Research Article
The Association between Microsatellite Alteration and Survival of Oral Cavity Squamous Cell Carcinoma Patients from an Endemic Betel Quid Chewing Area
Shih-An Liu1,3,5, Chen-Chi Wang1,3, Rong-San Jiang1, Wen-Yi Wang4 and Jin-Ching Lin2,3*
1Department of Otolaryngology, Taichung Veterans General Hospital, Taiwan
2Department of Radiation Oncology, Taichung Veterans General Hospital, Taichung, Taiwan
3Faculty of Medicine, School of Medicine, National Yang-Ming University, Taiwan
4Department of Nursing, Hung Kuang University, Taiwan
5Department of Medical Research, China Medical University, Taiwan
*Corresponding author: Jin-Ching Lin, Department of Radiation Oncology, Taichung Veterans General Hospital, Taichung, No. 1650, Sec 4, Taiwan Boulevard, Taichung, Taiwan, R.O.C
Published: 14 Feb, 2017
Cite this article as: Liu S-A, Wang C-C, Jiang R-S, Wang
W-Y, Lin J-C. The Association between
Microsatellite Alteration and Survival of
Oral Cavity Squamous Cell Carcinoma
Patients from an Endemic Betel Quid
Chewing Area. Clin Surg. 2017; 2: 1311.
Abstract
Background: The incidence of microsatellite alterations at the endemic betel quid chewing area and
its association with the survival of patients with oral cavity squamous cell carcinoma (OCSCC) is
not clear. Here we studied their possible relationship.
Methods: Subjects were 135 patients with histological confirmed OCSCC. From these patients we
obtained their cancerous tissues, corresponding surgical margins, and peripheral blood samples.
From these specimens, we analyzed the microsatellite alterations base on 10 oligonucleotide markers.
Specifically, specimens were assessed by automatic fragment analysis following amplification by
polymerase chain reactions.
Results: Of these specimens, 45 (33.3%) showed microsatellite instability (MSI) and 78 (57.8%)
showed loss of heterozygosity (LOH) for at least one marker. Using Kaplan-Meier’s analysis method,
microsatellite alterations of patients did not associate with their disease-specific survival. However,
the presence of MSI in surgical margins of the cancer increased the risk of local recurrence (odds
ratio: 7.49; 95% confidence interval: 3.34 ~ 16.80; P < 0.001).
Conclusion: The prognosis of OCSCC patients was not associated with microsatellite alterations in
region where betel quid chewing is prevalent. However, genomic examination of surgical margin
can possibly find out OCSCC patients who are prone to develop local recurrence.
Introduction
Microsatellite instability (MSI) and loss of heterozygosity (LOH) are the most common types of microsatellite alterations which have been reported to be associated with various type of cancer in the literature. Microsatellites are repeating segments of 1 to 6 base pairs in eukaryotic genomes [1]. Such repeated sequences are susceptible to inaccurate repetition during DNA duplication, and the failure to repair such errors leads to MSI [2]. For example, MSI is well-known to be related to the development and prognosis of colorectal cancer [3]. In addition, head and neck cancer patients with MSI are more likely to develop a second primary cancer [4]. Our previous study also showed that MSI in the dysplasia-free surgical margin of head and neck squamous cell carcinoma is associated with its local recurrence [2]. However, the relationship between survival of OCSCC patients and MSI remains controversial [5]. The loss of a functional allele at a heterozygous locus, or LOH, is correlated with allelic loss of a number of tumor suppressor genes [6]. A tumor suppressor gene adjacent to LOH can be deactivated, leading to uncontrolled cell growth [7]. A previous study found that LOH at D9S162 is associated with a poor recurrence-free survival in oral cancer patients [8]. Lee et al. [9] in a study on hypopharyngeal cancer patients also found that LOH is correlated with lymph node metastasis. However, most of abovementioned studies are in Western countries and little is known in Asian countries regarding MSI and LOH on the survival of OCSCC patients. Here we investigated microsatellite alterations in a betel quid-prevalent region and its association with the survival of OCSCC patients.
Table 1
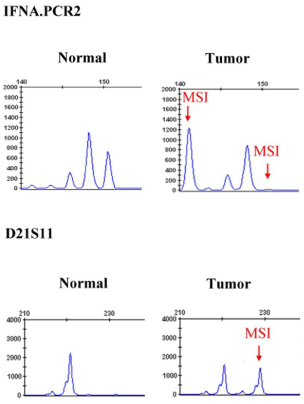
Figure 1
Figure 1
Representative sample of microsatellite instability (MSI) in selected
microsatellite markers.
Figure 2
Materials and Methods
This study was reviewed and approved by the Institutional
Review Board of the host Hospital. Potential participants were oral
cavity cancer patients planned for surgical intervention during a
four-year from April 2012 to April 2016. The detailed protocol was
first explained to participants and written consent obtained prior
to subject recruitment. We excluded those subjects who either had
declined for surgery, non-squamous cell carcinoma, incomplete
medical records, or refused to participate the study. Pathological
stage was determined according to the guidelines of the American
Joint Committee on Cancer (7th edition, 2009). Habits of participants
on cigarette smoking and betel quid chewing were quantitatively
recorded as follows. “One pack-year” represented smoking 20
cigarettes (1 pack) per day for 1 year and “one quid-year” represented
chewing one betel quid per day for 1 year. Due to various kinds of
alcoholic beverages were consumed by participants, we only divided
participants into three groups: non-user, social user, and heavy user.
Treatment plans for all participants were conducted in accordance
with the consensus guidelines of the oral cavity cancer team of the
host Hospital.
Detailed laboratory procedures were same as those in our
previous report [2]. In brief, histologically confirmed OCSCC
specimens and corresponding surgical margins were promptly
stored in liquid nitrogen. Peripheral blood (10 ml) was drawn before
operation and was placed in an EDTA-treated tube. The sample
was then centrifuged and the plasma was transferred to a 1.5-ml
microtube. The mononuclear cell layer was transferred into a clean
50 ml centrifuge tube, washed twice with a balanced salt solution, and
re-centrifuged. Samples were stored at –30°C until use. Total DNA
was extracted using the QIAamp DNA Mini kit (QIAGEN) according
to its instructions. The final DNA was dissolved in double-distilled
water and frozen at -30°C until further processing. Five binucleotide
microsatellites (D9S1748, D3S1079, THRB, D3S1234, D3S1300)
were selected based on literature review [10-12]. Three additional
binucleotide microsatellites (IFNA.PCR2, D2S206, D21S236) and
two tetranucleotide microsatellites (D21S1433, D21S11) were
also selected according to our previous work (Table 1). Multiplex
PCR reactions were performed with fluorescent-labeled forward
primers and the amplified PCR products were analyzed through
capillary array electrophoresis with the software Gene Scan (Applied
Biosystems Inc., Foster City, USA). All PCR products were purified
and sequenced with the ABI Big Dye Terminator (version 3.1) cycle
sequencing ready reaction kit and the ABI PRISM 3100 sequencer
(Applied Biosystems Inc., Foster City, CA). MSI was defined as the
presence of novel sized fragments in DNA obtained from tumor
subjects. These fragments were absent in the DNA of leukocytes taken
from peripheral blood (Figure 1). In addition, to provide a tumor
imbalance factor [11], the ratio of both microsatellite alleles (allele
2/allele 1) in the peripheral blood leukocyte DNA was divided by
the corresponding ratio found in tumor DNA (Figure 2). Imbalance
factor of values < 0.67 or > 1.5 were classified as LOH.
Statistical analyses
We used descriptive statistics to present the demographic data.
Student’s t test was used to compare continuous variables between
subgroups. Nominal or ordinal variables were analyzed using the
Chi-square test or Fisher’s exact test. The Kaplan-Meier method
was used to calculate disease-specific survival. Differences among
subgroups were assessed by the log-rank test. A backward stepwise
logistic regression model was used to find independent factors
correlated with local recurrence. All analyses were conducted in SPSS
for Windows, version 12.1 (SPSS, Chicago, IL) and a p< 0.05 was
considered statistically significant.
Table 2
Table 2
Descriptive and bivariate analysis of oral cavity squamous cell carcinoma patients with or without microsatellite instability (MSI).
Table 3
Table 3
Descriptive and bivariate analysis of oral cavity squamous cell carcinoma patients with or without loss of heterozygosity (LOH).
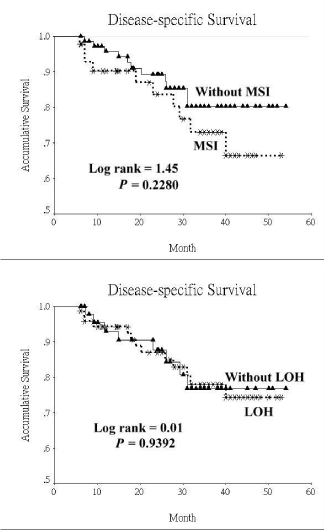
Figure 3
Figure 3
Disease-specific survival curves of oral cavity squamous cell
carcinoma patients based on the status of microsatellite alteration. (MSI:
microsatellite instability; LOH: loss of heterozygosity).
Table 4
Results
There were totally 171 oral cavity cancer patients planned to
receive surgical excision during the study period. Among these
participants, 5 (2.9%) declined to participate in the study; 6 (3.5%)
refused surgery and accepted organ preservation treatment instead,
and 3 (1.8%) did not have squamous cell carcinoma in their final
pathological reports. In addition to above-mentioned participants,
we also excluded 13 (7.6%) who had inadequate surgical margins
(< 5 mm) and 9 (5.3%) with dysplasia in at least one of the mucosa
margins. Complete data were obtained from 135 participants. The
average age of participants at presentation was 52.5+11.1 years and
they were mostly men (n=125, 92.6%). The commonest primary site
was the tongue (n=55, 40.7%), followed by buccal mucosa (n=49,
36.3%) and gum (n=11, 8.1%). In terms of personal habits of these
patients, 107 (79.3%) were smokers, 106 (78.5%) consumed alcohol
socially or heavily, and 99 (73.3%) habitually chewed betel quid.
Regarding pathological stages of disease, 43 (31.8%) were in stage I,
24 (17.7%) in stage II, 16 (11.9%) in stage III, and 52 (38.5%) in stage
IV. Only 33 (24.4%) of patients developed local recurrence during the
follow-up period. Among these recurrent cases, 28 (20.7%) expired
due to extensive local recurrence or complications during salvage
treatments; 4 (3.0%) developed cervical lymph node recurrence. None
of these showed distant metastasis. The average follow-up period was
25.5 (+14.7) months. A total of 45 participants (33.3%) had MSI in
their cancerous specimens for one marker or more. Most MSI was
localized to D21S236 (n=16, 35.6%), followed by IFNA.PCR2 (n=10,
22.2%), and THRB (n=9, 20.0%). More than half of these participants
(n=78, 57.8%) had LOH in their tumor specimens for least one
marker. The most frequently-detected positive marker for LOH was
IFNA.PCR2 (n=40, 51.3%), followed by D9S1748 (n=27, 34.6%),
and D3S1300 (n=21, 26.9%). Based on the detected microsatellite
alterations, participants were divided into two groups. (Table 2 and 3)
listed comparisons of variables between the two groups. Participants
with MSI when compared with those without tended to have
higher rates of both local recurrence and mortality. In addition, the
proportion of perineural invasion was also higher in those with MSI.
Significant differences were not found between the MSI and non-MSI
groups in terms of age, gender, personal habits, primary tumor sites,
histological characteristics, angiolymphatic invasion, extra capsular
invasion, pathological stage, and postoperative radiotherapy (Table 2).
Regarding LOH, all variables were not significantly different between
those with LOH and those without (Table 3). In terms of quantitative
data of personal habits, the average cigarette consumptions were
similar between participants with MSI/LOH and those without (MSI
vs. non-MSI: 32.5+23.2 vs. 29.5+15.6 pack-years, P=0.486; LOH vs.
non-LOH: 27.9+16.2 vs. 34.2+20.8 pack-years, P=0.079). However,
participants with microsatellite alterations showed betel quid more
than those without (MSI vs. non-MSI: 511+433 vs. 331+275 quidyears,
P=0.031; LOH vs. non-LOH: 415+366 vs. 363+321 quid-years,
P=0.459). Although the 4-year disease-specific survival rate was lower
in participants with MSI than those without, the difference was not
significant. (80.3 % vs. 66.4 %, P=0.2280). On the other hand, the
4-year disease-specific survival rates were similar between participants
with LOH and those without (76.8 % vs. 74.3 %, P=0.9392) (Figure 3).
There were totally 606 surgical margins were obtained from
the defects after tumor extirpation. Among these specimens, 516
were from mucosa, and 90 from deep soft tissues. Surgical margins
obtained from participants without microsatellite alteration were
excluded. Consequently, 392 surgical margins (from 87 informative
participants) were analyzed in the logistical regression model. Of
these specimens, 46 (11.7%) had MSI, whereas 89 (22.7%) had LOH
in at least one marker. For those patients who had local recurrence, 37
of their margins were found near the recurrent sites. Here, MSI was
more likely to be found in such margins when compared with that of
those without local recurrence (17 out of 37, 45.9% vs. 29 out of 355,
8.2%, P< 0.001). Also, the proportion of LOH in above-mentioned
margins was also higher than those without local recurrence (14 out
of 37, 37.8% vs. 75 out of 355, 21.1%, P=0.035). In the multivariate
analysis, the presence of MSI in surgical margins increased risk of
over 7 folds in developing local recurrence [Odds ratio (OR): 7.494;
95% confidence interval (CI): 3.342 ~ 16.80; P< 0.001]. Finally, late
stage was another independent risk factor for local recurrence (OR:
3.865; 95% CI: 1.584 ~ 9.427; P=0.003). Detailed data are shown in
(Table 4).
Discussion
The relationship between MSI and the survival of colorectal
cancer was well-documented [3]. Our OCSCC patients however
demonstrated no relationship between microsatellite alterations and
survival rates. The clinical implication and prevalence of MSI are
different across tumors of different primary sites [5]. For example,
MSI is associated with the survival of gastrointestinal cancer and
endometrial cancer [13,14]. The prognostic value of MSI in OCSCC
remains controversial. Lack of statistical power in the literature with
a low prevalence of MSI in head and neck cancer may underly such
controversy [5]. Murali et al. [8] reported that oral cancer patients
with LOH at D9S162 have poor recurrence-free survival rates. Here,
we found no difference in disease-specific survival between OCSCC
patients with LOH and those without. Not only did we select a
different set of microsatellite markers but we also used different
techniques. For example, we adopted modern automatic fragment
analysis after PCR amplification whereas their study resolved PCR
amplified samples in poly-acrylamide gels and used silver staining for
detection. Moreover, the cutoff value of tumor imbalance factor (or
LOH ratio) in our study was < 0.67 or >1.5, while other studies used
dissimilar cutoffs values (such as < 0.5 or >2) [5,15]. Such differences
in methodology could account for the discrepancy in results. The
incidence of MSI in head and neck cancer varies from 7.7 to 48 %
and the occurrence of LOH ranges from 29.6 to 86.7% [4,5,7-12,16].
In our study, the prevalence of MSI was 33.3% and that of LOH was
57.8%. One possible explanation for the different frequency of MSI/
LOH is the different microsatellite markers selected. Also, the studied
populations are different across different studies and predisposing
factors in different countries are also dissimilar. Interestingly, the
average consumption of betel quid was higher in our patients with
MSI than those without. A previous study reported that patients with
genomic alterations in tumor DNA have higher consumption of betel
quid (two-fold difference) than patients without. Therefore, apart
from MMR system mutation, betel quid chewing is believed to cause
genomic instability which can ultimately lead to carcinogenesis [17].
Despite the lack of association between MSI/LOH and diseasespecific
survival in our OCSCC patients, the presence of MSI in the
tumor-free surgical margins was linked to a higher risk of developing
local recurrence. Temam et al. [16] in their study on head and neck
squamous cell carcinoma patients also reported a similar finding.
MSI represents the molecular fingerprint of the deficient mismatch
repair (MMR) system [3], which repairs errors that occurred in
DNA replication [1]. Mutations of the MMR genes (such as MLH1,
MSH2, MSH6, and PMS2) can lead to lynch syndrome, which is
closely related to hereditary non-polyposis colorectal cancer [14].
Dysfunction of MMR system may lead to carcinogenesis as mutations
accumulated in pivotal genes. Accumulation of genetic modifications,
such as inactivation of the tumor suppressor genes may initiate oral
carcinogenesis after several years [5]. Partridge et al. [18] reported
that premalignant lesions with MSI likely lead to develop into head
and neck cancer. That could well explain the 7-fold increment on
the risk of local recurrence we found in our OCSCC patients with
MSI present in their surgical margins. On the other hand, our study
showed no connection between the presence of LOH in the surgical
margin and local recurrence of the cancer. A previous study in head
and neck cancer patients showed that the genetically altered margin
(or LOH) is associated with a higher risk of local recurrence as well
as second primary [19]. The discrepancy in results between the two
studies might be related to dysplasia margins were included in their
study whereas we excluded margins with any form of dysplasia.
There were some limitations in our study. First, the external
validity of our findings is limited as our patients analyzed were from
a single institute. Second, the statistical power was probably low due
to the relatively small sample size. Third, we only collected MSI/LOH
rather than tumor suppressor gene (such as P53) or gene methylation
status in specimens. Furthermore, the follow-up period was likely
not long enough to determine the survival benefit of MSI. Lastly,
although the therapeutic guidelines are standardized in our institute,
differences in treatment of patient could not be ruled out.
Conclusion
MSI and LOH in the microsatellite markers designated herein were not associated with disease-specific survival of OCSCC patients in Taiwan, which is a betel quid-prevalent country. MSI present in the dysplasia-free surgical margins increased the risk of local recurrence. Genomic examinations of surgical margins could be helpful in screening out those patients in risk of developing local recurrence. Adjuvant treatments might therefore be provided for them to improve their prognosis.
Acknowledgment
We thank the Biostatistics Task Force of Taichung Veterans General Hospital for assistance in statistics. Supported by grants from the National Science Council, Taiwan, Republic of China (NSC 101- 2314-B- 075A-005-MY3) and the Ministry of Health and Welfare, Taiwan, Republic of China (MOHW104-TDU-B-211-124-004).
References
- Watson MM, Berg M, Søreide K. Prevalence and implications of elevated microsatellite alterations at selected tetranucleotides in cancer. Br J Cancer. 2014; 111: 823-827.
- Lin JC, Wang CC, Jiang RS, Wang WY, Liu SA. Microsatellite alteration in head and neck squamous cell carcinoma patients from a betel quidprevalent region. Sci Rep. 2016; 6: 22614.
- Sinicrope FA, Sargent DJ. Molecular pathways: microsatellite instability in colorectal cancer: prognostic, predictive, and therapeutic implications. Clin Cancer Res. 2012; 18: 1506-1512.
- Deganello A, Gitti G, Mannelli G, Meccariello G, Gallo O. Risk factors for multiple malignancies in the head and neck. Otolaryngol Head Neck Surg. 2013; 149: 105-111.
- De Schutter H, Spaepen M, Mc Bride WH, Nuyts S. The clinical relevance of microsatellite alterations in head and neck squamous cell carcinoma: a critical review. Eur J Hum Genet. 2007; 15: 734-741.
- Choi KY, Choi HJ, Chung EJ, Lee DJ, Kim JH, Rho YS. Loss of heterozygosity in mammary serine protease inhibitor (maspin) and p53 at chromosome 17 and 18 in oral cavity squamous cell carcinoma. Head Neck. 2015; 37: 1239-1245.
- Ashazila MJ, Kannan TP, Venkatesh RN, Hoh BP. Microsatellite instability and loss of heterozygosity in oral squamous cell carcinoma in Malaysian population. Oral Oncol. 2011; 47: 358-364.
- Murali A, Sailasree R, Sebastian P, Rejnish Kumar R, Varghese BT, Kannan S. Loss of heterozygosity of D9S162: molecular predictor for treatment response in oral carcinoma. Oral Oncol. 2011; 47: 571-576.
- Lee SH, Lee NH, Jin SM, Rho YS, Jo SJ. Loss of heterozygosity of tumor suppressor genes (p16, Rb, E-cadherin, p53) in hypopharynx squamous cell carcinoma. Otolaryngol Head Neck Surg. 2011; 145: 64-70.
- Mahale A, Saranath D. Microsatellite alterations on chromosome 9 in chewing tobacco-induced oral squamous cell carcinomas from India. Oral Oncol. 2000; 36: 199-206.
- Arai K, Shibahara T, Yamamoto N, Noma H. The presence of candidate tumor suppressor gene loci at chromosome 3p for oral squamous cell carcinomas. Oral Oncol. 2002; 38: 763-771.
- Koy S, Plaschke J, Luksch H, Friedrich K, Kuhlisch E, Eckelt U, et al. Microsatellite instability and loss of heterozygosity in squamous cell carcinoma of the head and neck. Head Neck. 2008; 30: 1105-1113.
- Zhu L, Li Z, Wang Y, Zhang C, Liu Y, Qu X. Microsatellite instability and survival in gastric cancer: A systematic review and meta-analysis. Mol Clin Oncol. 2015; 3: 699-705.
- Sijmons RH, Hofstra RM. Review: Clinical aspects of hereditary DNA Mismatch repair gene mutations. DNA Repair (Amst). 2016; 38: 155-162.
- De Schutter H, Spaepen M, Van Opstal S, Vander Poorten V, Verbeken E, Nuyts S. The prevalence of microsatellite instability in head and neck squamous cell carcinoma. J Cancer Res Clin Oncol. 2009; 135: 485-490.
- Temam S, Casiraghi O, Lahaye JB, Bosq J, Zhou X, Julieron M, et al. Tetranucleotide microsatellite instability in surgical margins for prediction of local recurrence of head and neck squamous cell carcinoma. Clin Cancer Res. 2004; 10: 4022-4028.
- Zienolddiny S, Aguelon AM, Mironov N, Mathew B, Thomas G, Sankaranarayanan R, et al. Genomic instability in oral squamous cell carcinoma: relationship to betel-quid chewing. Oral Oncol. 2004; 40: 298- 303.
- Partridge M, Pateromichelakis S, Phillips E, Emilion GG, A'Hern RP, Langdon JD. A case-control study confirms that microsatellite assay can identify patients at risk of developing oral squamous cell carcinoma within a field of cancerization. Cancer Res. 2000; 60: 3893-3898.
- Tabor MP, Brakenhoff RH, van Houten VM, Kummer JA, Snel MH, Snijders PJ, et al. Persistence of genetically altered fields in head and neck cancer patients: biological and clinical implications. Clin Cancer Res. 2001; 7: 1523-1532.