Case Report
The Role of Two New Ratios as Predictive Factors of Oncologic Outcome in Stage III Colon and Intra-Peritoneal Rectal Cancer
Rizzo Gianluca*, Santullo Francesco, Zaccone Giuseppe, Vernes Elisa, Pafundi Donato Paolo, Biondi Alberto, Persiani Roberto, Verbo Alessandro, Mattana Claudio, Manno Alberto, Rubino Serena and Coco Claudio
Department of Digestive System and Metabolic Endocrine System, Abdominal Surgical Area University Hospital
Policlinics Foundation "Agostino Gemelli" - Catholic University of the Sacred Heart, Largo A. Gemelli, 8 - 00168
Rome, Italy
*Corresponding author: Gianluca Rizzo, Department of Digestive System and Metabolic Endocrine System, Abdominal Surgical Area University Hospital Policlinics Foundation "Agostino Gemelli" - Catholic University of the Sacred Heart, U.O.C. Chirurgia Generale 2 - Complesso Columbus Via Moscati, 31-33 – 00168 Rome, Italy
Published: 12 Jun, 2017
Cite this article as: Gianluca R, Francesco S, Giuseppe Z,
Elisa V, Paolo PD, Alberto B, et al. The
Role of Two New Ratios as Predictive
Factors of Oncologic Outcome in Stage
III Colon and Intra-Peritoneal Rectal
Cancer. Clin Surg. 2017; 2: 1504.
Abstract
Purpose: Aim of the study was to evaluate the role of 2 ratios as predictive factors of Overall Survival (OS) and Disease-Free Survival (DFS).
Methods: Stage III colon and intra-peritoneal rectal cancer patients treated from 2000 to 2010
entered the study. A statistical analysis was performed to identify variables predicting OS and DFS.
The role of 2 variables was evaluated: Length-Node ratio (maximal length of the tumor divided by
the lymph node ratio); Length-Node-Tumor ratio (maximal length of the tumor divided by the
product of lymph node ratio and pT stage).
Results: One hundred eighteen patients underwent to radical surgery. The median maximal length of
the tumor was 4 cm. The most frequent pT stage was pT3 (69.4%). The median number of harvested
lymph nodes and lymph node ratio was respectively 15 and 0.154. Median value of Length-Node
and Length-Node-Tumor ratio was respectively 27.083 and 8.889. After a median follow-up of 48
months, the cancer recurrence rate was 33.1%. The actuarial 5-y OS and 5-y DFS were respectively
71% and 66.1%. At multivariate analysis the male sex (p: 0.002), the occurrence of post-operative
complications (p: 0.002) and a value lower than median of Length-Node ratio (: 0.001) and Length-
Node-Tumor ratio (p< 0.001) were identified as independent factors predicting worse OS. A value
lower than median of Length-Node ratio (p: 0.017) and Length-Node-Tumor ratio (p: 0.006) were
identified as the only independent factors predicting worse DFS.
Conclusion: Length-Node and Length-Node-Tumor ratio seem to be prognostic factors significantly
related with the oncological outcome of stage III colorectal cancer.
Keywords: Colon cancer; Intra-peritoneal rectal cancer; Metastatic lymph node; Harvested
lymph nodes; Lymph node ratio; Oncologic outcome
Introduction
Colorectal cancer is the second most common cancer and an important cause of death [1-2].
Five-year survival after surgery for colorectal cancer is significantly related to cancer stage and varied
from 92%–100% for stage I to 0%-9% for stage IV. In III stage colorectal cancer the 5-year survival is
extremely variable, ranging from 33% to 83% [3]. In this group of colorectal cancer patients, several
studies demonstrated the validity of an adjuvant chemotherapy regimen in terms of survival benefit
and, actually, there is a wide range of treatment options from 5-FU to regimens including biological
agents [4-10]. Despite the many therapeutic options for the treatment of III stage colorectal cancer,
the cancer-related mortality remains high and variable. This heterogeneous variability needs a
patient selection to choose the most useful chemotherapeutic regimen. A better way to predict
survival in stage III colorectal cancer is based on the identification of prognostic factors.
Recently, interest has peaked in lymph nodes. The prognostic value of lymph nodes depends not
only on progression of the cancer, but also on other factors, such as the total number of harvested
lymph nodes and accurateness of histological examination [11]. In this contest, analogously to
gastric cancer, was analyzed the role of lymph node ratio. Lymph node ratio (defined as the ratio
between positive lymph node and the total number of harvested nodes) was evaluated as a predictor of recurrence, instead of the actual number of positive lymph nodes,
and several authors have confirmed the prognostic role of this ratio in
colorectal cancer [11-19].
Although studies not always reported coherent results, the
predictive role of size of the tumor in colorectal cancer has been
demonstrated [20-23]. Size of the tumor reflects not only duration
of time that the tumor has been present, but also the aggressiveness
of the tumor. For example, a large tumor could either reflect a slowly
spreading tumor, growing for a long period of time, or a more recent
quickly spreading tumor. Furthermore, the presence of positive lymph nodes can either be associated with a slowly spreading tumor,
which has been present for a long time, or with a recent, but aggressive
tumor. Therefore a tumor getting to a larger size before metastasizing
would have a better prognosis than those that metastasize at a small
volume.
A recent study, published in 2011 by Poritz et al. [24] introduced
a combining ratio, between tumor volume and the percent of positive
lymph nodes, which results as a predictor of 5-year cancer-specific
survival in colorectal cancer in terms of overall survival and diseasefree
survival. However, the exact measurement of tumor volume is a parameter not always available from pathology report. Generally, in
these reports, only one measurement, the maximum diameter, has
been described.
Based on the evidence of this study and hypothesizing that size of
tumor is really an important prognostic factor, especially if related to
lymph node ratio, we conduct a retrospective study with the following
aims:
• To identify, in a population of stage III colon and intraperitoneal
rectal cancer patients, predictive factors of Overall Survival
(OS) and Disease-Free Survival (DFS).
• To evaluate the prognostic role of two ratios including
tumor size, the lymph node ratio and pT stage as predictive factors
of OS and DFS.
Table 1
Table 2
Materials and Methods
This study retrospectively analyzes patients with stage III colon
and intraperitoneal rectal cancer, who underwent radical surgery at
our institution, from January 2000 until December 2010. All cancers
were diagnosed by endoscopic examination and defined by biopsy and
histological examination. Pre-operative cancer staging was performed
in all patients by chest and abdominal Computed Tomography (CT)
or by Abdominal Ultrasound (US) and chest X-ray. Patients who
underwent urgent surgery completed cancer staging during postoperative
hospital stay. Each specimen was analyzed and the tumor
stage was coded as described by the TNM 7th edition Staging of Colon
Cancer [25].
All patients underwent to radical colonic or rectal resection
(complete tumor resection with all margins histologically negative,
R0).
All stage III colon and intra-peritoneal rectal cancer patients
were evaluated for this study. Exclusion criteria were: patients with
carcinoma in situ; stage I, II and IV cancer; patients with synchronous
malignancies other than colorectal cancer; recurrent or perforated
colorectal cancer; colorectal cancer related to inflammatory bowel
disease; cancer of the appendix; extra-peritoneal rectal cancer; anal
canal cancer; patients who did not undergo radical surgical resection
(palliative resection or R1-R2 resection); patients who pre-operatively
received chemo- or radiotherapy; patients who post-operatively
received radiotherapy.
Post-operative adjuvant chemotherapy was recommended to all
patients. After evaluation and patient-selection by oncologist patients
received chemotherapy. All patients entered in a follow-up program
including:
• Hematological examination and screening, including
measuring levels of CEA and CA 19-9, performed at 1,6,12,18,24,36,48
and 60 months from surgery.
• Colonoscopy, performed at 12, 24, 36, 48 and 60 months
from surgery.
• Abdominal US and chest X-Ray, performed at 6, 18 and 30
months from surgery.
• CT-thorax-abdomen, performed at 12, 24, 36, 48 and 60
months from surgery.
Median and range time (months) of follow up were calculated
and the status of patients at last follow-up was recorded. Data were
inserted to a digital database.
A statistical analysis was performed. The variables and subgroups
analyzed are summarized on Table 1. The size of cancer was deducted
by histological report and corresponds to the maximum length of the
tumor measured in centimeters. Lymph node ratio was defined by this
formula: number of metastatic harvested lymph node divided by the
number of all harvested lymph nodes (metastatic and not metastatic).
Length-Node ratio was defined by this formula: maximum length of
the tumor (measured in centimeters) divided by the lymph node ratio.
Length-Node-Tumor ratio was defined by this formula: maximum
length of the tumor (measured in centimeters) divided by the product
of lymph node ratio and pT stage (varied from 1 to 4).
Outcome variables analyzed were:
• OS, defined as the time after primary treatment until
death (months; median survival and actuarial 5-year survival were
calculated).
• DFS, defined as the time after primary treatment until the
first cancer recurrence (months; median survival and actuarial 5-year
survival were calculated).
A Kaplan-Mayer analysis was performed to identify variables
significantly related to OS and DFS. A p-value of 0.05 was considered
to be the cut-off point for significance. Gender, age and variables
with p<0.125 were inserted in a multivariate model (Cox regression)
to evaluate whether these variables correlated independently with
survival.
All statistical analyses were performed by software IBM SPSS
Statistics version 21.0.0.0 for Mac OS X.
Table 3
Results
From 2000 to 2010, 118 patients (66 males and 52 females)
with a median age of 65 years (range: 31-94 years) underwent to
oncological resection for stage III colon and intra-peritoneal rectal
cancer. Baseline characteristics of patients entered in the study are
summarized in Table 2.
The most frequently involved site of tumor were the left colon
(63 patients, 53.4%), followed by the right colon (39 patients, 33.1%),
upper rectum (9 patients, 7.6%) and transverse colon (7 patients,
5.9%). Nearly all patients underwent an elective surgical intervention
(115 patients, 97.5%). A protective stoma was necessary only in 3
cases (2.5%). Eighty-six percent of tumors were adenocarcinoma,
most (57.6%) G1-2. The median value of tumor maximum length was
4 cm (range: 2 cm -13 cm). According to pathological examinations,
the primitive tumor was staged as pT1 in only 1 patient (0.8%), as
pT2 in 8 patients (6.8%), as pT3 in 82 patients (68.5%) and as pT4 in
27 patients (22.9%). The median number of harvested lymph nodes
was 15 (range 4-80) but more than 12 lymph nodes were harvested
in 80.5% of patients. The median number of metastatic lymph nodes
was 2 (ranging 1-45) and the median value of the lymph node ratio
was 0.154 (range 0.014-1.000). The median values of Length-Node
ratio and Length-Node-Tumor ratio were, respectively, 27.083 (range
3.750-350.000) and 8.889 (range 0.940-875.000). No short-term postoperative
deaths were recorded. Short-term post-operative morbidity
was recorded in 23.7% of cases and an anastomotic leakage was
observed in 4 cases (3.4%). Eighty-seven percent of patients received
post-operative chemotherapy. The remaining patients did not receive
adjuvant therapy because of comorbidity. The median follow-up period was 48 months (range: 7-132
months). Thirty-nine patients (33.1%) developed cancer recurrence: 4
of these (3.4%) were local recurrences and 35 were distant metastases
(29.8%), of which 20 (16.9%) were observed in the liver, 8 (6.7%) in
the lung, and 7 (5.9%) in other sites. At time of the last follow-up,
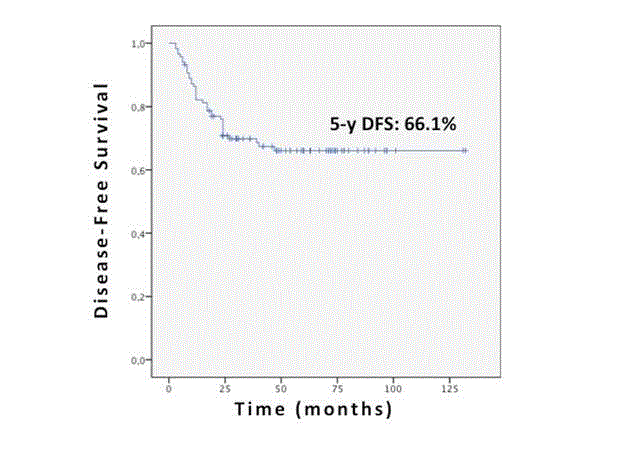
78 patients (66.1%) were alive without evidence of cancer recurrence
and 5 patients (4.2%) were alive with a cancer recurrence. Thirty-five
deaths were recorded: 25 cancer-related deaths (21.2%) and 10 (8.5%)
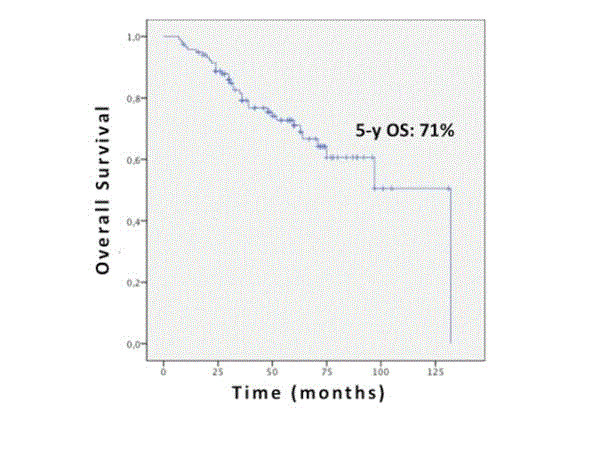
due to other causes. Actuarial 5-year OS and DFS were respectively
71% (Figure 1) and 66.1% (Figure 2).
About OS, at Kaplan-Mayer analysis (Table 3), the male sex (p:
0.005), the occurrence of short-term post-operative complications (p:
0.010), the presence of more than 2 metastatic lymph nodes (p: 0.022),
a number of harvested lymph nodes less than 12 (p: 0.031), a lymph
node ratio value higher than median value (p: 0.005), a Length-Node
ratio value less than median (p: 0.002) and a Length-Node-Tumor
ratio value less than median (p: 0.001) were significantly related
to a worse 5-year OS. At multivariate analysis, male sex (5-y OS in
male: 61.1% vs. 5-y OS in female: 82.9% - HR: 4.223; 95% C.I.: 1.705-
10.460; p: 0.002), the occurrence of post-operative complications (5-y
OS if complications occurred: 58.7% vs. 5-y OS if no complications
occurred: 75.1% - HR: 3.512; 95% C.I.: 1.613-7.646; p: 0.002), a
Length-Node ratio value less than median (HR: 105.396; 95% C.I.:
7.334-1514.716; p: 0.001) and a Length-Node-Tumor ratio less than
median (HR: 123.535; 95% C.I.: 8.596-1775.401; p< 0.001) were
observed as variables independently related to impaired OS at 5 years.
About DFS, at Kaplan-Mayer analysis (Table 4) the presence of
more than 2 metastatic lymph nodes (p: 0.003), a lymph node ratio
value higher than median (p: 0.013), a Length-Node Ratio value less
than median (p: 0.006) and a Length-Node-Tumor Ratio value less
than median (p: 0.002) were significantly related to a worse 5-year
DFS. At multivariate analysis only a Length-Node Ratio value less
than median (HR: 22.676; 95% C.I.: 1.990-258.362; p: 0.017) and a
Length-Node-Tumor Ratio less than median (HR: 29.957; 95% C.I.:
2.629-341.313; p: 0.006) were observed as variables independently
related to impaired DFS at 5 years.
Figure 1
Discussion
The TNM staging system is considered the most important
prognostic factor we have for predicting survival in colorectal cancer,
with mean survivals of 93%, 78%, 60% and 8% for stage I, II, III and
IV, respectively [26]. Size of the tumor is not part of the staging
system in colorectal cancer. However, several studies have shown a
correlation between tumor size and cancer-related survival [20-24]. A
recent Austrian study, performed on a total of 381 patients suffering from colorectal cancer at any pathological stage, demonstrated a
direct correlation between tumor size and the cancer-related survival.
The authors considered a cut-off of 4.5 cm and, applying it, tumors
exceeding this size were significantly associated with progressionfree
and cancer-specific survival [21]. This correlation is certainly
true for all pathological stages, hypothesizing that a larger tumor has
been in place longer, and it is therefore more likely to have a greater
intraparietal, lymphatic, and hematogenous invasiveness.
The metastatic spread to loco-regional lymph nodes is considered
as an important prognostic event in most solid cancers. Number of
lymph node harvested, the number of metastatic nodes and lymph
node ratio were more investigated as relevant prognostic factors of
oncological outcome in colorectal cancer. Regarding the number of
harvested nodes, in a systematic review on six studies analyzing stage
III colon cancer a positive correlation between number of lymph nodes
evaluated and improved survival was demonstrated [27]. The role of
the number of metastatic nodes is underlined on the 6th and 7th edition
of TNM classification who’s classified the stage III in three subgroups
on the basis of the number of positive nodes [28]. However, it seems
evident that the prognostic significance of 4 positive nodes on a total
of 4 examined is different when a total of 35 nodes were retrieved.
On the basis of this idea, several studies established the role of lymph
node ratio as prognostic factors for III stage colon cancer. In 2010, a
systematic review analyzed the prognostic role of lymph node ratio
in stage III colorectal cancer. This analysis, including sixteen studies
(on 33984 patients), demonstrated the prognostic role of lymph node
ratio, and its superiority to the number of metastatic nodes, both for
overall survival than for disease-free survival [13]. However, the INT-
0089 trial noted that the prognostic value of lymph node ratio was
maintained only if more than 10 lymph nodes were harvested [12].
The lymph node status could represent a way to understand the
role of tumor size as a predictor of the aggressiveness of the tumor.
Positive lymph nodes could be present in a slowly spreading large
tumor, being in place for many years, or in a small recent tumor that
has spread quickly. Thus, it seems intuitive to assume that tumors
that get to a larger size before metastasizing, have a better prognosis
than those metastasizing at a small volume. Taking in mind this
hypothesis, in 2011 Poritz et al. [24] demonstrated the prognostic
value of a ratio between the volume of the tumor (calculated by
multiplying the two largest tumor measurement) and lymph node
ratio. This study analyzed 63 patients, of who 35 were alive without
evidence of disease and 28 who developed distant metastases during
the 5-year follow-up. After analysis of DFS, the ratio was discovered
to be the only variable correlated significantly and independently with DFS. Furthermore, based on this evidence, the authors formulated a
complex algorithm to determine the probability that a particular case
of colorectal cancer would develop distant metastasis [24]. However,
the exact calculation of the volume of a neoplasm is a parameter
which is not always available in pathology report. Generally, in these
reports, only one measurement, the maximum diameter, has been
described. Furthermore, the practical use of the algorithm reported
by the authors appears to be not easily applicable to each patient.
To overcome this difficulty, but believing in the existence of a role
of tumor size as prognostic factor (protective factor) in relation to
the lymph node ratio (risk factor) and pT stage (risk factor), we
introduced the Length-Node ratio and the Length-Node-Tumor ratio,
easily calculated from data provided by each pathological report: the
maximum diameter of the tumor, the lymph node ratio and the pT
stage. As found in the study of Poritz et al., our study demonstrate
a statistically significant correlation between Length-Node-Tumor
ratio and the most important oncological outcome measurements:
OS and DFS. The prognostic role demonstrated by these ratios was
higher than other validated prognostic factors singularly taken, such
as lymph node harvested, number of metastatic nodes, lymph node
ratio and pT stage. Moreover, these ratios were the only variables
indipendently related to DFS.
About OS, also male sex and the occurrence of post-operative
complications resulted statistically related to worse survival. At a large
American study based on 39325 patients with colorectal cancer from
SEER database [29] women had significantly longer survival especially
after rectal resection; however, they present more emergently and at
an older age. Several studies tried to identify the cause of this survival
discrepancy between men and women in colorectal cancer. Especially
in rectal cancer, anatomic differences (narrow male pelvis) have been
offered as the explanation for differences in cancer survival between
men and women [30]; however, anatomy is unlikely to explain
differences in survival observed after colorectal cancer resection.
Some authors speculate that differences in circulating hormones or
in the immunologic response to cancer between women and men are
responsible for the survival advantage. McArdle et al. [30] argues that
poor survival in men may be the result of an ongoing inflammatory
response, in the form of elevated C-reactive protein, which seems
more detrimental in men than in women. From another point of
view, Wichmann et al. [31] speculates that circulating levels of
estrogen stimulate a protective immune response to tumors, whereas
circulating testosterone results in detrimental immune response.
Several authors have reported increased local tumour recurrences
after post-operative complications and, particularly, after anastomotic
dehiscence in patients undergoing resection for colorectal cancer
[32-34]. Moreover, a large meta-analysis, including 21 studies with
a combined patient population of 21,902 patients, demonstrate that
post-operative anastomotic leak has a clear and negative prognostic
impact on local recurrence and on survival, more underlined in
colorectal anastomoses [35]. During the acute and subsequent
chronic inflammation that accompanies a complication, a variety of
acute phase reactants and proinflammatory mediators are released.
In recent years, an increasing body of high quality experimental work
has demonstrated that many of these inflammatory biomarkers are
implicated in tumor proliferation, survival, avoidance of apoptosis,
progression to metastasis, and resistance to chemotherapy [36].
One example is the proinflammatory interleukin IL-1, which can
enhance the growth and progression of colorectal cancer, is highly
expressed in advanced cases, and antagonists of IL-1 inhibit tumor
growth in experimental models [37,38]. Other proinflammatory
mediators implicated in the molecular link between inflammation
and cancer include the tumor necrosis factor family of proteins, IL-
6, cyclooxygenase 2, matrix metalloproteases, nuclear factor kappa
Band the vascular endothelial growth factor family [36,39]. The
correlation between inflammatory response and cancer recurrence is
further supported by the emerging data from other cancer, especially
breast cancer. In this type of cancer, elevated markers of inflammation
resulted significantly associated with a reduced overall and diseasefree
survival [40,41].
Major limitations of this study include the retrospective design
of the study and the relatively small sample size, since these factors
could have introduced a selection bias.
Figure 2
Table 4
Conclusion
Length-Node and Length-Node-Tumor ratios seem to be important prognostic factors, significantly related with the oncological outcome of stage III colon and intra-peritoneal rectal cancer where the range of survival after surgery is very large. So, the introduction in clinical practice of these parameters, extremely simple to calculate, could help to identify colorecatal cancer patients with a major risk of cancer recurrence and with a potential need of a more aggressive chemotherapeutic regimen. More large studies are needed to confirm the prognostic role of these ratios.
Footnotes
RG and CC have made substantial contributions to conception,
design, analysis and interpretation of data, have been involved
in drafting the manuscript or revising it critically for important
intellectual content and have given final approval of the version
to be published. SF, ZG, PDP, RS and VE have made substantial
contributions to acquisition of data. BA, VA, MC, MA and PR have
given final approval of the version to be published.
RG, PR, MC and CC are members of the Italian Society of
Colorectal Surgeons (SICCR)
PR and CC are fellows of the American College of Surgeons
(FACS)
This study represents the master thesis of VE.
References
- Mennigen R, Senninger N, Laukoetter MG. Novel treatment options for perforations of the upper gastrointestinal tract: Endoscopic vacuum therapy and over-the-scope clips. World J Gastroenterol. 2014;20(24):7767-76.
- Kuehn F, Loske G, Schiffmann L, Gock M, Klar E. Endoscopic vacuum therapy for various defects of the upper gastrointestinal tract. Surg Endosc. 2017.
- Loske G, Schorsch T, Müller C. Intraluminal and intracavitary vacuum therapy for esophageal leakage: a new endoscopic minimally invasive approach. Endoscopy. 2011;43(6):540-4.
- Neumann PA, Mennigen R, Palmes D, Senninger N, Vowinkel T, Laukoetter MG. Pre-emptive endoscopic vacuum therapy for treatment of anastomotic ischemia after esophageal resections. Endoscopy. 2017;49(5):498-503.
- Loske G, Schorsch T, Dahm C, Martens E, Müller C. Iatrogenic perforation of esophagus successfully treated with Endoscopic Vacuum Therapy (EVT). EndoscInt Open. 2015;3(6):E547-51.