Commentaries and Perspectives
Degenerative Aneurysms of Chronic Type B Dissection Scan be Repaired with Branched Devices Either Using the True, False or Both Lumens of the Dissection
Rodrigues EO, Cunha RC, Ferreira DM, Bicalho GC and Ferreira MV*
Department of Vascular and Endovascular Surgery, SITE Endovascular, Rio de Janeiro, Brazil
*Corresponding author: Marcelo Ferreira, Department of Vascular and Endovascular Surgery, SITE Endovascular, Rio de Janeiro, Brazil
Published: 13 Jun, 2017
Cite this article as: Rodrigues EO, Cunha RC, Ferreira
DM, Bicalho GC, Ferreira MV.
Degenerative Aneurysms of Chronic
Type B Dissection Scan be Repaired
with Branched Devices Either Using
the True, False or Both Lumens of the
Dissection. Clin Surg. 2017; 2: 1511.
Commentary
Aortic dissection is a very challenging and dynamic pathology that may present itself in many forms. The INSTEAD trial has demonstrated that over a period of five years, treating uncomplicated
type B dissections with TEVAR (thoracic endovascular repair) has demonstrated better outcomes
than best medical treatment. Landmark analysis suggested a benefit of TEVAR for all end points
between 2 and 5 y; for example, for all-cause mortality (0% vs. 16.9%; P=0.0003), aorta-specific
mortality (0% vs. 16.9%; P=0.0005), and for progression (4.1% vs. 28.1%; P=0.004) [1].
The great discussion is whether sealing the proximal tear is sufficient to promote false lumen
thrombosis.
Clinical evidence demonstrated that aneurysm formation in chronic dissections is reported to
be present in 35% of the patients with 36% mortality over a period of three years [2].
Over the past years the use of fenestrated and branched stent grafts (F/BEVAR) to exclude the
false lumen completely has been proposed with promising early and mid-term results as the only
way to avoid late complications [3].
In this article we will demonstrate that not only should we correct the aorta to its full extent, but
also that the intentional placement of the endograft into the false lumen is feasible and sometimes
necessary in specific cases as the best alternative to promote adequate blood supply to the visceral
segment and exclude the dissected aorta.
Since 2012, our group has been routinely using either custom made device (CMD) or over the
shelf (OTS) devices to treat late complications of aortic dissection, especially when visceral arteries
are involved. The follow up of these patients have demonstrated complete false lumen thrombosis
and initially better patency of the visceral branches when compared to aortic aneurysms.
In our experience, the chronic septum between the false lumen and the true lumen, that is
believed to be thick and non complacent, does not seem to be an issue. The narrow true, which
is seen as an issue in chronic dissections, has neither been a problem to navigate the device or to
catheterize the visceral vessels.
The main strategy is to redirect the aortic blood in flow towards target vessels. Many times the
visceral vessels are irrigated by both lumens.
The main consideration that should be done is where to land the branched device to allow
adequate targets vessels catheterization. This can be done either by using the true lumen, the false
lumen or both lumens, as long as the target vessels can be connected to the branches of the device.
The decision to use one or the other relies upon convenience of the surgeon and the presence
of the re-entrances between the true lumen and the false lumen, and upon the diameter of the true
lumen observed in the angiotomography.
One of the main concerns when treating the aorta to its full extent is the spinal cord ischemia.
Ideally we propose staged interventions to allow adequate collateralization. The intra operative
strategy to prevent spinal cord injury is to maintain spinal fluid pressure less than12 mmHg and the
mean arterial pressure never below 80 mmHg.
As described by Dr. Griepp’s work [4] adequate internal iliac and vertebral blood supply should
be present to supply the spinal cord blood flow. Whenever necessary subclavian revascularization is advised before deploying the thoracic graft. All internal iliac
arteries should be revascularized with branch devices given that they
participate in medular perfusion.
When treating these aortic dissection cases, access is also a caveat
since most of these patients have been previously submitted to arch
corrections with bypasses to the supra aortic trunks. In Figure 1 we
see a case of a patient that presented onset of symptoms in 2015
with acute chest pain and was taken to the hospital and submitted
to emergency replacement of the arch with a dacron graft bypass to
the brachiocephalic trunk and to the left carotid artery, associated to
anantegrade deployment of a aortic endograft into the descending
aorta.
One year later he developed a chronic dissection with aneurysmal
degeneration of the entire descending and thoracoabdominal aorta,
with all the visceral vessels except the left renal revascularized from
the false lumen. We can observe in the tomography that three main
fenestrations connected the false lumen and the true lumen. In some
cases as demonstrated in Figure 2, most of the visceral vessels were
located within the false lumen except for the left renal. The problem
with in this particular case was gaining access due to the previous
dacron graft, as demonstrated in Figure 3.
Several intraoperative maneuvers had to be used in order to
overcome anatomical challenges:
1- In order to use the T branch, it was necessary to
accommodate a thoracic 36-30 mm tapered device to fit the OTS
T-branch which has a 34 mm proximal diameter.
2- Access to the false lumen from femoral access to deploy the
main graft in the false lumen: It was necessary to use a very small
fenestration located below the renal sin order to migrate the device
from the true lumen into the false lumen.
3- Previous arch surgery: Visceral branch catheterization
from the subclavian into the ascending aorta was very tortuous. It
was necessary to use the Tip Driven by the sheath maneuver.
4- Left renal catheterization from a small very narrow
communication between the two lumens located in the topography
of the celiac trunk. It was necessary to use the Snare ride technique to
adequately catheterize the vessel.
T-branch considerations/Possible modifications
The T-branch is an off the shelf (OTS) device developed to attend
most of the patient’s anatomy. It has four caudally oriented branches
positioned according to pre-determined clock positioned CT studies.
The celiac trunk branch is positioned at 1:00 o’clock, the superior
mesenteric artery 12:00 o’clock, the left renal at 3:00 o’clock and right
renal at 9:00 o’clock.
According to Torselo’s publication in the Journal of Endovascular
Therapy in 2013 the T-branch was able to attend 49%% of the patients
anatomies. In his own conclusion he wrote that another 14% of the
patients could have been suitable for the graft if additional maneuvers
had been performed, thus reaching a 63% suitability [4].
In our experience, after adapting and modifying the T-branch,
our applicability rates reach 80% of the cases or even more.
Even though the graft originally measures 202 cm, if necessary,
both proximal and distal stents can be withdrawn thus diminishing
proximal aortic coverage and decreasing the risk of spinal cord
ischemia.
In order to facilitate catheterization of the target vessels,
staged opening of the T-branch is very helpful. Ideally both the
celiac trunk (CT) and superior mesenteric artery (SMA) should be
selectively catheterized with a guide wire before placing the stent. Pre
catheterization of the CT avoids covering its origin when the SMA
Stent is deployed.
Another maneuver that becomes very helpful is to selectively use
the 4 branches of the T-branch according to each patient’s anatomy.
As demonstrated in Figure 4, the celiac branch was used to catheterize
the left renal and the left renal branch was used to catheterize the celiac
trunk. Although there was a switch in the original configuration the
final result demonstrated excellent blood flow in the visceral arteries.
Another important consideration is that any of the four branches
can be intentionally occluded with plugs or modified stent grafts with
coils. This can be done either during the procedure, or during a second
intervention to allow blood flow into the aneurysm sac functioning as
a medullary perfusion branch.
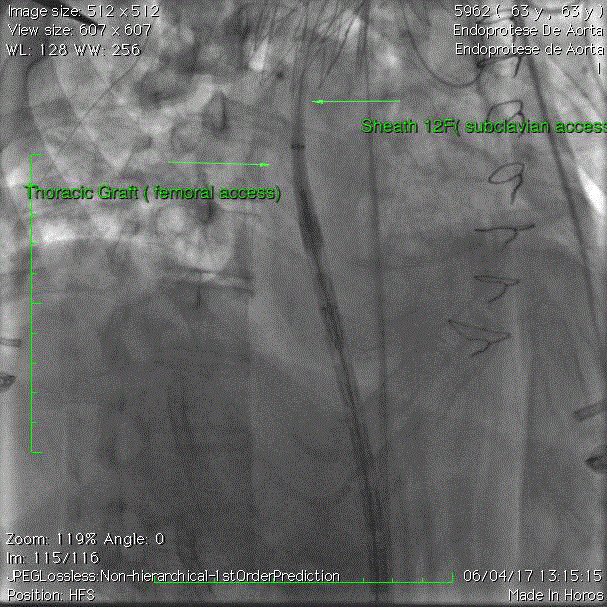
Tip driven by the sheath
To facilitate the migration of the branched endograft through
tortuous vessels and previous devices the trick is to use the maneuver
Driven by the Sheath. It is necessary to use a through and through
wire using the femoral and subclavian access. It is also important to
place a 12F sheath from the subclavian access as low as possible to
position the tip and insert the nose of the device. Once inside the tip
of the upper sheath, the nose from the graft remains attached to the
device, it moves upward as a unique piece.
The device migrates forward, guided by the tip of the upper access
sheath avoiding any contact with the aortic wall or any previous
device.
It is necessary to perform a uniform coordinated movement in
such a way that the surgeon advances the graft from below while the
assistant retrieves the sheath from above as demonstrated in Figure 5.
Snare ride technique
The snare ride originally created by our group in 2016,
published in the JEVT( epub ahead from print 2017 DOI:
10.1177/1526602817709465), is a technique that could be used as
a bailout technique whenever the target vessel catheterization is
difficult, like upward or short renal arteries without enough space to
be supported by a single wire. Figure 6 illustrates the technique.
In the example below, the left renal was very difficult to catheterize
from above and through the communication because of the distance
from the branch and also because it was short with an upward
orientation. Using the femoral access within the true lumen, the left
renal was catheterized with a cobra 2 catheter over a roadrunner wire
and then, once the vessel was catheterized, the catheter was exchanged
for an Indy Snare (It is important to note, that to our knowledge,
currently the Indy snare is the only snare that works over the wire).
From the subclavian access the communication was catheterized
and expanded with regular balloon angioplasty. The roadrunner guide
wire was exchanged for a Amplatz extra stiff wire. The Amplatz wire
was snared by using the IndySnare (Cook Bloomington USA) and
pushed inside the left renal artery. Once inside the artery we opened
the connection Stent, in this case a long Fluency (Bard) and then the
Snare was carried out. The snare besides helping the catheterization
also functioned as a anchor to stabilize stent deployment as
demonstrated in Figure 6.
Perspectives
Originally only type A and complicated type B dissections
undergone surgical repair. Nowadays, with increasing understanding
early repair have been indicated.
Clinical evidence has demonstrated that early repair seems to
have greater impact on future aortic remodelation, false lumen
thrombosis and lower aneurysm degeneration.
Restoring most, if not all, flow through the true lumen has been a
goal of surgical and endovascular treatment.
According to Dr. Chiesa [6], the most common situation
for TEVAR failure requiring conversion to open surgery is false
lumen enlargement leading to aneurysmal degeneration and
subsequent rupture. This is mainly due to false lumen reperfusion
and repressurization, caused by unsuccessful exclusion of the
proximal entry tear (i.e Type I-A endoleak or late migration) or distal
reperfusion from abdominal re-entry tears (even after successful
coverage of the proximal entry tear.
Size mismatch between the distal end of the stent graft and the
remarkably small diameter of a compressed distal true lumen (TL)
may contribute to relatively excessive over sizing that contributes
to distal endoleak or re-dissections, with stent induced distal redissection
(SIDR) associated with mortality of approximately 25%
[7].
Despite the different techniques to promote true lumen
expansion, due to the chronicity of the dissection, aortic remodeling
does not occur in all cases. Due to this physical phenomenon a shift
in paradigm has been proposed and many aortic centers drew their
attention toward the residual pressure in the false lumen.
In the past when the primary entry tear was located mainly in the
thoracic aorta, besides the correction with a TEVAR, many centers
have advocated different techniques to obliterate the false lumen to
avoid late retrograde filling.
Mosop and coworker first published a series of patients treated
with proximal TEVAR combined with the use of distal bare metal
stents. This technique which became known as the PETITCOAT
technique [8].
Another proposed technique done to avoid retrograde filling
was proposed by Hofferberth and colleagues [9]. They advocated
the aggressive expansion and disruption of the intimal membrane
to equalize pressures between true and false lumen. This technique
which was first described in acute expansions coined the acronym
STABILISE which stands for the stent assisted balloon induced
intimal disruption and relamination in aortic dissection repair.
The first techniques to induce false lumen thrombosis was coil
embolization. Due to the effort and cost needed to treat large patent
false lumens with individual embolic coils, many centers used
Amplatzer plugs. Due to elevated costs and low effectiveness, both of
techniques were soon abandoned.
Kolbel and colleagues proposed the Candy Plug technique that
was originally done by a modified thoracic stent graft to plug the false
lumen. Later on, the Knickerbocker technique was developed and
based on the dilation of the middle part of a large diameter stent graft
that is placed in the true lumen to rupture the dissection membrane.
A short bulbous segment of the stent graft is forcefully dilated using a
compliant balloon to rupture the dissection membrane [10].
Interestingly Dr. Mastracci compiled a chapter review published
in 2016 addressing all these techniques used for false lumen
embolization. Her conclusion after reviewing literature was that
the role of false lumen embolization was not clear and had yet to be
defined as a routine protocol for aortic dissections [11].
In 2013 Dr. Greenberg published a ten year review that included
30 patients treated with fenestrated endografts. In order to minimize
the risk of paraplegia and to allow for aortic remodeling, staged
procedures separated by two months interval were done. His group
observed that, aside from the proximal sealing and fixation zone
there exist three regions of potential failures: the intergraft joint the
interface between a branch and the distal visceral or brachiocephalic
artery and the distal sealing zone [12].
Despite all the difficulties they obtained good results and
concluded that FEVAR was feasible and could be used to treat these
patients.
The following year, Dr. Verho even demonstrated his initial
experience and outcomes of fenestrated/branched endografts in
chronic post dissection thoracoabdominal aneurysms. A total of 31
patients (25 male, mean age 65 11.4 years) were treated. Technical
success was 93.5% and 30-day mortality 9.6% [3].
Management of the aortic dissection can, however be challenging
when several important brachiocephalic or visceral arteries originate
from different lumens. Under such circumstances thrombosis of
the false lumen may actually disrupt blood flow to vital organs and
structures and result in devastating complications such as paraplegia,
cerebrovascular lesions, bowel ischemia and renal failure [13].
At all instances access from the false lumen to the proximal
landing zone in the true lumen is essential. The rationale for using
the false lumen as the route for placement of an endovascular graft
in an aortic dissection is based on many factors. The true lumen is
smaller in almost all aortic dissections and particularly in chronic
stages can be very restrictive. The false lumen will most often allow
full extension of the graft [12].
Alternatively, the false lumen intentional placement (FLIP)
technique can be used, as demonstrated throughout the chapter [14].
Dr. Watanabe et al. [15] performed repair of a dissecting
thoracoabdominal aneurysm with a compressed true lumen by
first performing visceral debranching, followed by placement of a
bifurcated graft in the false lumen.
Recently published in the annals of vascular surgery, Dr.
Kaumann and colleagues published two case reports where the
thoracic endograft was intentionally plotted within the false lumen
to correct a chronic aortic dissection with satisfactory outcomes [16].
Before performing aortic dissection endovascular corrections,
careful planning is fundamental. It is necessary to decide before hand
which lumen to look for and was to land your main graft in order to
promote adequate visceral perfusion.
Our group have been performing endovascular repair of
chronic type B dissections with branched devices since 2012. Recent
experience has demonstrated that the OTS T-branch attends most
patients’ anatomies. Even in patients with narrow true lumens we
have implemented the use of OTS devices with good results.
Whenever the patient has a unfavorable anatomy we advocate all
the intra operative graft modifications previously mentioned. Even in
patients with cephalic oriented branches are suitable for the T-branch.
As previously described the snare ride technique can be used as
bail out for cephalic oriented branches as well as those with difficult
catheterization. The main advantage of this maneuver is that the Indy
snare, allows you to gain extra support to catheterize vessels. It is less
morbid than retroperitoneal retrograde catheterization, and is less
costly.
Another advantage is that by using the snare ride, you can connect
the target vessels through previous fenestrations and is not necessary
to further fenestrate the aorta with perforating devices.
We strongly believe that it’s very important to stop all the blood
flow to the false lumen to prevent the pressurization and aortic
degeneration of it.
In conclusion, the Branched devices either CMD or OTS can
be used to repair Degenerative Thoracoabdominal aneurysms after
Chronic Dissections. In situations where most visceral vessels are
located within the false lumen, and the true lumen is too narrow, the
graft can be deployed within the false lumen thus allowing adequate
visceral perfusion, eventually both lumens can be used depending on
the visceral vessels anatomy after the dissection as we demonstrated
in the illustrative case here reported.
The important message is: to avoid blood flow in the false lumen
outside the device implanted and exclude all the possible reentries
that may result in late aortic degeneration.
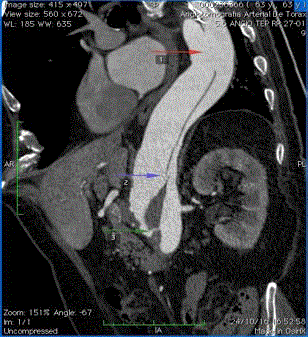
Figure 1
Figure 1
3D MPR Tomography reconstruction demonstrating main
fenestrations between the true lumen and the false lumen (the colored
arrows demonstrate the fenestrations).
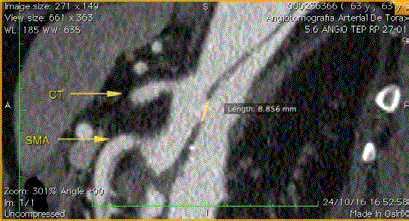
Figure 2
Figure 2
3D MPR Tomography reconstruction demonstrating the main
visceral vessels located in the false lumen.
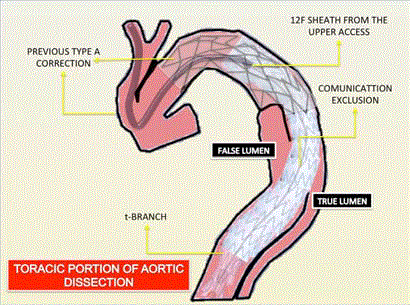
Figure3
Figure 3
Schematic illustration demonstrating the sinuous path the sheath
assumed due to previous dacron graft.
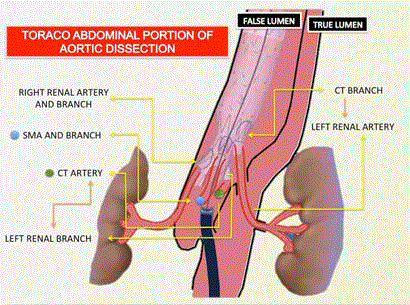
Figure 4
Figure 4
Schematic illustration how the branches of the T-branch may be
modified to attend patient’s anatomy. In this case the celiac trunk branch
was used to catheterize the left renal through the dissection, whereas the left
renal branch was used to catheterize the celiac trunk.
Figure 5
Figure 6
References
- Nienaber CA, Kische S, Rousseau H, Eggebrecht H, Rehders TC, Kundt G, et al. Endovascular repair of type B aorticdissection: long-term results of the randomized investigation of stent graftsin aortic dissection trial. Circ Cardiovasc Interv. 2013;6(4):407-16.
- Scali ST, Feezor RJ, Chang CK, Stone DH, Hess PJ, Martin TD, et al. Efficacy of thoracic endovascular stent repair forchronic type B aortic dissection with aneurysmal degeneration. J Vasc Surg.2013;58(1):10-7.
- Luozzo GD, Wilderman M, Pawale A,McCullough J, Griepp RB. Planned Staged Repair of Thoracoabdominal AorticAneurysms to Minimize Spinal Cord Injury: A Proof of Concept. Aorta (Stamford). 2015;3(5):177-80.
- Oikonomou K, Kopp R, Katsargyris A, Pfister K, Verhoeven EL, Kasprzak P. Outcomes of fenestrated/branchedendografting in post-dissection thoracoabdominal aortic aneurysms. Eur J VascEndovasc Surg. 2014;48(6):641-8.
- Bisdas T, Donas KP, Bosiers M, TorselloG, Austermann M. Anatomical suitability of the T-branch stent-graft in patientswith thoracoabdominal aortic aneurysms treated using custom-made multibranchedendografts. J Endovasc Ther. 2013;20(5):672-7.
- Chiesa R,Melissano G. Aortic Dissections, Patient true stories and the innovations that saved their lives. 2016:321-337.
- Dong Z, Fu W, Wang Y, Wang C, YanZ, Guo D, et al. Stent graft- induced new entry after endovascular repair forStanford type B aortic dissection. J VascSurg. 2010;52:1450e7.
- Nienaber CA, Kische S, Zeller T,Rehders TC, Schneider H, Lorenzen B, et al. Provisional extension to inducecomplete attachment after stent graft placement in type B aortic dissection:the PETTICOAT concept. Endovasc Ther. 2006; 13:786-46.
- Hofferberth SC, Foley PT, Newcomb AE,Yap KK, Yii MY, Nixon IK, et al. Combined proximal endografting with distalbare-metal stenting for management of aortic dissection. Ann Thorac Surg.2012;93(1):95-102.
- Kölbel T, Lohrenz C, Kieback A, Diener H, Debus ES, Larena-Avellaneda A. Distal false lumen occlusion in aorticdissection with a homemade extra-large vascular plug: the candy-plug technique.J Endovasc Ther. 2013;20(4):484-9.
- MastracciT. Aortic Dissections, Patient true stories and the innovations that savedtheir lives. 2016:305-13.
- Kitagawa A,;GreenbergRK,;Eagleton MJ,;Mastracci TM,;Roselli EE. Fenestrated andbranched endovascular aortic repair for chronic type B aortic dissection withthoracoabdominal aneurysms. J Vascular Surgery. 2013;58:625-34.
- Urbanski PP, Bougioukakis P, Deja MA,Diegeler A, Irimie V, Lenos A, et al. Open aortic arch surgery in chronicdissection with visceral arteries originating from different lumens. Eur JCardiothorac Surg. 2016;49(5):1382-90.
- Quinones-BaldrichW, Saleem T. Aortic Dissections, Patient true stories and the innovations that savedtheir lives. 2016:349-56.
- Watanabe Y,;KurataniT,;Shirakawa Y,;Torikai K,;Shimamura K,;Sawa Y. HybridEndovascular repair of dissecting thoracoabdominal aortic aneurysm with stentgraft implantation through the fasle lumen. J Vasc Surg . 2014;59:264-7.
- Kamman AV, William DM, Patel HJ.Thoracic Endovascular Aortic Repair into the false lumen in chronic aorticdissection. Ann Vasc Surg. 2017.