Research Article
Hepatoid Adenocarcinoma Presenting as a Necrotizing Soft Tissue Infection of the Abdominal Wall
Kristin Shields1*, Panna Codner,2, Christopher Dodgion2, Lewis Somberg2 and John A Weigelt2
1Department of Surgery, Western Plains Medical Complex, Dodge City, KS, USA
2Department of Surgery, Medical College of Wisconsin, Milwaukee, Wisconsin, USA
*Corresponding author: Kristin Shields, Department of Surgery, Western Plains Medical Complex. Dodge City, KS, USA
Published: 06 Jun, 2017
Cite this article as: Shields K, Codner P, Dodgion C,
Somberg L, Weigelt JA. Hepatoid
Adenocarcinoma Presenting as a
Necrotizing Soft Tissue Infection of the
Abdominal Wall. Clin Surg. 2017; 2:
1490.
Abstract
A 46 y/o presented with a necrotizing soft tissue infection of the anterior abdominal wall. She denied recent trauma. She had recently been treated for sciatica, but had not been on any long term medications. Her symptoms worsened over the last week. She had an abdominal mass with diffuse abdominal tenderness. Her initial treatment was consistent with principles for managing a complicated skin and soft tissue infection including resuscitation, broad spectrum antibiotics and excision and incision of tissues. Tissues removed included her abdominal mass. Her pathology eventually revealed a hepatoid carcinoma of unknown but likely gynecologic origin. While uncommon, an underlying malignancy is a consideration in a patient with a necrotizing soft tissue infection whose clinical presentation is not typical.
Introduction
While advanced neoplastic diseases of the abdomen occasionally present with invasion through the abdominal wall, it is less common to present with a necrotizing soft tissue infection of the abdominal wall. Perforated colon and rectal cancers are reported to present in this fashion, but never a hepatoid adenocarcinoma [1]. We present an interesting case of hepatoid adenocarcinoma (HAC) presenting as a necrotizing soft tissue infection.
Case Presentation
A 46 year old woman with a history of sciatica presented with 1 week of malaise and weakness.
She had noticed worsening of her sciatica for the past month and was being treated by a chiropractor.
Additional symptoms included leg swelling, loss of appetite, and shortness of breath with ambulation.
She appeared ill, was jaundiced, tachycardic and tachypneic. She had an abdominal wall mass,
pitting edema of the abdominal wall, and diffuses abdominal tenderness. Her hemoglobin was 4.6
g/dl, white blood cell count 42.3 × 103/μL, creatinine 1.5 mg/dL, sodium 135 mmol/L, bicarbonate
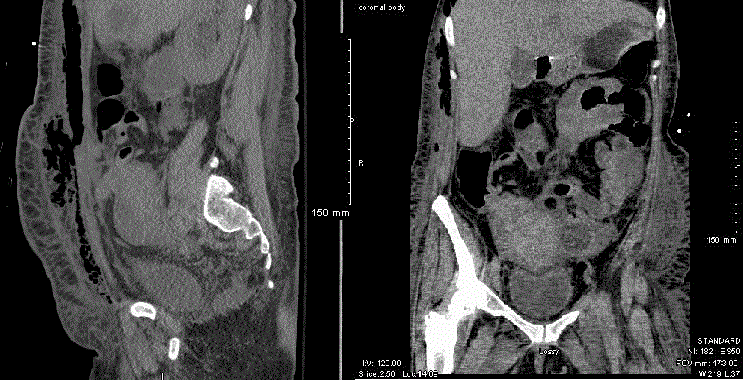
16 mmol/L, lactic acid 2.3 mmol/L and albumin 1.8g/dL. She underwent a computed tomography
(CT) scan revealing a small bowel fistula from the right lower quadrant to the anterior abdominal
wall with extensive air and fluid dissecting through the musculature and subcutaneous fat of the
abdominal wall, inguinal adenopathy, 3.9 cm left ovarian cyst, and bilateral mild hydronephrosis
(Figure 1). The initial differential diagnosis included inflammatory bowel disease and malignancy.
The patient was admitted to the surgical intensive care unit, administered broad spectrum
antibiotics, and given a blood transfusion. She was taken to the operating room where diagnostic
laparoscopy revealed mucopurulent material in the peritoneal cavity and the omentum and right
colon adherent to the abdominal wall. An abdominal incision was made to debride the anterior
abdominal wall mass during which copious purulent and dishwater-like fluid as well as necrotic
fat and muscle was noted. The omentum was adhered to the peritoneal contents and there was a
rock-hard mass in the posterior cul-de-sac. After a thorough washout, the patient’s abdomen was
temporarily closed and she continued resuscitation in the intensive care unit. The following day she
returned to the operating room for a sigmoidoscopy and extended right hemicolectomy as well as
further debridement of necrotic skin, fascia, and fat from the abdominal wall.
Intra-operative cultures grew Escherichia coli, Streptococcus anginosis, Candida albicans,
Bacteroides fragilis, and mixed anaerobic flora. Over the next week, she returned to the operating
room multiple times for washouts, an end ileostomy, mucous fistula, colonoscopy, clinical breast
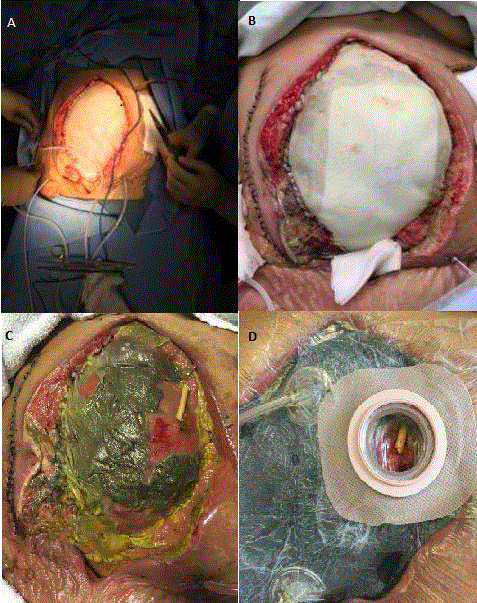
examination, partial oophorectomy and closure of the fascial defect using StratticeTM reconstructive
tissue matrix (LifeCell Corp, Branchburg, NJ). The enterocutaneous fistula was isolated from the wound using a drain. The tissue defect was covered with a negative pressure dressing (Figure 2). The right colectomy specimen had
metastatic carcinoma in the terminal ileum, appendix, and cecum
as well as 2 of 6 pericolonic lymph nodes. Extensive lymphovascular
invasion was present. The carcinoma stained positive for HEPPAR-1
and CK AE1/AE3. The cells were negative for CK7, CK20, PAX-8
(paired box gene 8), mammaglobin, ER (estrogen receptor), TTF-1,
S100, calretinin, and HBME-1 (anti-mesothelioma antibody). The
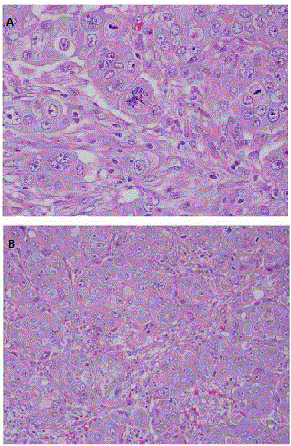
ovarian specimen revealed sheets and nests of cells with abundant
eosinophilic cytoplasm, pleomorphic nuclei with prominent
nucleoli, brisk mitotic activity which was all consistent with HCC
(hepatocellular carcinoma) (Figure 3). Additional staining revealed
SALL-4, CDX-2, and GATA-3 negativity, and CK-19 positivity.
This discounted the possibility of a yolk sac tumor and supported a
hepatoid carcinoma of unknown but likely gynecologic origin.
The patient’s serum tumor markers revealed an elevated CEA of
6.6 (normal< 3), CA-125 of 310 (normal< 35), and CA 19-9 of 35,
and a normal AFP of 7.1 (normal< 5.5). A tri-phasic contrasted CT
study did not reveal any evidence of HCC. Given the aggressiveness
of this rare tumor type, the widespread disease, and the patient’s
ECOG status of 4, no chemotherapy was recommended. The patient
transitioned to inpatient hospice and died of her disease less than one
month later.
Table 1
Figure 1
Figure 1
CT Abdomen/Pelvis reveals extensive dissection of air and fluid in
the subcutaneous tissues of the abdominal wall as well as large pelvic tumor
and a left ovarian cyst.
Discussion
Hepatoid adenocarcinoma is a rare extrahepatic neoplasm
most often described as originating in the stomach. Other sites for
this tumor include the ovary, duodenum, urinary tract, pancreas,
gallbladder, testicle, uterus, or lung. This tumor type was first
reported in 1970 by Bourreille et al. [2] and remains a diagnostic and
treatment challenge. It is more common in men, and the average age
of diagnosis is 64 years [3].
HAC morphologically displays hepatoid differentiation such as
polygonal cells in trabecular or glandular formation, bile canaliculi
or bile production, and sinusoidal formation [4]. HAC tumors
have a propensity for lymphovascular invasion [5]. It is difficult to
differentiate HAC from hepatocellular carcinoma, especially when
HAC metastasizes to the liver. HAC tumors have differential immunestaining
patterns, with multiple different patterns reported in the
literature. Table 1 displays commonly used immunohistochemical
markers for HAC versus HCC and HYST [6-13]. Typically, HAC
stains positive for AFP, mCEA, glypican-3, TTF-1, napsin A, Hep par
1, HEA-125, and MOC-31 positive. PLUNC and CDX-2 (a marker
of adenocarcinomas of intestinal origin) can be negative or positive
[14,15]. The cytokeratin profile is usually CK 7, CK 8, CK 18, and CK
19 positive, and CK 5, CK 6, CK 14, and CK 20 negative [16,17]. In
this patient, the immunostaining profile of CK-19, CK AE1/AE3 and
HEPPAR 1 positivity directed our concern to either HCC or HAC.
In this patient, there was no radiologic evidence of hepatocellular
carcinoma on CT imaging, and no evidence intraoperatively of HCC.
Given the extensive amount of pelvic tumor, the direct extension
into pelvic bone, and the worsening sciatic pain pre-dating other
symptoms, it is likely that this patient’s carcinoma started in the pelvis.
In hepatoid tumors possibly arising from gynecologic structures, it is
important to rule out hepatoid yolk sac tumor, which is a distinct
entity. While both yolk sac tumor and HAC stain positive for AFP
and glypican 3, hepatoid yolk sac tumors will also stain positive for
SALL 4. This patient’s tumor was SALL-4 negative, supporting the
diagnosis of HAC over yolk sac tumor. Hepatoid yolk sac tumors
generally occur in younger patients than HACs, with an average age
of 22 years. Patients with yolk sac tumors often also have a diagnosis
of gonadal dysgenesis [18].
The median survival of HAC is 11 months, and there is no
standardized treatment approach. The recommended treatment of
HAC of presumed gastrointestinal origin is surgical resection and
adjuvant chemotherapy- FOLFOX (leucovorin calcium, fluorouracil,
oxaliplatin) or FOLFIRI (leucovorin calcium, fluorouracil, irinotecan) with or without bevacizumab. Alternatively, capcitabine
with a platinum agent (oxaliplatin or cisplatin) has been used
for treatment [3,14]. Treatment of pulmonary HAC is resection
followed by cisplatin and gemcitabine, and sometimes followed by
localized radiation therapy [19]. Hepatoid carcinoma of the ovary is
less common, and has been treated with carboplatin and paclitaxel,
often in conjunction with surgical resection. Given the pathologic
similarity to hepatocellular carcinoma, sorafenib has been used with
variable responses [16]. Our patient had an unresectable tumor and
a poor functional status precluding any chemotherapy. Alpha fetoprotein
is usually elevated in the serum of patients with HAC and, if
initially elevated, can be used as a tumor marker to monitor treatment
response. However, not all patients display an elevated serum AFP.
Figure 2
Figure 2
A. Wound after placement of StratticeTM mesh. B. Post-operative
day #1 after mesh placement. C. After 1 week negative pressure therapy,
showing granulation atop StratticeTM mesh and staining from enterocutaneous
fistula. D. Fistula isolation from negative pressure dressing.
Figure 3
Figure 3
A: Colon specimen at 400X- polygonal cells with abundant
eosinophilic cytoplasm, mitotic activity and bizarre nuclei. B: Ovarian
specimen at 200X, again demonstrating sheets of cells with eosinophilic
cytoplasm and prominent nucleoli.
Conclusion
It is important to keep neoplastic processes as a differential diagnosis in patients with necrotizing soft tissue infections, especially when presenting with systemic evidence of malignancy. While immunohistochemistry can assist in differentiating HAC from hepatocellular carcinoma, the most convincing evidence is a lack of radiologic or surgical findings of HCC. As this case appeared to have originated in the pelvis, immunohistochemical markers were used to exclude hepatoid yolk sac tumor. HAC is a rare tumor, with its treatment is often determined by the organ system which is involved. However, a combination of surgical resection and chemotherapy has been reported to have treatment efficacy in patients presenting with early stage disease.
References
- Marron C, McArdle G, Rao M, Sinclair S, Moorehead J. Perforated carcinoma of the caecum presenting as a necrotizing fasciitis of the abdominal wall, the key to early diagnosis and management. BMC Surg. 2006;6:11.
- Bourreille J, Metayer P, Sauger F, Matray F, Fondimare A. Existence of alpha feto protein during gastric-origin secondary cancer of the liver. Presse Med. 1970;78(28):1277-8.
- Shen Z, Liu X, Lu B, Ye M. Hepatoid adenocarcinoma of the stomach: A case report of a rare type of gastric cancer. Oncol Lett. 2016;11(2):1077-80.
- Sinard J, Macleay L, Melamed J. Hepatoid adenocarcinoma in the urinary bladder. Unusual localization of a newly recognized tumor type. Cancer. 1994;73(7):1919-25.
- Osada M, Aishima S, Hirahashi M, Takizawa N, Takahashi S, Nakamura K, et al. Combination of hepatocellular markers is useful for prognostication in gastric hepatoid adenocarcinoma. Hum Pathol. 2014;45(6):1243-50.
- Su J, Chen Y, Wang R, Wu C, Lee S, Lee T. Clinicopathological characteristics in the differential diagnosis of hepatoid adenocarcinoma: a literature review. World J Gastroenterol. 2013;19(3):321-7.
- Randolph LK, Hopkins MK, Hopkins MP, Wasdahl DA. Hepatoid carcinoma of the ovary: A case report and review of the literature. Gynecol Oncol Rep. 2015;13:64-7.
- Horie Y, Kato M. Hepatoid variant of yolk sac tumor of the testis. Pathol Int. 2000;50(9):754-8.
- Devouassoux-Shisheboran M, Schammel DP, Tavassoli FA. Ovarian hepatoid yolk sac tumours: morphological, immunohistochemical and ultrastructural features. Histopathology. 1999;34(5):462-9.
- Moran CA, Suster S. Hepatoid yolk sac tumors of the mediastinum: a clinicopathologic and immunohistochemical study of four cases. Am J Surg Pathol. 1997;21(10):1210-4.
- Pandey M, Truica C. Hepatoid carcinoma of the ovary. J Clin Oncol. 2011;29(15):e446-8.
- Putra J, Ornvold K, Suriawinata A. Primary Hepatic Yolk Sac Tumor: A Case Report of a Ruptured Tumor in a 22- month- old Boy. JSM Clin Case Rep. 2014;2(1):1015.
- Chu P, Weiss L. Expression of Cytokeratin 5/6 in Epithelial Neoplasms: An Immunohistochemical Study of 509 Cases. Mod Pathol. 2002;15(1):6-10.
- Ogbonna O, Sakruti S, Sulieman M, Ali A, Shokrani B, Oneal P. Hepatoid adenocarcinoma of the duodenum: an unusual location. Case Rep Oncol, 2016;9(1):182-7.
- Reis H, Padden J, Ahrens M, Putter C, Bertmas S, Pott LL, et al. Differential proteomic and tissue expression analyses identify valuable diagnostic biomarkers of hepatocellular differentiation and hepatoid adenocarcinomas. Pathology. 2015;47(6):543-50.
- Mazouz A, Amaadour L, Ameurtesse H, Chban L, Amarti A, Kettani F, et al. Primary hepatoid carcinoma of the ovary: a case report. Pan Afr Med J. 2015;20:93.
- Gavranic T, Park Y. A novel approach using sorafenib in alpha fetoprotein-producing hepatoid adenocarcinoma of the lung. J Natl Compr Canc Netw. 2015;13(4):387-91.
- Ulbright T. Gonadoblastoma and hepatoid and endometrioid-like yolk sac tumor: an update. Int J Gynecol Pathol. 2014;33(4):365-73.
- Haninger D, Kloecker G, Bousamra L, Nowacki M, Sione S. Hepatoid adenocarcinoma of the lung: report of five cases and review of the literature. Mod Pathol. 2014;27(4):535-42.