Br J Neurosurg.2015;29(6):865-7.
- SetacciC, Chisci E, Setacci F, Iacoponi F, de Donato G, Rossi A. Siena carotid arterystenting score: a risk modelling study for individual patients. Stroke; ajournal of cerebral circulation. 2010;41(6):1259-65.
- RicottaJJ, Aburahma A, Ascher E, Eskandari M, Faries P, Lal BK, et al. Updated Societyfor Vascular Surgery guidelines for management of extracranial carotid disease.J Vasc Surg. 2011;54(3):e1-31.
- AmatoB, Bianco T, Compagna R, Siano M, Esposito G, Buffone G, et al. Surgicalresection of carotid body paragangliomas: 10 years of experience. Am J Surg.2014;207(2):293-8.
- NishijimaH, Asakage T, Sugasawa M. Malignant carotid body tumor with systemic metastases.Ann Otol Rhinol Laryngol. 2011;120(6):381-5.
- Pathology and Genetics of Tumours of the Endocrine Organs.WHO Classification of Tumours [press release]. Lyon France: IARC Press. 2004.
- AT. Carotid Body Tumours: A Review. InternationalJournal of Clinical Medicine. 2015;6:119-31.
- ShamblinWR, ReMine WH, Sheps SG, Harrison EG, Jr. Carotid body tumor (chemodectoma).Clinicopathologic analysis of ninety cases. Am J Surgery. 1971;122(6):732-9.
- AnandVK, Alemar GO, Sanders TS. Management of the internal carotid artery duringcarotid body tumor surgery. Laryngoscope. 1995;105(3 Pt 1):231-5.
- ZengG, Zhao J, Ma Y, Huang B. Resection of carotid body tumors and the additionalchoice of intraoperative shunt in complicated tumors. Ann Vasc Surg. 2012;26(4):511-5.
- McCollumCH, Wheeler WG, Noon GP, DeBakey ME. Aneurysms of the extracranial carotidartery. Twenty-one years' experience. Am J Surg. 1979;137(2):196-200.
- RossetE, Albertini JN, Magnan PE, Ede B, Thomassin JM, Branchereau A. Surgicaltreatment of extracranial internal carotid artery aneurysms. J Vasc Surg.2000;31(4):713-23.
- McCannRL. Basic data related to peripheral artery aneurysms. Ann Vasc Surg.1990;4(4):411-4.
- MalikovS, Thomassin JM, Magnan PE, Keshelava G, Bartoli M, Branchereau A. Opensurgical reconstruction of the internal carotid artery aneurysm at the base ofthe skull. J Vasc Surg. 2010;51(2):323-9.
- TheEC/IC Bypass Study Group. Failure of extracranial-intracranial arterial bypassto reduce the risk of ischemic stroke. Results of an international randomizedtrial. N Engl J Med. 1985;313(19):1191-200.
- ShahinianH, Dornier C, Fisch U. Parapharyngeal space tumors: the infratemporal fossaapproach. Skull Base Surg. 1995;5(2):73-81.
- PrasadSC, Piccirillo E, Chovanec M, La Melia C, De Donato G, Sanna M. Lateral skullbase approaches in the management of benign parapharyngeal space tumors. AurisNasus Larynx. 2015;42(3):189-98.
- VikatmaaP, Mäkitie AA, Railo M, Törnwall J, Albäck A, Lepäntalo M. Midlinemandibulotomy and interposition grafting for lesions involving the internalcarotid artery below the skull base. J Vasc Surg. 2009;49(1):86-92.
- Cassoni A, Terenzi V, Della Monaca M, Bartoli D, Battisti A,Rajabtork Zadeh O, et al. Parapharyngeal space benign tumours: our experience.J craniomaxillofac surg. 2014;42(2):101-5.
- KtenidisK, Lioupis A, Megalopoulos A, Antoniadis K, Kiskinis D. New exposure techniquefor management of giant internal carotid artery aneurysm. J Vasc Surg.2011;54(2):522-5.
- HoB, Jang DW, Van Rompaey J, Figueroa R, Brown JJ, Carrau RL, et al. Landmarksfor endoscopic approach to the parapharyngeal internal carotid artery: aradiographic and cadaveric study. Laryngoscope. 2014;124(9):1995-2001.
- ChanJY, Li RJ, Lim M, Hinojosa AQ, Boahene KD. Endoscopic transvestibularparamandibular exploration of the infratemporal fossa and parapharyngeal space:a minimally invasive approach to the middle cranial base. Laryngoscope.2011;121(10):2075-80.
- Trinidad-HernándezM, Introcaso JH, White JV. Combined open and endovascular treatment of asaccular aneurysm and redundant loop of the internal carotid artery. J VascSurg. 2006;44(3):642-6.
- DavisL, Zeitouni A, Makhoul N, Steinmetz OK. Surgical Exposure to Control the DistalInternal Carotid Artery at the Base of the Skull during Carotid AneurysmRepair. Ann Vasc Surg. 2016;34:268 e5-8.
- ScerratiA, Ercan S, Wu P, Zhang J, Ammirati M. Intrapetrous Internal Carotid Artery:Evaluation of Exposure, Mobilization and Surgical Maneuvers Feasibility from aRetrosigmoid Approach in a Cadaveric Model. World Neurosurg. 2016;91:443-50.
- Eliason JL, Netterville JL, Guzman RJ, Passman MA, NaslundTC. Skull base resection with cervical-to-petrous carotid artery bypass tofacilitate repair of distal internal carotid artery lesions. Cardiovascularsurgery. 2002;10(1):31-7.
- DewLA, Shelton C, Harnsberger HR, Thompson BG Jr. Surgical exposure of the petrousinternal carotid artery: practical application for skull base surgery.Laryngoscope. 1997;107(7):967-76.
- BerettaF, Hemida SA, Andaluz N, Zuccarello M, Keller JT. Exposure of the cervicalinternal carotid artery: surgical steps to the cranial base and morphometricstudy. Neurosurgery. 2006;59(1 Suppl 1):ONS25-34; discussion ONS25-34.
- CarrauRL, Myers EN, Johnson JT. Management of tumors arising in the parapharyngealspace. Laryngoscope. 1990;100(6):583-9.
- DimitrijevicMV, Jesic SD, Mikic AA, Arsovic NA, Tomanovic NR. Parapharyngeal space tumors:61 case reviews. Int J Oral Maxillofac Surg. 2010;39(10):983-9.
- MaloneJP, Agrawal A, Schuller DE. Safety and efficacy of transcervical resection ofparapharyngeal space neoplasms. Ann Otol Rhinol Laryngol. 2001;110(12):1093-8.
- JungehuelsingM, Guntinas-Lichius O, Klussmann JP, Eckel HE, Stennert E. Modifications of themidline mandibulotomy for access to the parapharyngeal space. Laryngoscope.2010;120(8):1557-62.
- TengMS, Genden EM, Buchbinder D, Urken ML. Subcutaneous mandibulotomy: a newsurgical access for large tumors of the parapharyngeal space. Laryngoscope.2003;113(11):1893-7.
- FischUP, Oldring DJ, Senning A. Surgical therapy of internal carotid artery lesionsof the skull base and temporal bone. Otolaryngol Head Neck Surg (1979).1980;88(5):548-54.
- WelshP, Pradier R, Repetto R. Fibromuscular dysplasia of the distal cervicalinternal carotid artery. J Cardiovasc Surg (Torino). 1981;22(4):321-6.
- PurdueGF, Pellegrini RV, Arena S. Aneurysms of the high internal carotid artery: anew approach. Surgery. 1981;89(2):268-70.
- GlassockME 3rd, Smith PG, Bond AG, Whitaker SR, Bartels LJ. Management ofaneurysms of the petrous portion of the internal carotid artery by resectionand primary anastomosis. Laryngoscope. 1983;93(11 Pt 1):1445-53.
- PellegriniRV, Manzetti GW, DiMarco RF, Bekoe S, Arena SA, Marrangoni AG. The directsurgical management of lesions of the high internal carotid artery. JCardiovasc Surg (Torino). 1984;25(1):29-35.
- SandmannW, Hennerici M, Aulich A, Kniemeyer H, Kremer KW. Progress in carotid arterysurgery at the base of the skull. J Vasc Surg. 1984;1(6):734-43.
- FisherDF Jr, Clagett GP, Parker JI, Fry RE, Poor MR, Finn RA, et al. Mandibularsubluxation for high carotid exposure. J Vasc Surg. 1984;1(6):727-33.
- LarsenPE, Smead WL. Vertical ramus osteotomy for improved exposure of the distalinternal carotid artery: a new technique. J Vasc Surgery. 1992;15(1):226-31.
- AlimiYS, Di Mauro P, Fiacre E, Magnan J, Juhan C. Blunt injury to the internalcarotid artery at the base of the skull: six cases of venous graft restoration.J Vasc Surg. 1996;24(2):249-57.