Case Report
Dealing with the Unexpected but Chasing the Inevitable: Atrio-Ventricular Disruption Following Aortic Valve Replacement in an Elderly Patient
Avtaar Singh S1*, Capoccia M2 and Al-Attar N3
1Department of Cardiothoracic, Golden Jubilee National Hospital, UK
2Department of Transplant, Golden Jubilee National Hospital, UK
3Consultant Cardiac Surgeon, Golden Jubilee National Hospital, UK
*Corresponding author: Sanjeet Avtaar Singh, Department of Cardiothoracic, Scottish National Advanced Heart Failure Service, Golden Jubilee National Hospital, Clydebank, Glasgow, UK
Published: 14 Apr, 2017
Cite this article as: Avtaar Singh S, Capoccia M, Al-Attar
N. Dealing with the Unexpected but
Chasing the Inevitable: Atrio-Ventricular
Disruption Following Aortic Valve
Replacement in an Elderly Patient. Clin
Surg. 2017; 2: 1402.
Abstract
Atrioventricular groove ruptures is almost always lethal. We report the successful repair of a rupture of the atrioventricular groove in an elderly patient who underwent an aortic valve replacement and the sequel of complications preceding this.
Introduction
Atrio-ventricular groove rupture remains a challenging and feared event for any cardiac surgeon. The outcome is almost always fatal. Here we present the occurrence of this event following aortic valve replacement in an elderly patient.
Case Presentation
A 73 year old lady was referred to our hospital with a 12-month history of worsening shortness of breath on exertion. Three years earlier, she presented with shortness of breath and chest pain. A
thallium scan suggested a large area of reversibility and a subsequent angiogram showed 2-vessel
disease. Drug Eluting Stents were inserted into her left anterior descending and circumflex arteries.
She stopped smoking forty years earlier and her past medical history included hyperlipidaemia and
hypertension. Her past surgical history included a Right sided mastectomy for breast cancer in 2001,
alongside adjuvant radiotherapy. She also underwent a trabeculectomy for glaucoma in 2004.
On examination, she had a short stature of 4 ft 11”. She had a slow rising pulse with an ejection
systolic murmur radiating to her carotids. Echocardiography revealed good LV systolic function
and a peak gradient across the aortic valve of 75 mmHg with a mean of 41 mmHg. The aortic valve
surface area was calculated at 0.7 cm2. She also has mild MR and a systolic pulmonary artery pressure
of 30 mmHg. The decision was made for her to undergo a bio-prosthetic aortic valve replacement.
During surgery, her native aortic valve was tricuspid with 3 heavily calcified cusps which
were excised and the annulus was cleaned from debris. The peri-operative TOE showed extensive
calcifications of the mitral annulus confirming the pre-operative findings on the angiogram. While
weaning from bypass significant bleeding was noted from the posterior aspect of the heart raising
suspicion of a spontaneous atrio-ventricular groove rupture, which was confirmed on subsequent
inspection. Cardiopulmonary bypass was reinstituted and the breach repaired using a pericardial
patch. Complete haemostasis was achieved and cardiopulmonary bypass discontinued uneventfully.
The early postoperative course was relatively uneventful. The following morning weaning from
the ventilator was commenced but sudden collapse with no cardiac output required resuscitation
manoeuvres and emergency chest reopening. Findings were consistent with significant bleeding
from further myocardial rupture on the left ventricular side. The defect was repaired without
cardiopulmonary bypass using two 4/0 Prolene pledgeted sutures; in addition, the whole anterior
and lateral ventricular walls were re-enforced with 2 patches of pericardium fixed to the surface
with Bio Glue achieving complete haemostasis. The insertion of an intra-aortic balloon pump was
deemed necessary to supplement the already significant inotropic support.
She remained critical requiring prolonged ventilation with several failed weaning attempts.
Her poor neurological status and the development of abdominal distension led to further imaging
investigations, which confirmed a likely infarct in the high right occipital/occipito-parietal region and significant bowel ischaemia. General surgical opinion was sought and a laparotomy performed: small bowel resection was required with
demarcation levels just beyond the duodeno-jejunal flexure around
and 18 inches proximal to the terminal ileum. Post resection, there
remained approximately 6cm of proximal duodenum just distally
to the DJ flexure and around 35 cm of terminal ileum, which were
both tied off with a view to re-explore the abdomen in 48 h prior
to anastomosis. During re-exploration a further 5cm of terminal
Ileum was resected and an end to end anastomosis was carried out as
planned (Figure 1).
Following extubation, she had a slow recovery requiring TPN and
pharmacological treatment for onset of atrial flutter with subsequent
DC cardio version in the absence of atrial clots on TOE assessment.
Finally, she was discharged to her local hospital for continuity of care.
She was readmitted to our hospital 2 months later because
of increasing shortness of breath. A CT Pulmonary Angiogram
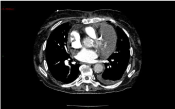
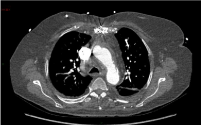
had already revealed a large (\R\5 cm) left pericardial haematoma
compressing the origin of the RVOT/pulmonary trunk. A subsequent
Gated CT of the Aorta showed a complex pseudo-aneurysm
measuring up to 3 cm in diameter, which appeared to communicate
with the distal left main stem with most of its blood supply from a
wide neck (2.5 mm) fistulous connection with the aortic isthmus. A
large pericardial haematoma measuring at least 9.5 cm in maximal
diameter with significant external compression of the right ventricular
outflow tract was confirmed (Figure 2).
Following MDT discussion, it was agreed that further surgical
intervention would not be a suitable option. Instead, a trans-catheter
closure would be attempted (Figure 3).
The procedure was challenging but eventually a device was
successfully deployed to close the defect.
A repeat Aorta gated CT performed 2 days post procedure showed the closure device within the mitro-aortic curtain but the
pseudo-aneurysm of the left main stem remained unchanged with
an increasing haematoma (84 × 55 × 37 mm) compressing the
pulmonary trunk.
A week later, she developed features suggestive of tamponade
confirmed on echocardiographic assessment with haemodynamic
compromise. A pericardiocentesis was attempted but abandoned
shortly after. Further deterioration and ongoing sepsis led to her
death after five months following her aortic valve replacement.
Figure 1
Figure 2
Figure 3
Discussion
Atrio-ventricular rupture is usually witnessed following mitral
replacement in the presence of a heavily calcified annulus [1,2]
although it may occur in other less common scenarios such as aortic
valve replacement, excessive cardiac manipulation or trauma [3-5].
The outcome is almost always fatal. It is not surprising that it has
been widely recognised as one of the worst nightmares for a cardiac
surgeon regardless the level of experience.
It is always tempting to show off by discussing successful outcomes
or reporting clinical cases where we have made something to be proud
of. On this occasion, we sought to share a not so successful clinical
case in order to stress how easy it can be to become overconfident
after an initial successful repair and then having to reconsider your
boundaries and accept defeat.
Despite unnecessary manipulation of the heart during the
procedure, the inevitable did happen. Distortion of the mitral annulus
already compromised by the presence of significant calcifications may
have certainly played a role. Other likely contributing factors include
cardiomegaly, previous myocardial infarction and advanced age. The
atrioventricular groove remains a naturally weakened transitional
area in view of its embriological features where the conduction
tissue is the only muscular continuity between atrial and ventricular
chambers [6-8]. The early postoperative event requiring further
repair following emergency reopening in intensive care proved to be
untimely complication. The initial successful combination of suturing
and surgical sealants only delayed the point of no return confirming
the challenge and demand related to such cases.
References
- Kwon JT, Jung TE, Lee DH. The rupture of atrioventricular groove after mitral valve replacement in an elderly patient. J Cardiothorac Surg. 2014;9:28.
- Lee ME, Tamboli M, Lee AW. Use of a sandwich technique to repair a left ventricular rupture after mitral valve replacement. Tex Heart Inst J. 2014;41(2):195-7.
- Lawton JS, Deshpande SP, Zanaboni PB, Damiano RJ. Spontaneous Atrioventricular Groove Disruption during Off-Pump Coronary Artery Bypass Grafting. Ann Thorac Surg 2005;79:339-41.
- Nakamura T, Izutani H, Shibukawa T, Higuchi T. Aortoventricular disruption after aortic valve replacement: a rare complication. Interact Cardio Vasc Thorac Surg. 2010;11:447-8.
- Slater AD, Subramanian S, Huang J, Bouvette M, Pagni S, Dowling RD. Repair of mitral valve and left atrioventricular disruption caused by blunt chest trauma. Ann Thorac Surg. 2009;87(4):1289-90.
- El-Essawi A, Biancosino C, Anssar M, Kutschka I, Breitenbach I, Harringer W. Atrioventricular disruption managed by ex-situ repair and autotransplantation. J Heart Valve Dis. 2007;16(4):359-61.
- Kalangos A, Jornod N, Rognon R, Faidutti B. Successful repair of a right ventricular rupture at the atrioventricular groove. Ann Thorac Surg. 1996;61(3):995-7.
- Wessels A, Markman MW, Vermeulen JL, Anderson RH, Moorman AF, Lamers WH. The development of the atrioventricular junction in the human heart. Circ Res. 1996;78(1):110-7.