Review Article
Pediatric Hydrocele: A Comprehensive Review
Natasha Fourie and Behrouz Banieghbal*
Department of Paediatric Surgery, Stellenbosch University, South Africa
*Corresponding author: Behrouz Banieghbal, Stellenbosch University, Tygerberg Children’s Hospital, PO Box 241 Cape Town 8000, South Africa
Published: 28 Apr, 2017
Cite this article as: Fourie N, Banieghbal B. Pediatric
Hydrocele: A Comprehensive Review.
Clin Surg. 2017; 2: 1448.
Abstract
Pediatric hydrocele is a benign common condition seen by surgeons. It is almost always occur
in males although a female equivalent is described. A hydrocele is a collection of fluid within the
processus vaginalis (PV) that produces swelling in the inguinal region or scrotum. An inguinal hernia
occurs when abdominal organs protrude a large PV, into the inguinal canal or scrotum. Inguinal
hernia and hydrocele share a similar etiology and pathophysiology and may coexist. In a healthy
male neonate, the testicle is surrounded by a closed cavity; the tunica vaginalis of the scrotum. In
postnatal life, this is a potential space that should not communicate with the peritoneal cavity of
the abdomen. However up to 60% of neonates have hydroceles. The natural history of PV is that of
spontaneous closure due to poorly understood reasons. After 2 years of age, only 0.8% of males have
a clinically palpable hydrocele and surgery is recommended for this group. The surgical techniques
involve PV resection or closure at internal ring via open surgery or laparoscopically, whichever
techniques used; the outcome is highly successful and minimal complications are reported.
Keywords: Pediatric hydrocele; Inguinal hernia
Introduction
History
The description of the abdominal cavity and the tunica vaginalis is attributed to Galen in 176 AD
[1]. However, a clear description of the inguinal anatomy and its relationship to groin hernias and
hydroceles was not recorded until the 19th century. The first surgically treated series was published
in 1934 by a German surgeon [2]. However, earlier successful surgical treatments similar to modern
surgery were noted as early as 1915 [3].
Pathogenesis
Hydrocele is defined as an accumulation of serous fluid in a body sac, normally in the scrotum.
In an attempt to understand the pathophysiology of pediatric hydroceles, it is necessary to first
clarify the normal embryology of testicular descent. During fetal development, the testicle is formed
after the migration of Y-containing germ cells from the yolk sac onto the gonadal ridge at 6 weeks
of gestation. Gonadal ridge is a mesenchymal structure that is located medial to the mesonephros.
Subsequently and during the rest of fetal development, the testicle descends through the
posterior abdominal wall and through the inguinal canal by the shortening of a cordlike structure
(gubernaculum). The exact mechanism of this regression is not fully understood and is probably due
to a complex local hormonal combination produced by the testicle. There are several androgenic
hormones implicated in testicular descent; notably testosterone, which act directly by shorting and
finally regressing of Gubernaculum, the descent of testis is however far more complex than that,
and it is beyond the scope of the mini-review [4,5]. As part of its descent through the inguinal canal,
an opening appears at the internal ring by a saclike extension of the peritoneum, the Processus
Vaginalis (PV). There is no information available on the mechanism for the appearance of this
extension.
After the testicle descends, the PV obliterates and either disappears or becomes a fibrous cord
without a lumen. The distal tip of the PV remains as a membrane around the testicle, which is
commonly referred to as the tunica vaginalis.
This obliteration of PV disconnects the inguinal region and scrotum from the abdomen, and
therefore, no abdominal organs or peritoneal fluid can pass into the scrotum or inguinal canal. If
the PV does not close, it is referred to as a Patent Processus Vaginalis (PPV). If the PPV is small in
caliber and only large enough to allow fluid to pass, the condition is referred to as a communicating
hydrocele. If the PPV is larger, allowing ovary, intestine, omentum, or other abdominal contents to
protrude, the condition is referred to as a hernia.
A number of theories have been postulated concerning the
failure of PV closure at birth. The most likely pathogenesis is the
role of smooth muscle within the PV and its subsequent contraction,
resulting in the closure of the PV. Smooth muscle of unknown
origin has been identified in PPV tissue in both males and females
(Figure 1), including adult patients, but it is not present in the normal
peritoneum. Paradoxically, higher amounts of smooth muscle have
been noted in inguinal hernia sacs than in the PPV of children with
hydroceles. This suggests that smooth muscle keeps the PPV open
rather than closed. Further research is needed to explain these
contradictory findings [6-9].
Familial inheritance is a well-known entity in which approximately
8% of parents reported having a groin operation as a child, more
likely for an inguinal hernia rather than a hydrocele [10]. Few people
remember the age of surgery, and hence, no clear data are available.
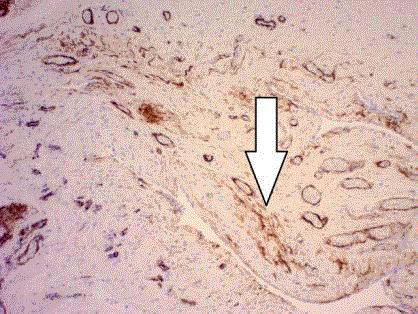
Figure 1
Figure 1
Caldesmon immunohistochemical confirmations of smooth muscle
remnant (white arrow) in an excised hernial sac (Courtesy of Dr. P. Schubert,
Pathologist) X100 magnification.
Figure 2
Figure 3
Differential Diagnosis
A hydrocele, based on its cause, can be categorized as
primary or secondary
A primary hydrocele is due to the failure of the PV to close and
is by far the most common pathology seen by pediatric surgeons. A
primary hydrocele is the result of a poorly understood abnormality
in PV closure.
In some cases, PV is closed at the time of surgery and simple
drainage of a large fluid collection is all that is needed.
A secondary hydrocele in children is often due breakdown of
hematoma after groin surgery, mistaken ligation and excision of PV
during routine surgery. Secondary hydrocele can have a different
etiology in the adult population. Its pathology is more complicated,
and there are exhaustive descriptive pathologies. Careful clinical
examination is sufficient in most cases to ascertain as to the primary
or secondary nature of the hydrocele; however, in equivocal cases, an
ultrasound is often required as an aid to diagnosis.
In decreasing order of incidence in children
Inflammatory conditions: Otherwise known as epididymitis/
orchitis, the former is associated with descending infection from the
urinary tract via the vas deferens as well as occasional case reports
in patients with ano-rectal malformation. If there is any doubt, an
urgent ultrasound may help with the diagnosis. In most cases, broadspectrum
antibiotics are prescribed for 7-10 days.
Torsion of appendix testis: One of the remnants of the
Mullerian ducts in males persists as an appendage on the upper pole
of the testes. For unknown reasons, this tissue can undergo torsion,
causing significant pain. Despite ultrasonic confirmation, parents are
sufficiently concerned about testicular torsion and the severity of pain
that they request scrotal exploration and excision of the pathology.
Omental plug at the internal ring: In this pathology, the PPV
is large, but an omental plug obstructs the entrance to the PV. This
pathology is essentially an inguinal hernia. The treatment is to repair
the inguinal hernia after releasing the omentum, which is partially
attached to the PV.
Testicular torsion: This is a complex rare pathology noted in
teenagers where the testis twists on its axis, causing ischemia. Urgent
de-torsion and pexy of both side are mandatory.
Tumors: There are numerous primary and metastatic tumors
(occasionally called a sanctuary location) that can result in minimal
fluid collection. Clinically, these conditions are readily distinguishable
from a primary hydrocele. Treatment is by trans-inguinal resection in
most cases.
Parasitic infection: The most quoted parasitic infection in the
hemi-scrotum is due to Wuchereria bancrofti round worms [11]
although Echinococcosis infections are also described [12]. Clinically,
they can be difficult to diagnose preoperatively, and ultrasonography
is necessary to identify testicular pathology in equivocal cases. They
are managed by medical, supportive bandaging and rarely surgery.
Ascites: Increased pressure from excess fluid of the intraabdominal
cavity could result in a large hydrocele that was not present
prior to the disease process. The common cause is ascites due to renal
or hepatic failure as well as a newly placed ventricular peritoneal
shunt. Management is directed towards the cause of ascites.
Post-varicocele fluid collection: This condition is due to
lymphatic fluid collection after surgical ligation of spermatic vessels
for varicocele. Lymphatic sparing ligation is advocated to avoid this
complication. There is no consensus as to the best modality for its
definitive treatment.
Figure 4
Figure 5
Figure 6
Incidence
There are no demographic or racial differences. It is not easy to
ascertain an exact hydrocele incidence because of underreporting by
many parents or children. Scrotal fluid collection is present in 60% to
80% of males at birth but declines to under 0.8% over 2 years of age.
The overall male to female ratio is calculated at 500:1 [13].
Clinical presentation of pediatric hydrocele
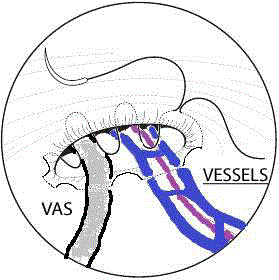
A hydrocele in males is a collection of fluid in a potential space
between the tunica vaginalis and the testicle. Hydroceles may be
considered communicating due to a small-caliber PPV that allows
only fluid to pass into the scrotum. If there is no connection between
the hydrocele and the abdominal cavity, the condition is called a
non-communicating hydrocele. Non-communicating hydroceles
are common in infants, with an incidence close to 60% in newborns.
The non-communicating hydrocele usually spontaneously reabsorbs
before the age of 2 years. In most children with a congenital hydrocele,
there is a long history of an asymptomatic, painless, soft fullness in
the hemi-scrotum that is usually noticed by the caregiver or during
routine school physical screenings. Pain is not a symptom, and if
present, the possibility of an incarcerated or strangulated inguinal
hernia must be considered.
Communicating hydroceles vary in size during a 24 h period, with
an increase in volume during daytime activity. If communicating, a
large hemi-scrotal fullness may extend into the inguinal area and can
be associated with discomfort during physical activity.
Occasionally, a hydrocele can extend from the scrotum through
the inguinal canal into the retro-peritoneum as an abdomino-scrotal
hydrocele. This is due to a minute “trap-door” opening in the PPV;
fluid that enters the hydrocele becomes entrapped, and the hydrocele
continues to enlarge upward toward the abdomen. This condition
can be mistaken for an indirect inguinal hernia during clinical
examination and is often operated on.
When a small communication with the peritoneum persists and
the PPV obliterates further distally, a hydrocele of the cord can form.
It presents as a tense, fluid-containing, painless, round, mobile mass.
It is palpable in the inguinal canal or upper scrotum and usually
resorbs by 1-2 years of age.
An acute presentation of a hydrocele may be secondary to an
underlying disease process inside the tunica vaginalis or testis.
In many patients, there is an acute respiratory tract infection or
gastroenteritis associated with significant scrotal pain due to sudden
distension of a small hydrocele. In this unusual circumstance,
increased intra-abdominal pressure during coughing or straining
forces large amounts of fluid through a small-caliber PPV.
The majority of premature hydroceles resolve with time, and
therefore, surgery is only considered if the hydrocele is present at the
age of 2 years and is not decreasing further in size [2].
Physical examination is normally sufficient to distinguish
a hydrocele from an inguinal hernia. If the clinician is able to feel
the spermatic cord above the mass, a hydrocele can be confidently
diagnosed. This may be difficult to appreciate in the presence of a
tense inguino-scrotal hydrocele. An additional feature in the clinical
findings of a hydrocele includes the ability to trans-illuminate. This
does not fully exclude an inguinal hernia, since an incarcerated
inguinal hernia in premature infants can also trans-illuminate [14].
Imaging studies
Most cases of pediatric hydroceles can be diagnosed with a good
history and adequate physical examination alone. A minority of
patients require further radiological investigation to aid in diagnosis.
Historically, contrast herniography was performed, but this has been
replaced by ultrasonography.
Herniography: This procedure is of historical interest only. Water
soluble contrast is injected into the peritoneal cavity via an infraumbilical
fluoroscopic-guided injection. With gravity, the contrast
accumulates into the PPV, which is identified by serial plain X-rays.
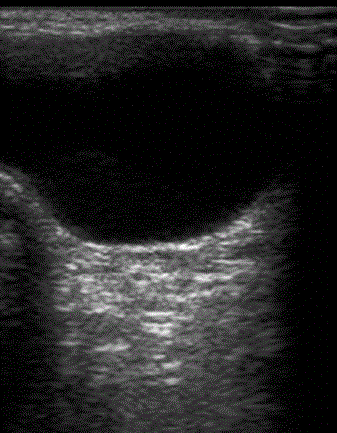
Ultrasonography, using a 7.5 MHz transducer, is the current
modality of choice as an aid for diagnosis, with a reported accuracy of
91.7% [15], compared to herniography, this modality has the distinct
advantage of being noninvasive (Figure 2). Indications for usage of
ultrasonography include: trauma with possible testicular rupture,
torsion of a testicle or ovary, concern of a testicular or spermatic cord
tumor, or equivocal physical findings. Ultrasound performed with the
patient at rest and straining: standing, coughing or crying. Detection
of the inflow of peritoneal fluid in the inguinal canal is diagnostic for
a hydrocele with ultrasound examination.
Surgical techniques
Unlike in adults, sclerotherapy has no place in the definitive
management of pediatric hydroceles. Standard open surgical
management has been the gold standard for the definitive treatment of
hydroceles, and the procedure is identical to an inguinal herniotomy.
The main difference is that the PPV is small in caliber. Excision of
a section of PV is thought to be as effective as ligation, with reports
in other series that have shown low or no recurrence with nonligation
of the sac in inguinal hernia [16,17]. The safety and efficacy
of laparoscopic repair for both an inguinal hernia and a hydrocele in
children are demonstrated to be similar to open procedures without
any minor or major complications. Several known advantages of
the laparoscopic approach are that it is a less painful approach for
patients, patients return to their normal activity more rapidly, and it
provides superior cosmetic results.
All techniques are performed under general anesthesia, the
child’s abdomen, groin, and scrotum are cleaned and draped. The
patient is placed in the supine position. Thereafter, there are at least
four surgical options available.
Open inguinal herniotomy for pediatric hydrocele: High
ligation of the PPV with or without drainage of the scrotal hydrocele
is the standard method of open repair for a hydrocele performed via
an inguinal incision [2].
In older children, the internal inguinal ring is more lateral,
increasing the distance between the two rings. The external ring lies
superior and lateral to the pubic tubercle.
A transverse skin incision is made in the groin, above the
external inguinal ring on the symptomatic side, within a skin crease.
Dermis and the subcutaneous tissues are spread bluntly, exposing
Scarpa’s fascia. The fascia is incised to expose the external oblique
fascia mediolaterally. The inguinal ligament is exposed laterally and
medially up to the level of the external ring. The external oblique
fascia, 1 cm to 2 cm above the inguinal ligament, is opened superiorly
and laterally near the external inguinal ring in the direction of the
muscle fibers. Two clamps are placed on both cut edges of the fascia
(Figure 3). The spermatic cord containing PPV is mobilized through
the opening of the fascia and the cord structures are elevated out of
the wound. Fibers attached to the hernia sac are released until a clear
space inferior to the cord is created and artery forceps are placed
underneath this space. The spermatic fascia is bluntly dissected the,
vas deferens and vessels are dissected away from the sac. A clamp is
placed across the vas deferens and vessels, and the proximal end of the
hernia sac is freed of cord structures to the level of the internal ring
and ligated with monofilament absorbable sutures. A small window
is cut into the hydrocele and fluid drained from the distal sac and /or
scrotum with digital pressure. The testicle is pulled down, and cord
structures returned to the scrotum. The two clamps are elevated on
the edges of the external oblique fascia, and closed with interrupted
absorbable sutures. Surgical wound is closed in layers; Scarpa’s fascia
is closed with a single interrupted absorbable suture, skin with an
absorbable subcuticular continuous suture and a wound dressing is
applied.
There are at least 3 laparoscopic approaches, practiced mostly in
academic teaching centers. The simplicity of the operation and the
fine dissection required to separate the cord vessels and vas deferens
make laparoscopic PPV excision an ideal procedure for pediatric
surgical trainees in minimally invasive surgery. During laparoscopy,
the contralateral inguinal ring can be inspected for patency. These
approaches are gaining popularity with pediatric surgeons with
cheaper ports and instrumentations; increased cost is no longer
an important debate. These techniques are particularly useful for
recurrent cases where a scarred groin is avoided by attending to open
PV trans-abdominally.
The laparoscopic approaches to pediatric hydrocele include:
ligation or excision of a section of PPV and extra- or intra-peritoneal
repair.
Intra-peritoneal PPV excision with non-ligation: The surgeon
and assistant stand at the head of the operating table with the screen/
monitor at the foot of the table.
A 5 mm supra or infra-umbilical camera port is inserted via
the open Hasson approach. Pneumoperitoneum is achieved via
insufflation of CO2 at pressures of 8 mmHg to 10 mmHg. PPV, vas
deferens, and testicular vessels are identified. The contralateral side
is evaluated for a concurrent hydrocele, which is found in 10% of
cases and can be repaired at the same time. Under direct vision, two
other 3 mm instruments are placed, one in the right lower quadrant
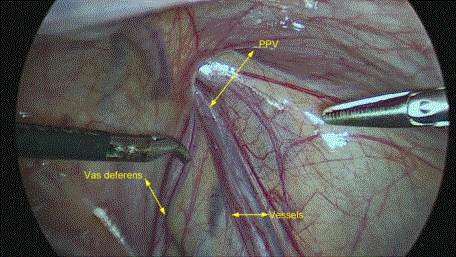
and left lower quadrant at 90 degree working angles (Figure 4). In
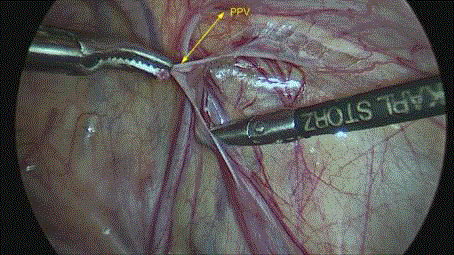
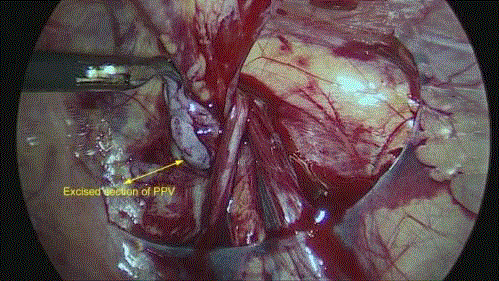
one approach, favored by the authors, a section of PPV is excised
without its ligation (Figures 5-8). The pneumoperitoneum is released,
the umbilical incision is closed with an absorbable suture, and the
wounds of the two working sites are closed with stri-strips. There is
no report of recurrence with this technique in 75 cases over a 5-year
follow-up (accepted manuscript, awaiting publication).
Intra-peritoneal purse string closure pf PPV: Here, a subperitoneal
saline injection is used to separate the vas deferens and
testicular vessels from the peritoneum. The peritoneum overlying
the testicular vessels and vas deferens is grasped, and 2 ml to 4 ml of
saline is injected. The neck of the hernia sac is incised with a scissor
or diathermy. An absorbable suture is passed under visual control
through the abdominal wall. The purse string suture is placed around
the hernia ring and must include the whole ring of the peritoneum.
Exit and entry sites of successive bites of peritoneum should be as
close as possible (Figure 9). Intra-peritoneal pressure is decreased to
2 mmHg to 4 mmHg before tying the suture. Complete closure of the
processes vaginalis is tested by increasing intra-peritoneal pressure to
15 mmHg and palpating for crepitus; if present, another suture can be
placed. Ports are removed under direct vision, and the insufflation is
terminated. Wounds are closed as described. Recurrence rate of 1.4%
are reported with this technique [18].
Extra-peritoneal purse string closure of PPV (2 or one trocar
technique): After placement of the camera port and creation of
pneumoperitoneum, a 3 mm grasper is placed midway between the
umbilicus and the supra pubic tubercle under direct vision. A small
stab wound is made just lateral to the internal ring, and a hernia
hook containing a 3/0 non-absorbable suture is passed through the
tract into the pre-peritoneal space. The deep ring is dissected from
medial, lateral, and posterior aspects. Care is taken to avoid the vas
deferens and testicular vessels. The tip of the hook is used to pierce
the peritoneum. Halfway around the internal ring, the suture is
pulled into the peritoneum, and the hook is withdrawn and reinserted
into the antero-medial peritoneal space. The medial semicircle of
the internal ring is similarly dissected as before. The hook then reenters
the peritoneum. The suture is placed through the eye of the
hook, it is withdrawn, and the suture entirely encircles the internal
ring. Pneumoperitoneum is released, and the suture is tied extracorporally.
The complete closure of the PV can again be tested by
increasing the intra-abdominal pressure, and palpating for crepitus
as described before. A recurrence rate of about 1% has been recorded
in this approach [19-22].
Figure 7
Figure 8
Figure 9
Complication of Surgery
There are many anesthetic complications, notwithstanding the
recently described toxicity of volatile anesthetic agents in children <4
years of age [23].
Open and laparoscopic techniques can result in a myriad of
complications, which, although rare, are routinely discussed with
parents/caregivers as part of the consent process. Commonly
mentioned complications include partial to complete transection of
the vas deferens as well as spermatic cord vessel injury. Recurrence
is due to failure in recognizing and resecting the small-caliber PPV
by inexperienced surgeons. However, unlike inguinal herniotomy,
damage to other structures, such as the bladder, are never reported.
Wound infections or dehiscence are rare, and there is no literature
regarding trocar injury during laparoscopic PPV surgery.
Conclusion
The exact mechanism of the formation of hydroceles and nonclosure of PV is yet to be determined.
The definitive repair of hydroceles in children over 2 years of age
is a relatively simple, regardless of the technique used. It has a very
high success rate with minimal morbidity.
References
- Charles S. A short history of anatomy from Greeks to Harvey. 1957;46-56.
- Langer M. About the Hernia and Hydrokele of the Childhood. Arch f Klin Chir. 1934;18:418.
- Gross RE. Hydrocele. In: The Surgery of Infancy and Childhood. London: WB Saunders Publishers; 1953. p. 463-6.
- Hutson JM, Li R, Southwell BR, Newgreen D, Cousinery M. Regulation of testicular descent. Pediatr Surg Int. 2015;31(4):317-25.
- Sadler TW. Urogenital system. Langman’s Medical Embryology, 9th ed. Philadelphia: Lippincott Williams; 2004. p. 321-62
- Stickel WH, Manner M. Female hydrocele (cyst of the canal of Nuck): sonographic appearance of a rare and little-known disorder. J Ultrasound Med. 2004;23(3):429-32.
- Schneider CA, Festa S, Spillert CR, Bruce CJ, Lazaro EJ. Hydrocele of the canal of Nuck. N J Med. 1994;91(1):37-8.
- Tanyel FC, Mufutuoclu S, Dagdeviren A, Kaymas FF, Buyukpamukch N. Myofibroblast defined by electron microscopy suggest the dedifferentiation of smooth muscle within the sac walls associated with congenital hernia. BUJ international. 2001;87:251-5.
- Mouravas VK, Koletsa T, Sfougaris DK, Philippopoulos A, Petropoulos AS, Zavitsanakis A, et al. Smooth muscle cell differentiation in the processus vaginalis of children with hernia or hydrocele. Hernia. 2010;14(2):187-91.
- Burcharth J, Pommergaard HC, Rosenberg J. The inheritance of groin hernia: a systematic review. Hernia. 2013;17(2):183-9.
- Aguiar-Santos AM, Leal-Cruz M, Netto MJ, Carrera A, Lima G, Rocha A. Lymph scrotum: an unusual urological presentation of lymphatic filariasis. A case series study. Rev Inst Med Trop Sao Paulo. 2009;51(4):179-83.
- Khan RA, Wahab S, Chana RS, Fareed R. Isolated retroperitoneal hydatid cyst in a child: a rare cause of acute scrotal swelling? J Pediatr Surg. 2010;45(8):1717-9.
- Ben-Ari J, Merlob P, Mimouni F, Rosen O, Reisner SH. The prevalence of high insertion of scrotum, hydrocele and mobile testis in the newborn infant (36-42 weeks gestation). Eur J Pediatr. 1989;148(6):563-4.
- Naji H, Ingolfsson I, Isacson D, Svensson JF. Decision making in the management of hydroceles in infants and children. Eur J Pediatr. 2012;171(5):807-10.
- Hasanuzzaman SM, Chowdhury LH, Sarker RN, Bari MS, Talukder SA, Islam MK. Ultrasonographic evaluation of contralateral exploration of patent processus vaginalis in unilateral inguinal hernia. Mymensingh Med J. 2011;20(2):192-6.
- Mohta A, Jain N, Irniraya KP, Saluja SS, Sharma S, Gupta A. Non-ligation of the hernial sac during herniotomy: a prospective study. Pediatr Surg Int. 2003;19(6):451-2.
- Rafiei MH, Jazini A. Is the ligation of hernial sac necessary in herniotomy for children? A randomized controlled trial of evaluating surgical complications and duration. Adv Biomed Res. 2015;4:97.
- Yang XD, Wu Y, Xiang B, Wong K, Pei J, Li FY. Ten year experience of laparoscopic repair of pediatric hydrocele and the long-term follow-up results. J Pediatr Surg. 2015;50(11):1987-90.
- Saka R, Okuyama, Sasaki T, Nose S, Yoneyama C. Safety and efficacy of laparoscopic percutaneous extra peritoneal closure of inguinal hernias and hydroceles in children: a comparison with traditional open repair. J Laparoendosc Adv Surg Tech A. 2014;24(1):55-8.
- Wang Z, Xu L, Chen Z, Yao C, Su Z. Modified single-port minilaparoscopic extraperitoneal repair for pediatric hydrocele: a single-center experience with 279 surgeries. World J Urol. 2014;32(6):1613-8.
- Shalaby R, Ismail M, Gouda S, Yehya AA, Gamaan I, Ibrahim R, et al. Laparoscopic management of recurrent inguinal hernia in childhood. J Pediatr Surg. 2015;50(11):1903-8.
- Peng Y, Li C, Han Z, Nie X, Lin W. Modified single-port vs two-port laparoscopic herniorrhaphy for children with concealed deferent duct: a retrospective study from a single institution. Hernia. 2016.
- Lin D, Liu J, Kramberg L, Ruggiero A, Cottrell J, Kass IS. Early-life singleepisode sevoflurane exposure impairs social behavior and cognition later in life. Brain Behav. 2016;6(9):e00514.