Review Article
Atrial Myxoma: From Diagnosis to Management
Boutayeb A*, Mahfoudi L and Moughil S
Department of Cardiovascular Surgery, Ibn Sina University Hospital, Rabat, Morocco
*Corresponding author: Alaae Boutayeb, Department of Cardiovascular Surgery, Ibn Sina University Hospital, 6766- Madinat Al Irfane 10000 Rabat, Morocco
Published: 07 Jun, 2017
Cite this article as: Boutayeb A, Mahfoudi L, Moughil S.
Atrial Myxoma: From Diagnosis to
Management. Clin Surg. 2017; 2: 1498.
Abstract
The purpose of this paper is to review and discuss all the topics and issues related to the diagnosis and
management of atrial myxomas. These tumors represent the most common primary heart tumors
and may present with a wide range of symptom spectrum making the diagnosis sometimes difficult.
Echocardiography is the gold standard tool for diagnosis as well as follow-up. Due to its potential
serious consequences, myxoma should be removed as soon as possible. While surgery results in
excellent overall survival and freedom from reoperation rates, annual follow-up is recommended
particularly in familial cases.
Keywords: Myxoma; Cardiac tumor; Carney complex
Introduction
Myxoma is a neoplasm composed of stellate to plump cytologically bland mesenchymal cells set in a myxoid stroma [1]. Myxoma was first diagnosed in 1952 by Goldberg [2]. This neoplasm is the most common cardiac benign tumor. Nevertheless, many of its features still remain unknown or controversial. In this paper, we review recent scientific data concerning diagnosis and management modalities of myxomas.
Epidemiology
Myxomas represent 0.25% of all heart diseases [3]. Even if their annual incidence rarely exceeds
0.5 to 1 case per million individuals [4,5], myxomas are the most frequent primary cardiac tumors
in adults and represent more than 50% to 85% of benign ones [2,6,7].
Epidemiologically, myxomas show a female predominance with a sex ratio of 3:1 [4,5] and
are generally classified into two main epidemiologic forms: the familial and the sporadic. On one
hand, the latter type, representing 95% of all cases [7], affects mainly middle age women. Tumor
is more commonly unique and localized in the left atrium. On the other hand, the familial type
affects more frequently young males. Tumors are usually multiple and may occur in other cardiac
chambers. This type has autosomal dominant inheritance and falls under Carney's syndrome. The
latter was described in 1985 and combines cardiac and extracardiac myxomas as well as cutaneous
pigmentation (lentiginosis periorificial, café-au-lait spots, blue nevi) and endocrine tumors (Cushing
syndrome, breast fibroadenoma, testicular tumor, acromegaly...) [8,9]. Carney complex is a multiple
neoplasia syndrome caused by PRKAR1A, inactivating mutations associated with chromosome
17q22-q24. These genes encode the cAMP’s (cyclic adenosine monophosphate) regulatory subunit
of the dependent protein kinase A. Nevertheless, their role still remains imprecisely known [10,11].
Histogenesis
Myxoma’s origin remains inadequately understood as the theory stating that it derives from
thrombi is progressively rejected [12,13]. Furthermore, myxomas were alleged to arise from
microscopic endocardial structures located in the fossa ovalis, known as Prichard structures [14].
By detecting the antigens and genetic material of the Herpes simplex virus type 1 (HSV1) in 70%
of cases of operated myxoma, Li et al. [15] suggest that cardiac myxomas could result from chronic
inflammation of the endocardium induced by viral infection.
The conflicting hypotheses on the histogenesis of cardiac myxoma originate from two main
contributing factors: the heterogeneous phenotype of myxoma cells, as well as the different approaches
in their morphological and immuno-histochemical characterization. Nevertheless, it is relevant to
point out that, currently, most authors believe myxoma derives from multipotent mesenchymal
stem cells [16]. Indeed, myxoma tumoral cells exhibit two characteristics. On one hand, it exhibits
the phenotypic markers of embryonic endothelial-to-mesenchymal transformation, which precedes
terminal differentiation of endocardial cushion formation involved in cardiac septation and valve formation. On the other hand, it displays markers of primitive cardiac mesenchymal differentiation [17]. This hypothesis is further
supported by the presence of substantial similarities between the
following three types of cells: the endothelial lining ones, the cells
within the cushion tissue and finally the cardiac myxoma’s lepidic
ones [6]. However, it still remains unclear from which the cardiac
myxoma derives from but there are currently three main leads. The
first one is the embryonic remnants of cardiac cushions, the second,
the primitive multi-potential mesenchymal cells existing in adult
hearts and the third, the ectopic de novo re-expression of early
cardiomiogenic phenotype in adult cardiac cells [17].
Finally, other authors support the nervous origin of myxomas.
Based on immuno-histochemical data, Krikler et al. [18] affirm that
myxomas originate from a neuroendocrine tissue. The observed
pattern of neuroendocrine markers and distribution of VWF/FVIII,
CD34 and SMA (smooth muscle actin) led Krikler et al. [18] to suggest
a possible neuroendocrine origin of cardiac myxoma. This origin was
further supported by the findings of Teraccianno et al. [19]. In fact,
they detected strong and diffuse immune-histochemical staining of
CALB2 (calretinin/calbindin 2 protein normally detected in the cells
of the central and peripheral neural tissue) in the cytoplasm and
nucleus of neoplastic cells in all examined sporadic cardiac myxoma
cases [20].
Figure 1
Anatomical Pathology
Myxomas are intra-cavitary tumors that mainly affect atrial
chamber (75% LA and 18% RA). They arise from inter-atrial septum
near the fossa ovalis but rarely anteriorly or posteriorly to the atrial
walls, or even auricles. In 6% of cases, these tumors originate in the
ventricles (RV 8%, 4% LV) or in the atrioventricular valves (1%).
Complementarily, myxomas can be bi-atrial or, more rarely, multifocal
[21].
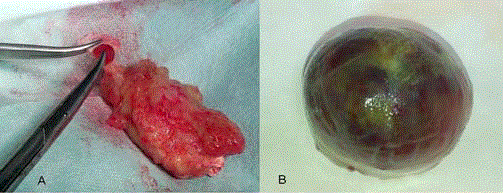
Macroscopically, the myxomas’ size varies from 1 to 15 cm,
with an average of 5 cm to 6 cm [22,23]. Myxomas can be either
pedunculated or have a wide base of implantation.
St. John Sutton’s classification distinguishes between two forms
of myxomas: solid and papillary [24]. The first group represents 2/3
of myxomas and corresponds to solid tumors, sometimes polypoid,
with unstriated and smooth surfaces related to a high superficial
collagenation. Papillary myxomas are brittler and characterized by
their irregular shape. One particular characteristic, explained by the
secretory activity of these tumors, is the release of metalloproteinase
and enzymes which degrades continuously the extracellular matrix
and therefore creates an imbalance between the process of synthesis
and tissue fragmentation [25]. These characteristics explain why
obstructive heart failure is usually associated with solid tumors while
embolic events represent the most common clinical feature of fragile
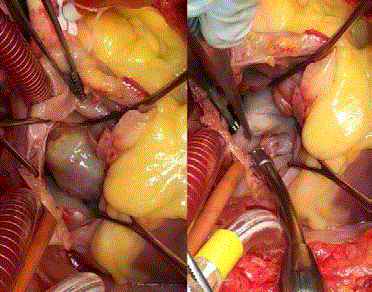
papillary myxoma [26] (Figure 1).
The histology of cardiac myxoma resembles closely the
mesenchymal tissue, forming vascular structures. It is characterized
by a myxoma stroma rich in elastin, collagen and proteoglycans in
which reside small fusiform or stellate cells with round or oval nuclei
and scarce eosinophilic cytoplasm [27]. These cells are sometimes
multi-nucleated. They are shaped and structured in chained rings, or
in nests all around the capillaries. Other cells can also be observed like
lymphocytes, plasma cells, histiocytes and mast cells, which may all
together contribute to systemic manifestations [27].
Myxomas are often infused by thin-walled vessels lacking
pericytes, versus thick-walled ones located at the implantation
base. The tumor surface is often covered with a layer of flattened
endothelial cells which form small vascular spaces or invaginations.
In some locations, this endothelium is covered by lipid cells.
Most authors agree on the benign aspect of myxomas.
Nevertheless, these tumors have a local and remote extensive
potential [8]. As a consequence, myxomas are considered biologically
benign but “functionally malignant” tumors. In fact, many brain
metastases localizations, as well as arterial and bone (sternum, spine
and pelvis...) have been described in the literature [28-30]. Some
authors believe that these metastases result from the persistence
within the tumor fragments disseminated of live tumor cells capable
to grow and multiply [31]. This tumor progression is promoted by
the autocrine expression of growth factors and angiogenesis (VEGF,
vascular endothelial growth factor and PDGF, platelet-derived
growth factor), which acts upon the cytoplasmic receptors [32-35].
Similarly, inflammatory cytokine (IL6, IL8), abundantly secreted
by the neoplastic tissue, may also enhance CM (cardiac myxoma)
vascularization and migration as well as proliferation of their cells
[21]. Furthermore, others antigenic signals are induced by monocyte
chemotactic protein-1 (MCP-1) and thymidine phosphorylase (TP)
which are expressed by myxoma cells [36]. Further studies are needed
to prove, first, the involvement of other markers such as MIA and
S100 in tumoral growth; second, the recurrence or even the exact
nature of the extra-cardiac dissemination or malignant potential of
cardiac myxoma [21].
Besides, it was found that some recurrent lesions may exhibit
more aggressive histology and significantly faster cell proliferation
[37-39]. While some authors suggest successive malignant alteration
of benign myxomas, others think that these tumors correspond to
undiagnosed malignant primary tumors [40-43].
Diagnosis
Clinical presentations of cardiac myxomas are polymorphic and
unspecific. They depend for the most part on their base, shape, size and
mobility. As a consequence, a wide spectrum of clinical manifestations
ranging from asymptomatic forms, identified erratically, to severe
ones with complications involving life-threatening prognosis. While
in the series of Beghetti et al. [44] more than half of the patients
were diagnosed during a routine examination, others stated that
only 10% to 15% of myxomas are asymptomatic and constitute the
prerogative of small tumors [21]. The data in the literature related to
the myxoma’s growth remains imprecise but it can be inferred from a
certain set of observations. Oldershaw et al. [45] reported three cases
of myxoma quiescent in patients ranging from 7 to 16 years! Roudaut
and Allal reported respectively 1 and 3 cases of left atrial myxomas
which developed within the 8, 11, 12 and 14 months following the first echocardiography without visible image [46-47]. The reported
growth rate of left atrial myxomas varies from 0 to 1.3 - 7mm/month
[48-49].
Hemodynamic consequences
The hemodynamic consequences reflect in signs of left heart
failure (dyspnea, paroxysmal nocturnal dyspnea, orthopnea or
pulmonary edema) or right one (venous hyper pressure, lower limb
edema, and hepatomegaly). The symptoms are often progressive but
may have a positional character. Because of their atrial localization,
myxomas can compromise systemic or pulmonary venous drainage
or hinder valve motion. On one hand, they can create a barrier to the
passage of blood from the atria to the ventricles. This obstruction,
progressive or intermittent, often simulates a mitral or tricuspid
stenosis and can cause dyspnea, malaise, or sudden death [50]. This
intra-cardiac obstruction is found in approximately 50% of cases, but
may appear later in the disease evolution [24,51]. On the other hand,
these tumors can cause atrioventricular valvular regurgitation mainly
due to impairment of valve closure or even leaflet damage [52].
Indeed, several valvular destruction mechanisms have been reported:
mechanical destruction, chemical or infectious. In our experience, we
have operated a young patient for whom we discovered a small crack
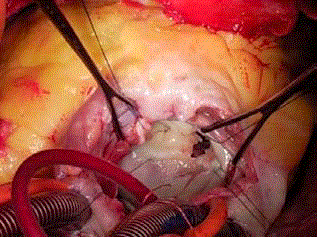
in the mitral valve after resection of left atrial myxoma. Mitral valve
repair was performed with a satisfactory result (Figure 2).
Embolic events
Embolic complications represent a serious complication of
myxoma. This phenomenon is related to the migration of the tumor
or its fragmentation, or even the posting of thrombi and vegetations
adherent to the tumor surface.
Patients (30% to 45%) with left atrial myxoma get complicated
with systemic emboli [53]. While all organs may be affected,
nevertheless, the central nervous system remains the most affected
(more than 50% of cases) [54-57]. Cases of retinal emboli, renal
mesenteric coronary or lower limbs have been reported [55,58-63].
It’s widely admitted to consider at high risk of embolism, patients
presenting with some threatening echocardiographic images papillary
of large multi-lobed tumors, or intermittently prolapsing mass
[64]. Keeling searching for common characteristics of patients who
experienced embolic events in their series, highlighted pedunculated
myxoma (76.9%), an atrial tachyarrhythmia in six (46.2%),
involvement of mitral valve tissue (38.5%), as well as prolapse through
the mitral valve (30.8%). Moreover, the combination of two factors
was detected in almost all patients [5].
Right atrial myxomas seem to be less emboligenic. Although it
affects only 10% of cases, this complication can cause massive and
fatal pulmonary embolism or pulmonary arterial obstruction with
secondary pulmonary hypertension. Furthermore, paradoxical
embolisms seem to be rare [65].
General signs
General signs appear in approximately 90% of patients and may
be the sole symptoms in 30% of cases [7,66]. It generally consists of
fever, weight loss, Raynaud's phenomenon, arthralgia and myalgia.
These symptoms are often accompanied by a procession of biological
abnormalities, such as chronic anemia, thrombocytopenia, or the
inflammatory syndrome (leukocytosis, elevated ESR and CRP as well
as hypergammaglobulinemia).
These events reflect an inflammatory response as well as immune
reaction against the tumor, or even immune response reaction to
the heart muscle mediated by the presence of neoplasm [5]. These
reactions invove the activation of numerous humoral and cellular
cascades but they can also be explained by embolic or mechanical
phenomena (destruction of blood elements) [7,21,68].
All constitutional manifestations are usually reversible and
completely resolved after complete surgical excision of tumor
tissue. However, these parameters may undergo a change in cases of
recurrence of the disease [5].
Rarely, myxoma can become infected. This added infection making
the tumor more friable increases the embolic risk. Streptococcus is
the most frequently incriminated germ. More rarely, other bacterial
or fungal agents were found (Enterococcus faecalis, Staphylococcus
lugdunensis, Gemella morbillorum, Porphyromonas asaccharolytica,
Candida albicans and Histoplasma capsulatum) [69-70].
Paraclinical investigations
The diagnostic significance of the ECG (electrocardiography)
and chest X-ray remains very limited. These two para-clinical tests
provide only non-specific signs. Illustratively, ECG may show atrial
fibrillation, bundle branch block or even abnormal P waves. Similarly,
the chest radiograph can emphasize on cardiomegaly secondary to
atrial cavities enlargement.
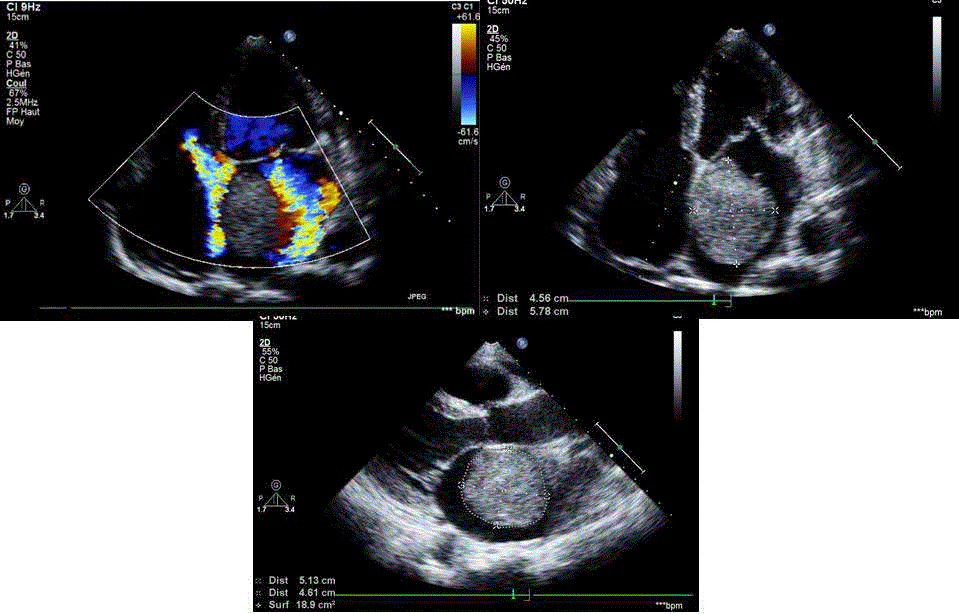
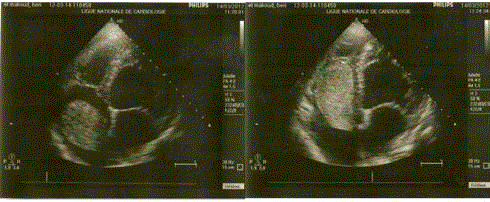
Currently, echocardiography remains the key examination tool
for the diagnosis of atrial myxoma. It enables the diagnosis and
determines the localization, shape, and size of the tumor and its
various connections with the adjacent cardiac structures (Figure 3
and 4).
Trans-thoracic echocardiography is the most commonly used.
It typically provides all the information necessary prior to surgical
resection, but transesophageal echocardiography has, to our
knowledge, enhanced specificity and sensitivity. Transesophageal
echocardiograms detect tumors as small as 1–3 mm in diameter. It
is particularly helpful to evaluate the posterior left atrial wall, atrial
septum, and right atrium, which often are not well displayed on transthoracic
examination, in order to potentially exclude the possibility of
bi-atrial multiple tumors [71]. Ultra-fast CT, PET (Positron Emission
Tomography) scan and especially cardiac MRI (Magnetic Resonance
Imaging) provide also complementary information on myxomas.
However, these investigations should be reserved for cases in which
the diagnosis or characterization of the tumor remains unclear after
an echocardiographic evaluation [72]. In exceptional cases, the
diagnosis is made on the basis of the embolic material’s histological
examination [73].
Figure 2
Figure 3
Figure 4
Natural History & Surgery
In general terms, myxoma represents an emergency. Typically,
the treatment has to be provided subsequent to the diagnosis given
the sudden death risk and embolism affecting approximately 10% of
patients waiting for surgery [74]. This approach is widely accepted;
however, some authors think that emergency management appears
to be less clearly indicated in some stable patients having tumors less
than 2 cm large [64]. In such patients, the risk of embolism seems
to be low and is not amplified by a 2 days’ preoperative assessment.
The latter will allow performing surgery under better conditions and,
obviously, with improved outcomes, particularly in elderly or high
risk patients [64].
Since the first surgical excision under ECC (extracorporeal
circulation) of atrial myxoma by Craaford in 1954 [75], surgery
remains the gold standard despite isolated experiences relating the
feasibility of percutaneous resection in some particular cases [76]
Surgery is usually performed through a median sternotomy
and cardiopulmonary bypass. It is important to minimize cardiac
manipulation to prevent embolic complications. Furthermore, the
vent should be inserted after aortic clamping in cases of left atrial
myxoma.
Minimally, invasive surgery has been increasingly used in the field
of cardiac surgery and was applied to excision of myxoma. Ko et al.
[77] reported the case of 3 patients who received excision of left atrial
myxoma. The procedures were performed through a right anterior
sub-mammary mini thoracotomy or right parasternal incision with
the assistance of endoscopy during femoro–femoral cardiopulmonary
bypass. Ravikumar et al. [78] reported five consecutive patients with
cardiac myxoma who underwent minimally invasive excision of
the tumors. In two patients, the approach was the right parasternal
mini thoracotomy and the subsequent 3 patients had direct-access
partial sternotomy. Yu and colleagues reported the use of extensive
thoracoscopic surgery in resecting cardiac myxomas in 12 cases with
10 in the left atria and 2 in the right atria. Extensive thoracoscopic
surgical resection of myxoma was successfully performed in all cases
through three minimal incisions, with the largest incision being less
than 3 cm long. It ensured a safe outcome and achieved complete
tumor resection [79].
Despite CPB (cardiopulmonary bypass) and aortic cross clamping
durations, these experiences confirm the feasibility and the positive
results of this approach. Indeed, minimally invasive surgery can
deliver the promise of expediency, safety, minimal discomfort, less
postoperative pain, quick functional recuperation, excellent cosmetic
healing, shortened hospital stays, and therefore cost savings.
There is no real consensus on the modalities of surgical
management of cardiac myxomas. In the study conducted by Jones
et al. [80] the frequency of the various operative approaches was as
follows: biatrial, 38%; left atriotomy, 34%; and transseptal, 28%.
Jones et al. [80] stated that the surgical approach to atrial
myxomas should (1) allow minimal manipulation of the tumor, (2)
provide adequate exposure for complete resection of the tumor, (3)
allow inspection of all four heart chambers, (4) minimize recurrence,
and (5) be safe and efficient. It is therefore conclusive that the left
atriotomy by itself is an approach that does not meet all these criteria.
The impossibility of an exploration of the four heart chambers can
be easily compensated by new methods of medical imaging and
echocardiography. However, due to sub-optimal exposure especially
in large mass, this route requires an excessive manipulation of the
tumor with a risk of secondary fragmentation. The main limitation
of the left atriotomy probably remains the impossibility to obtain
wide resection of implantation sites with free margins. In fact, 34 to
85% cases had a subendocardial, not full-thickness, resection of the interatrially based tumor [81,82]. We strongly agree with Garatti and
his associates [83] who propose this approach to patients with small
myxoma attached to the posterior wall of the left atrium or even the
mitral valve.
The bi-atrial approach was introduced and promoted by Cooley
in 1973. It brings several advantages: (1) definition of tumor pedicle
by direct visualization, (2) minimal manipulation of the tumor, (3)
adequate margins of excision (4) inspection of all heart chambers,
and (5) secure closure of the atrial septal defect [84]. Trans-septal
approach (Figure 5) encompasses all these benefits, even with
minimally invasive surgery. Nevertheless, this approach seems to be
correlated to a lower risk of bleeding and rhythm disorders, as well as
postoperative conduction [85,86].
Another important problem in the surgical treatment of atrial
myxoma is solving the tumor basis in order to prevent tumor
recurrence. Thus, ideally, surgery has to allow complete atrial
excision of the tumor with an adequate rim of interatrial septum.
The site of the attachment should be respected with a clear margin.
Myxomas not attached to the septum may undergo a full or a
subendocardial wall thickness resection or even thermocauterization.
Thereafter, all defects must be repaired by direct closure or by using
synthetic or pericardial patch. An inspection of the various cavities
and an extensive washing with serum are highly recommended to
remove any tumor fragments. Moreover, atrioventricular valves
function should be controlled per-operatively with saline and
subsequently with transesophageal echocardiography.
Right sided atrial myxomas are approached through right
atriotomy. Unless using femoral venous cannulation, both the
superior and the inferior venae cavae have to be cannulated directly,
and, as much as possible, by using the right angled cannula to avoid
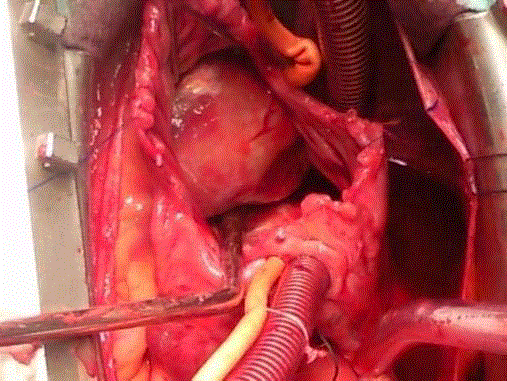
tumoral fragmentation and embolization (Figure 6).
We already reported the case of a 57 year-old woman with a
large right atrial myxoma and severe left ventricular dysfunction who
underwent successful on-pump beating heart resection. The mass was
completely removed after a side-clamping of both the base as well as
the surrounded atrial septum [87].
Data from the literature show that myxoma’s surgery remains
associated with low mortality. In most series, hospital mortality rarely
exceeds 3% [66]. Virtually all deaths are in patients with advanced
disability or old age, the mode of death being generally related not
to the atrial myxoma itself but to coexisting cardiac or degenerative
disease [66]. The major postoperative complication remains the
rhythm disorders. Among the 83 patients who underwent excision of
myxoma, Centofanti et al. [88] observed atrial arrhythmias in 31 cases
(37.3%). More recently, Garrati et al. [83] and Lad et al. [89] observed
transient atrial fibrillation in respectively 6 out of 98 cases and 1 out
of 56 cases. The observed low incidence of atrial arrhythmias in these
two studies may further strengthen the conservative nature of the
trans-septal approach. Moreover, normal sinus rhythm was restored
in all these cases using amiodarone [83,88,89].
On the long run, survivors are generally in good health unless they
have other disease processes or residual effects from a preoperative
embolic event [90]. The most common complication is recurrence.
It may occur within just a few months or up to several years after
the initial resection (average of 4 years) [21]. There is also a report
of a patient whose secondary lesion was diagnosed 20 years after the
primary tumor’s removal [91]. This complication is more frequent
in the familial types (12%–22%) versus the sporadic ones (1%- 4%).
Similarly, the recurrence rate in sporadic myxomas with abnormal
DNA was significantly higher compared to overall sporadic cases
(12%–40%) [92]. According to McCarthy and colleagues [93], the
risk of tumor recurrence is much higher in the familial variety (10%),
in the syndrome of Carney complex (21%), and in the presence of
multiple myxomas (33%).
The explanation of the intracardiac recurrence is related to the
following features: (1) family predisposition, (2) unrecognized
multicentric origin of primary lesion, (3) incomplete resection
or intraoperative dissemination of tumor cells, and, (4) the de
novo proliferation of the pre-tumor or reserve cells present in the
endocardium [94]. The role of incomplete resection still remains
controversial since recurrent tumors often do not resurge at the same
site of the original lesion. Consequently, it is necessary to perform
annual echocardiography throughout a patient’s life, and particularly
in patients with multifocal, atypical, or familial myxomas. In other
cases, echocardiography at 5-year intervals seems adequate [95-99].
Figure 5
Figure 6
Conclusion
Myxomas are the most frequent among cardiac tumors and
may present with a wide range of symptom spectrum. Due to their
potential fatal consequences, myxomas are usually removed as soon as
diagnosed. Early diagnosis is currently obtained by echocardiography
which represents the gold standard tool for diagnosis.
Various surgical approaches are possible. Most centers usually
prefer the biatrial and transseptal approaches. Surgery with
complete excision of the tumor, results in excellent survival rate but
do not exclude the risk of reoperation. Thus, annual follow-up is recommended particularly in familial type. Further studies are still
required to assess histogenesis and genetics of atrial myxoma.
References
- Burke AP, Tazeelar H, Gomez-Roman JJ. Benign tumours of pluripotent mesenchyme. In: Travis WD, Brambilla E, Muller- Hermelink HK, Harris CC, eds. World Health Organization tumours of the lung, pleura, thymus and heart. Lyon: IARC Press. 2004;260-5.
- Goldberg HP, Glenn F, Dotter CT. Myxoma of the left atrium: diagnosis made during life with operative and postmortem findings. Circulation. 1952;6(5):762-7.
- Fayard JM, Maurice P. Myxome cardiaque. Conférence de Cardiologie. 1980;25:31-3
- Yoon DH, Roberts W. Sex distribution in cardiac myxomas. Am J Cardiol. 2002;90(5):563-5.
- Keeling IM, Oberwalder P, Anelli-Monti M, Schuchlenz H, Demel U, Tilz GP, et al. Cardiac myxomas: 24 years of experience in 49 patients. Eur J Cardiothorac Surg. 2002;22(6):971-7.
- Wold LE, Lie JT. Cardiac myxomas: a clinicopathologic profile. Am J Pathol. 1980;101(1):219-40.
- Reynen K. Cardiac myxomas. N Engl J Med. 1995;333(24):1610-7.
- Bertherat J. Carney complex (CNC). Orphanet J Rare Dis. 2006;1:21.
- Stratakis CA, Kirschner LS, Carney JA. Clinical and molecular features of the Carney complex: diagnostic criteria and recommendations for patient evaluation. J Clin Endocrinol Metab. 2001;86(9):4041-6.
- Stratakis CA. Mutations of the gene encoding the protein kinase A type I-alpha regulatory subunit (PRKAR1A) in patients with the “complex of spotty skin pigmentation, myxomas, endocrine overactivity, and schwannomas” (Carney complex). Ann N Y Acad Sci. 2002;968:3-21.
- Kirschner LS, Sandrini F, Monbo J, Lin JP, Carney JA, Stratakis CA. Genetic heterogeneity and spectrum of mutations of the PRKAR1A gene in patients with the carney complex. Hum Mol Genet. 2000;9(20):3037-46.
- Salyer WR, Page DL, Hutchins GM. The development of cardiac myxomas and papillary endocardial lesions from mural thrombus. Am Heart J. 1975;89(1):4-17.
- Salyer WR, Salyer DC. Myxoma-like features of organizing thrombi in arteries and veins. Arch Pathol. 1975;99(6):307-11.
- Prichard RW. Tumors of the heart; review of the subject and report of 150 cases. AMA Arch Pathol. 1951;51(1):98-128.
- Li Y, Pan Z, Ji Y, Sheppard M, Jeffries DJ, Archard LC, et al. Herpes simplex virus type 1 infection associated with atrial myxoma. Am J Pathol. 2003;163(6):2407-12.
- Singhal P, Luk A, Rao V, Butany J. Molecular basis of cardiac myxomas. Int J Mol Sci. 2014;15(1):1315-37.
- Orlandi A, Ciucci A, Ferlosio A, Genta R, Spagnoli LG, Gabbiani G. Cardiac myxoma cells exhibit embryonic endocardial stem cell features. J Pathol. 2006;209(2):231-9.
- Krikler DM, Rode J, Davies MJ, Woolf N, Moss E. Atrial myxoma: a tumour in search of its origins. Br Heart J. 1992;67(1):89-91.
- Terracciano LM, Mhawech P, Suess K, D'Armiento M, Lehmann FS, Jundt G, et al. Calretinin as a marker for cardiac myxoma. Diagnostic and histogenetic considerations. Am J Clin Pathol. 2000;114(5):754-9.
- Brookes SJ, Steele PA, Costa M. Calretinin immunoreactivity in cholinergic motor neurons, interneuron's and vasomotor neurons in the guinea-pig small intestine. Cell Tissue Res. 1991;263(3):471-81.
- Gosev I, Paic F, Ðuric Z, Gošev M, Ivcevic S, Bulic Jakuš F, et al. Cardiac myxoma the great imitators: Comprehensive histopathological and molecular approach. Int J Cardiol. 2013;164: 7-20.
- Barah D, Parida S. Cardiac myxoma: molecular markers, critical disease pathways, drug targets, and putative targeting miRs. Cancer Ther. 2009;7:77-96.
- Barh D, Kumar A, Chatterjee S, Liloglou T. Molecular features, markers, drug targets, and prospective targeted therapeutics in cardiac myxoma. Curr Cancer Drug Targets. 2009;9(6):705-16.
- St. John Sutton MG, Mercier LA, Giuliani ER, Lie JT. Atrial myxomas: a review of clinical experience in 40 patients. Mayo Clin Pro. 1980;55:371- 6.
- Orlandi A, Ciucci A, Ferlosio A, Pellegrino A, Chiariello L, Spagnoli LG. Increased expression and activity of matrix metalloproteinases characterize embolic cardiac myxomas. Am J Pathol. 2005;166(6):1619-28.
- Swartz MF, Lutz CJ, Chandan VS, Landas S, Fink GW. Atrial myxomas: pathologic types, tumor location, and presenting symptoms. J Card Surg. 2006;21(4): 435-40.
- Altundag MB, Ertas G, Ucer AR, Durmus S, Abanuz H, CalikoÄŸlu T, et al. Brain metastasis of cardiac myxoma: case report and review of the literature. J Neurooncol. 2005;75(2):181-4.
- Okada N, Yamamura T, Kitano Y, Nakamura T, Kamido H, Matsuzawa Y, et al. Metastasizing atrial myxoma: a case with multiple subcutaneous tumours. Br J Dermatol. 1986;115(2):239-42.
- Hirsch BE, Sehkar L, Kamerer DB. Metastatic atrial myxoma to the temporal bone: case report. Am J Otol. 1991;12(3):207-9.
- Moiyadi AV, Moiyadi AA, Sampath S, Kalpana SR, Mahadevan A, Shankar SK, et al. Intracranial metastasis from a glandular variant of atrial myxoma. Acta Neurochir (Wien). 2007;149(11):1157-62.
- Shinfeld A, Katsumata T, Westaby S. Recurrent cardiac myxoma: seeding or multifocal disease? Ann Thorac Surg. 1998;66(1):285-8.
- Gaumann A, Strubel G, Bode-Lesniewska B, Schmidtmann I, Kriegsmann J, Kirkpatrick CJ. The role of tumor vascularisation in benign and malignant cardiovascular neoplasms: a comparison of cardiac myxoma and sarcomas of the pulmonary artery. Oncol Rep. 2008;20(2):309-18.
- Sakamoto H, Sakamaki T, Kanda T, Tsuchiya Y, Sato M, Sato H, et al. Vascular endothelial growth factor is an autocrine growth factor for cardiac myxoma cells. Circ J. 2004;68(5):488-93.
- Kono T, Koide N, Hama Y, Kitahara H, Nakano H, Suzuki J, et al. Expression of vascular endothelial growth factor and angiogenesis in cardiac myxoma: a study of fifteen patients. J Thorac Cardiovasc Surg. 2000;119(1):101-7.
- Bennett KR, Gu JW, Adair TH, Heath BJ. Elevated plasma concentration of vascular endothelial growth factor in cardiac myxoma. J Thorac Cardiovasc Surg. 2001;122(1):193-4.
- Zhang T, Koide N, Wada Y, Tsukioka K, Takayama K, Kono T, et al. Significance of monocyte chemotactic protein-1 and thymidine phosphorylase in angiogenesis of human cardiac myxoma. Circ J. 2003;67(1):54-60.
- Hou YC, Chang S, Lo HM, Hsiao CH, Lin FY. Recurrent cardiac myxoma with multiple distant metastasis and malignant change. J Formos Med Assoc. 2001;100(1):63-5.
- Sakovich VA, GrinshteÄn IuI, Vershinin IV. [Malignant transformation of left atrial myxoma]. Klin Med (Mosk). 2004;82(2):65-7.
- Kasugai T, Sakurai M, Yutani C, Hirota S, Waki N, Adachi S, et al. Sequential malignant transformation of cardiac myxoma. Acta Pathol Jpn. 1990;40(9):687-92.
- Awamleh P, Alberca MT, Gamallo C, Enrech S, Sarraj A. Left atrium myxosarcoma: an exceptional cardiac malignant primary tumor. Clin Cardiol. 2007;30(6):306-8.
- Donatelli F, Pocar M, Moneta A, Mariani MA, Pelenghi S, Triggiani M, et al. Primary cardiac malignancy presenting as left atrial myxoma. Clinical and surgical considerations. Minerva Chir. 1996;51(7-8):585-8.
- Ariki H, Mizuno S, Tsunekawa T, Doi T. [Left atrial sarcoma with the initial diagnosis of myxoma]. Kyobu Geka. 2005;58(6):462-5.
- Hammond GL, Strong WW, Cohen LS, Silverman M, Garnet R, LiVolsi VA, et al. Chondrosarcoma simulating malignant atrial myxoma. J Thorac Cardiovasc Surg. 1976;72(4):575-80.
- Beghetti M, Gow RM, Haney I, Mawson J, Williams WG, Freedom RM. Pediatric primary benign cardiac tumors: a 15-year review. Am Heart J. 1997;134(6):1107-14.
- Oldershaw PJ, St john Sutton MG, Gibson RV. Long asymptomatic period of atrial myxomas. Thorax. 1980;35:70-1.
- Roudaut R, Gosse P, Dallocchio M. Rapid growth of a left atrial myxoma shown by echocardiography. Br Heart J. 1987;58(4):413-6.
- Allal J, Coisne D, Ciber M, Malin F, Christiaens L, Foullon P, et al. [Spontaneous course of myxoma of the left atrium]. Arch Mal Coeur Vaiss. 1988;81(10):1263-6.
- Vazir A, Douthwaite H. Rapidly growing left atrial myxoma: a case report. J Med Case Rep. 2011;5:417.
- Kourkoveli P, Tsiapras D, Grisbolaki E, Karapanagiotou O, Kampanarou S, Kyrzopoulos S, et al. Growth rate of an apical left ventricular myxoma using serial two dimensional echocardiographic and computed tomography observations over twelve months: a case report. J Med Case Rep. 2014;8:60.
- Türkmen N, Eren B, Fedakar R, Comunoglu N. An unusual cause of sudden death: cardiac myxoma. Adv Ther. 2007;24(3):529-32.
- Bjessmo S, Ivert T. Cardiac myxoma: 40 years' experience in 63 patients. Ann Thorac Surg. 1997;63(3):697-700.
- Whitlock R, Evans R, Lonn E, Teoh K. Giant left atrial myxoma and associated mitral valve pathology. J Cardiothorac Vasc Anesth. 2007;21(1):103-5.
- Fyke FE 3rd, Seqard JB, Edwards WD, Miller FA Jr, Reeder GS, Schattenberg TT, et al. Primary cardiac tumors: experience with 30 consecutive patients since the introduction of two-dimensional echocardiography. J Am Coll Cardiol. 1985;5:1465.
- Lee VH, Connolly HM, Brown RD. Central nervous system manifestations of cardiac myxoma. Arch Neurol. 2007;64(8):1115-20.
- Herbst M, Wattjes MP, Urbach H, Inhetvin-Hutter C, Becker D, Klockgether T, et al. Cerebral embolism from left atrial myxoma leading to cerebral and retinal aneurysms: a case report. AJNR Am J Neuroradiol. 2005;26(3):666-9.
- Alvarez-Sabín J, Lozano M, Sastre-Garriga J, Montoyo J, Murtra M, Abilleira S, et al. Transient ischemic attack: a common initial manifestation of cardiac myxomas. Eur Neurol. 2001;45(3):165-70.
- Sabolek M, Bachus-Banaschak K, Bachus R, Arnold G, Storch A. Multiple cerebral aneurysms as delayed complication of left cardiac myxoma: a case report and review. Acta Neurol Scand. 2005;111(6):345-50.
- Neff CM, McCowan CL. Complete aortic occlusion caused by cardiac myxoma emboli. Am J Emerg Med. 2008;26(1):110[e1–2].
- Coley C, Lee KR, Steiner M, Thompson CS. Complete embolization of a left atrial myxoma resulting in acute lower extremity ischemia. Tex Heart Inst J. 2005;32(2):238-40.
- Dominguez Garcia D, Arranz Duran J, Yoldi Bocanegra R, Trugeda Padilla A, Guerra Jorge MJ, Pino Capote JA. Embolism at the iliac bifurcation secondary to an atrial myxoma. Rev Esp Anestesiol Reanim. 2000;47(10):476-9.
- Demir M, Akpinar O, Acarturk E. Atrial myxoma: an unusual cause of myocardial infarction. Tex Heart Inst J. 2005;32(3):445-7.
- Luczak D, Firek B, Gepner K, Szwed H. Acute inferolateral myocardial infarction as the first manifestation of left atrial myxoma — a case report. Kardiol Pol. 2005;63(3):324-7.
- Braun S, Schrotter H, Reynen K, Schwencke C, Strasser RH. Myocardial infarction as complication of left atrial myxoma. Int J Cardiol. 2005;101(1):115-21.
- Selkane C, Amahzoune B, Chavanis N, Raisky O, Robin J, Ninet J. et al. Changing Management of Cardiac Myxoma Based on a Series of 40 Cases With Long-Term Follow-Up. Ann Thorac Surg. 2003;76:1935-8.
- Guhathakurta S, Riordan JP. Surgical treatment of right atrial myxoma. Tex Heart Inst J. 2000;27(1):61-3.
- Kirklin JW, Barratt-Boyes BG. Cardiac tumors. In Cardiac Surgery. 4th Edtn. 2013;2256.
- Mendoza CE, Rosado MF, Bernal L. The role of interleukin-6 in cases of cardiac myxoma. Clinical features, immunologic abnormalities, and a possible role in recurrence. Tex Heart Inst J. 2001;28(1):3-7.
- Yokomuro H, Yoshihara K, Watanabe Y, Shiono N, Koyama N, Takanashi Y. The variations in the immunologic features and interleukin-6 levels for the surgical treatment of cardiac myxomas. Surg Today. 2007;37(9):750-3.
- Uchino K, Mochida Y, Ebina T, Tobe M, Kobayashi S, Yano Y, et al. Infected left atrial myxoma. Intern Med. 2002;41(11):957-60.
- Falasca K, Ucciferri C, Mancino P, Di Girolamo A, Vecchiet J. Infected atrial myxoma: a rare cause of fever. Infez Med. 2008;16(1):40-2.
- Pérez de Isla L, de Castro R, Zamorano JL, Almería C, Moreno R, Moreno M, et al. Diagnosis and treatment of cardiac Myxomas by transesophageal echocardiography. Am J Cardiol. 2002;90:1419 -21.
- Araoz PA, Mulvagh SL, Tazelaar HD, Julsrud PR, Breen JF. CT and MR imaging of benign primary cardiac neoplasms with echocardiographic correlation. Radiographics. 2000;20:1303-19.
- Val-Bernal JF, Acebo E, Gomez-Roman JJ, Garijo MF. Anticipated diagnosis of left atrial myxoma following histological investigation of limb embolectomy specimens: a report of two cases. Pathol Int. 2003;53(7):489-94.
- Crafoord C. Discussion on mitral stenosis and mitral insufficiency. In: Lam CR, editor. Proceedings of the International Symposium on Cardiovascular Surgery, Henry Ford Hospital, Detroit, March 1955. Philadelphia: WB Saunders. 1955:202.
- Konecny T, Reeder G, Noseworthy PA, Konecny D, Carney JA, Asirvatham SJ. Percutaneous ablation and retrieval of a right atrial myxoma. Heart Lung Circ. 2014;23(11):e244-7.
- Ko PJ, Chang CH, Lin PJ, Chu JJ, Tsai FC, Hsueh C, et al. Video-Assisted Minimal Access in Excision of Left Atrial Myxoma. Ann Thorac Surg. 1998;66:1301-5.
- Ravikumar E, Pawar N, Gnanamuthu R, Sundar P, Cherian M, Thomas S. Minimal access approach for surgical management of cardiac tumors. Ann Thorac Surg. 2000;70(3):1077-9.
- Yu S, Xu X, Zhao B, Jin Z, Gao Z, Wang Y, et al. Totally Thoracoscopic Surgical Resection of Cardiac Myxoma in 12 Patients Ann Thorac Surg. 2010;90:674-6.
- Jones DR, Warden HE, Murray GF, Hill RC, Graeber GM, Cruzzavala JL, et al. Biatrial approach to cardiac myxomas: a 30-year clinical experience. Ann Thorac Surg. 1995;59:851-5.
- Meyns B, Vancleemput J, Flameng W, Daenen W. Surgery for cardiac myxoma. A 20-year experience with long-term follow-up. Eur J Cardiothorac Surg. 1993;7(8):437-40.
- Actis Dato GM, De Benedictis M, Actis Dato A Jr, Ricci A, Sommariva L, De Paulis R. Long-term follow-up of cardiac myxomas (7-31 years). J Cardiovasc Surg (Torino). 1993;34(2):141-3.
- Garatti A, Nano G, Canziani A, Gagliardotto P, Mossuto E, Frigiola A, et al. Surgical Excision of Cardiac Myxomas: Twenty Years Experience at a Single Institution. Ann Thorac Surg. 2012;93:825-31.
- Kabbani SS, Cooley DA. Atrial myxoma. Surgical considerations. J Thorac Cardiovasc Surg. 1973;65(5):731-7.
- Utley JR, Leyland SA, Nguyenduy T. Comparison of outcomes with three atrial incisions for mitral valve operations. Right lateral, superior septal, and trans-septal. J Thorac Cardiovasc Surg. 1995;109:582-7.
- Kumar N, Saad E, Prabhakar G, De Vol E, Duran CMG. Extended TRANS-septal versus conventional left atriotomy: early postoperative study. Ann Thorac Surg. 1995;60:426-30.
- Boutayeb A, Marmade L, Bensouda A, Moughil S. Righ atrial myxoma and severe left ventricular dysfunction: which explanations? Which management? Heart Lung Circ. 2013;22(4):309-11.
- Centofanti P, Di Rosa E, Deorsola L, Actis Dato GM, Patane F, La Torre M, et al. Primary Cardiac Tumors: Early and Late Results of Surgical Treatment in 91 Patients. Ann Thorac Surg. 1999;68:1236-41.
- Lad VS, Jain J, Agarwala S, Sinha VK, Khandekar JV, Agrawal NB, et al. Right atrial trans-septal approach for left atrial Myxomas. Nine-year experience. Heart Lung Circ. 2006;15:38-43.
- Hanson EC, Gill CC, Razavi M, Loop FD. The surgical treatment of atrial myxomas. Clinical experience and late results in 33 patients. J Thorac Cardiovasc Surg. 1985;89:298.
- Dubel HP, Borges AC, Bohm J, Gliech V, Rutsch W. Left atrial myxoma: second recurrence 20 years after the first operation. Z Kardiol. 2005;94(1):49-51.
- McCarthy PM, Piehler JM, Schaff HV, Pluth JR, Orszulak TA, Vidaillet HJ Jr, et al. The significance of multiple, recurrent, and "complex" cardiac myxomas. J Thorac Cardiovasc Surg. 1986;91(3):389-96.
- Reber D, Birnbaum DE. Recurrent cardiac myxoma: why it occurs. A case report with literature review. J. Cardiovasc Surg (Torino). 2001;42(3):345-8.
- Belhassen B, Rogowski O, Glick A, Viskin S, IIan M, Rosso R, et al. Radiofrequency ablation of accessory pathways: a 14 year experience at the Tel Aviv Medical Center in 508 patients. Isr Med Assoc J. 2007;9(4):265-70.
- Abhishek F, Heist EK, Barrett C, Danik S, Blendea D, Correnti C, et al. Effectiveness of a strategy to reduce major vascular complications from catheter ablation of atrial fibrillation. J Interv Card Electrophysiol. 2011;30(3):211-5.
- Abhishek F, Heist EK, Barrett C, Danik S, Blendea D, Correnti C, et al. Effectiveness of a strategy to reduce major vascular complications from catheter ablation of atrial fibrillation. J Interv Card Electrophysiol. 2011;30(3):211-5.
- Rubio Alvarez J, Martinez de Alegria A, Sierra Quiroga J, Adrio Nazar B, Rubio Taboada C, Martinez Comendador JM. Rapid growth of left atrial myxoma after radiofrequency ablation. Tex Heart Inst J. 2013;40(4):459-61.
- Ancona R, Comenale Pinto S, Caso P, Di Palma V, Pisacane F, Martiniello AR, et al. Right atrial mass following transcatheter radiofrequency ablation for recurrent atrial fibrillation: thrombus, endocarditis or myxoma? Monaldi Arch Chest Dis. 2009;72(1):40-2.
- Reardon MJ, Smythe WR. Cardiac neoplasms. In: Cohn LH, Edmunds LH Jr, editors. Cardiac surgery in the adult. 2nd ed. New York: McGraw-Hill. 2003:1373-400.
- Roskell DE, Biddolph SC. Proliferating cell nuclear antigen expression grossly overestimates cellular proliferation in cardiac myxomas. Eur J Med Res. 1999;4(3):105.