Research Article
Pathogenic Mechanisms for Spermatocele and Epididymal Cyst Formation
Kathyrn LaRusso1, Jeffery Miller4, Kahlil Saad2 and George F Steinhardt3*
1Department of Surgery, University of Texas Health Center, USA
2Department of Urology, Wayne State University, USA
3Department Urology, Helen DeVos Children's Hospital, USA
4Department of Pathology, Spectrum Health, USA
*Corresponding author: George Steinhardt, Department Urology, Helen DeVos Children's Hospital, NE Suite 3300, Grand Rapids, Michigan 49506, USA
Published: 27 Dec, 2016
Cite this article as: LaRusso K, Miller J, Saad K, Steinhardt
GF. Pathogenic Mechanisms for
Spermatocele and Epididymal Cyst
Formation. Clin Surg. 2016; 1: 1261.
Abstract
Introduction: Spermatoceles (SC) and Epididymal Cysts (EC) are commonly diagnosed with the
onset of symptoms or more often as incidental sonographic findings. In experimental animals, fetal
tissues exposed to estrogen mimetic compounds demonstrate the development of abnormal cystic
structures in the epididymis of adult animals. The only known cause of EC in adult humans is
fetal exposure to the potent estrogen diethylstilbestrol (DES). Additionally, previous studies have
also shown that Estrogen Receptor knockout (ERKO) mice demonstrate disruption of luminal fluid
resorption in the efferent ducts in the head of the epididymis which is the usual anatomic location
for both SC and EC. This study investigates the possible association of undue estrogen stimulation
and the occurrence of spermatoceles (SC) and epididymal cysts (EC) by analyzing human SC and
EC tissue for the estrogen inducible protein lactoferrin (LF) as well as estrogen receptor 1 (ESR1)
and estrogen receptor 2 (ESR2).
Methods: An IRB approved study identified 10 patients with well-documented SC and 7 patients
with EC between the years 2007-2012. Formalin fixed paraffin embedded blocks of surgically
removed tissue were stained using immunohistochemistry (IHC) for lactoferrin (LF), estrogen
receptor1 (ESR1) and estrogen receptor 2 (ESR2).
Results: Both ECs and SCs stained positive for LF, ER1 and ER2. Results are summarized in the
table below.
Conclusion: IHC staining for LR, ER1, and ER2 suggests estrogen stimulation and disruption as a
possible pathogenesis of both EC and SC.
Keywords: Spermatocele, Epididymal Cyst; ESR
Introduction
Epididymal cysts (EC) or fluid-filled collections at the head of the epididymis are relatively
common and generally asymptomatic. Lined with pseudo stratified columnar epithelium, they are
histologically distinguished from spermatoceles, which are lined by simple cuboidal epithelium and
contain spermatozoa or sperm fragments in the lumen [1]. Spermatoceles tend to be large and may
be removed surgically with symptoms of pain or discomfort [2]. Clinically, the difference between
spermatoceles and epididymal cysts is not clear cut and the distinction between the two may be
largely arbitrary based on size. Recent studies have suggested an increasing frequency in epididymal
cysts in humans [3-5].
The male reproductive system, including the epididymis, is dependent on both androgen and
estrogen-mediated interactions for normal development and fertility. Estrogen-mediated processes
are regulated by the estrogen receptors ESR-1 and ESR-2 which are variably localized throughout the
male reproductive system dependent upon species, localizing techniques and developmental stage
[5]. The receptors mediate gene transcription through direct and indirect signaling pathways. ESR-
1 has been shown to regulate key factors in epididymal luminal contents including ion transport
channels and aquaporin channel expression [6]. Lactoferrin (LF) is an estrogen inducible nonheme
binding multifunctional glycoprotein that transports iron, modulates the immune system,
and inhibits bacterial growth [7]. Though LF expression in the epididymis is not well characterized,
LF has been identified as an important component of seminal fluid and serves to coat spermatozoa
during transit through male and female reproductive tracts.
According to the Theory of Testicular Dysgenesis exposure
to estrogens during early stages of development may account for
the increasing frequency of male reproductive problems including
hypospadias, testicular maldescent, infertility and testicular cancer
[8,9]. Both experimental and clinical studies have shown that prenatal
exposure to the potent estrogen diethylstilbestrol (DES) causes the
development of EC among other lesions [10,11]. The amount of
prenatal estrogen exposure may be correlated with levels of LF found
in the epididymis [12]. Indeed the experimental observations that
linked LF expression, EC formation and prenatal exposure to DES
constituted a landmark study in the fields of developmental andrology
and environmental toxicology [13].
This study investigates both SC and EC with a novel assessment
of well characterized clinical histories, operative notes and surgical
specimens utilizing immunohistochemistry to compare the
expression of LF, ESR-1 and ESR-2 in these tissues.
Table 1
Table 1
Depicts the number of patients, average presenting age of the patients, presenting symptom and average size of the spermatoceles (SC) vs. epididymal cysts (EC)..
Table 2
Table 2
Depicts the number of specimens that were positive for LF, ESR1 and
ESR2 for patients with SC vs EC. p-value by Fisher’s Exact Test.
Materials and Methods
In this IRB approved study, hospital pathology reports were
queried for the words "epididymal cyst" and "spermatocele" between
the years 2007 and 2012. Patient records were then reviewed as well
as the dictated operative notes and microscopic slides from the
pathology specimens. A total of 17 patients were identified (SC=10
and EC=7) for whom the diagnosis was consistent with the clinical
history, the operative dictation, and the microscopic pathologic
findings. Formalin fixed and paraffin embedded tissue blocks were
retrieved from the surgical tissue archive for IHC assessment. 4
μM formalin-fixed paraffin embedded sections were stained for
Lactoferrin (83440) [14] at a concentration of 6ug/ml), ERα (Ventana,
clone SP1, used neat) and ERβ (Dako, clone pp65/10, diluted 1:20)
using a Ventana Benchmark Ultra stainer. Briefly, antigen retrieval
for ERα and ERβ was accomplished online using Ventana CC1 for 32
minutes. No retrieval was used for the lactoferrin. Primary antibodies
were incubated for 32 minutes. Visualization was achieved using the
Ventana Ultraview DAB kit followed by hematoxylin staining.
We initially intended to grade the intensity of staining in the
specimens, but as most specimens were either strongly positive
or negative for LF, ESR-1 and ESR-2 we tabulated only positive or
negative staining.
Results and Discussion
The number of patients with either SC or EC, the average presenting
age, presenting symptoms and average size SC vs EC are presented in
Table 1. The age range for the 17 patients whose tissue was examined
was from 15 to 83 years, and while ECs were younger, this was not
statistically significant by a paired t-test. For both SC and EC patients
the most common presenting symptom was scrotal pain followed by
the awareness of a scrotal mass, though no statistical significance by
Fisher’s exact test was demonstrated. One of the spermatoceles was
incidentally present in the surgical specimen that was obtained at
the time of orchiectomy for testicular cancer (embryonal carcinoma)
and another patient had the incidental diagnosis for spermatocele
made at the time of orchiectomy for prostatic cancer. One37-yearold
patient with EC had the incidental diagnosis made at the time of
orchiectomy for an intraabdominal testis. The volume of the SC and
EC were calculated by the volume of the ellipsoid formula; from the
table it is clear that the SC were much larger than EC, though this
failed to reach significance by a paired t-test.
IHC positivity for LF, ESR1 and ESR2 is reported in Table 2 as
simply present or absent. Regarding LF, 80% of spermatoceles and 71%
of epididymal cysts stained positive. In total, 60% of spermatoceles
and 71% of epididymal cysts stained positive for ER1 and 70% of
spermatoceles and 29% of epididymal cysts stained positive for
ER2. In the spermatocele group, there were two patients that did
not stain positive of LF - one patient had embryonal carcinoma and
the other patient did also not stain positive for ER1 or ER2. Within
the epididymal cyst group, there was no correlation between those
that did not stain positive for LF and those that stained positive for
ER1/ER2. Statistical analysis of staining patterns was done with the
Fisher’s exact test. Figures 1,2 and 3 demonstrate characteristic IHC
staining for LF, ESR1 and ESR2 in this study.
LF is an estrogen inducible protein [14,15] that is present
throughout the lumen of the epididymis and serves an important role
in coating spermatozoa [16]. It has been established that LF mRNA
is present in the epididymal cells which produce LF in significant
amounts in response to estrogen stimulation [17]. In addition to
LF elaboration, apical epididymal cells function to secrete and
absorb luminal substances as well as fluid so as to create a balanced
environment for sperm maturation [18,19]. These processes including
fluid transport are controlled by a balance of androgen and estrogen
action [20]. ESR1 is constitutively expressed in the epididymis of
experimental animals but is down regulated by high doses of estrogen
[6]. Prior experimental studies demonstrate that ER1 regulates fluid
resorption in the efferent duct serving to concentrate sperm before
entering the distal portions of the epididymis. Enhanced exposure to
estrogens may impact the expression of ER1 and affect the active and
passive transport of ions and water in the epididymal caput perhaps
explaining the pathogenesis for both EC and SC. Such pathogenic
mechanisms are further suggested by experiments which show ESR-
1 knockout mice to have impaired fluid resorption in the efferent
ductules in the epididymis which results in fluid accumulation
and retention in the rete testis with resultant seminiferous tubular
degeneration and atrophy [21].
Estrogen stimulation via endocrine disrupting chemicals is a topic
of major interest and concern [22,23]. It is undeniable that a whole
host of man-made organochloro-compounds are weakly estrogenic
in cell systems. Furthermore, in many experimental circumstances
fetal and post-natal exposure to these chemicals causes a wide variety
of anatomic abnormalities in the reproductive tract of male animals.
The intense staining for the estrogen inducible protein LF in both
EC and SC implicates estrogen action in the pathogenesis of these
structures. This idea is further supported by the intense staining of
both ESR-1 and ESR-2 in both EC and SC. It seems likely that SC
and EC represent large and small versions of the same pathologic
processes occurring in the caput epididymis. Our findings suggest the
possibility that their occurrence may be yet another consequence of
estrogen mimetic endocrine disrupting chemicals.
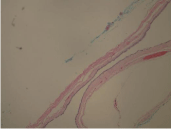
Figure 1A
Figure 1A
Is a photomicrograph of representative EC tissue stained with
H&E at 10X showing typical pseudo-stratified columnar epithelium.
Figure 1B
Figure 1B
Demonstrates IHC staining for LF in the same tissue at 20X for
LF. The staining shows diffuse and intense throughout the epithelial cells
of the EC.
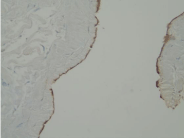
Figure 2A
Figure 2A
Demonstrates IHC staining of EC tissue for ESR-2 at 20X. Notice intense localization not only in the epithelium of the EC but also in the epithelium of the adjacent epididymal tubules.
Figure 2B
Figure 2B
Demonstrates ICC staining of EC tissue for ESR-2 at 20X. While the EC epithelium is negative for ESR-2, the adjacent epididymal tubules show intense localization.
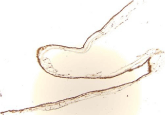
Figure 3A
Figure 3B
Figure 3B
Demonstrates IHC staining for ESR-1 in SC tissue at 10X. The
ESR-1 staining is quite intense.
Figure 3C
Figure 3C
Demonstrates IHC staining for ESR-2 in SC tissue at 20x. The ESR-2 staining is quite intense.
Conclusion
When comparing well characterized patients with SC vs. EC we find that both groups present either with scrotal discomfort or appreciation of a scrotal mass. Generally patients with EC are a bit younger and the scrotal mass is smaller. IHC staining for the estrogen inducible protein LF is equally prominent in tissue from both EC and SC patients.IHC staining for ESR-1 is equally intense and prevalent in EC and SC while ESR-2 staining is intense and equally prevalent tissue from SC patients while the tissue from EC is as intense, but somewhat less prevalent. These findings suggest no substantial difference between EC and SC and implicate endocrine disruption by estrogen mimetic chemicals in their pathogenesis. Given the prevalence of EC in our prior studies, we would predict an increased performance of spermatocelectomy in the future.
Acknowledgement
We wish to thank Christina T Teng, PhD for her generous gift of mouse anti human lactoferrin antibody.
References
- Huggins C, Noonan WJ. Spermatocele, including x-ray treatment. J Urol. 1938; 39: 784-790.
- Nesbitt JA. Hydrocele, Spermatocele, In: Graham S, Editor –in-Chief. Glenn’s Urologic Surgery. 6th ed. Philadelphia: Lippincott Williams & Wilkins. 2004: 598-602.
- Homayoon K, Steinhardt GF. Epididymal cysts in children: Natural history. J Urol. 2004; 171: 1274- 1278.
- Posey ZQ, Ahn HJ, Junewick J, Chen JJ, Steinhardt GF. Rate and associations of epididymal cysts in pediatric scrotal ultrasounds. J Urol. 2010; 184: 1739-1742.
- Weatherly D, Wise P, Shawn M, Loeb A, Chen Y, Chen JJ, et al. Epididymal Cysts: Are they associated with Infertility? Am J Men’s Health. 2016: 1-5.
- Joseph A, Shur BD, Hess RA. Estrogen, Efferent Ductules, and the Epididymis. Biol Reprod. 2011; 84: 207-217.
- Teng CT, Liu Y, Yang N, Walmer D, Panella T. Differential molecular mechanism of the estrogen action that regulates lactoferrin gene in human and mouse. Mol Endo. 1992; 6: 1969-1981.
- Sharpe RM. Pathways of endocrine disruption during male sexual differentiation and masculinization. Best Prac Res Clin Endocrinol Metab. 2006; 20: 91-100.
- Skakkebek NE, Meyts RD, Main KM. Testicular dysgenesis syndrome: An increasing common developmental disorder with environment aspects. Human Reprod. 2001; 16: 972-978.
- Gill WB, Schumacher GF, Bibbo M, Straus FH, Schoenberg HW. Association of diethylstilbestrol exposure in utero with cryptorchidism, testicular hypoplasia and semen abnormalities. J Urol. 1979; 122: 36-39.
- Newbold RR, Hanson RB, Jefferson WN, Bullock BC, Haseman J, McLachlan JA. Proliferative lesions and reproductive tract tumors in male descendants of mice exposed developmentally to diethylstilbestrol. Carcinogenesis. 2000; 21: 1355-1363.
- Pearl CA, Roser JF. Expression of lactoferrin in the boar epididymis: effects of reduced estrogen. Domest Anim Endocrinology. 2008; 34: 153-159.
- McLachlan JA, Newbold RR, Bullock B. Reproductive tract lesions in male mice exposed prenatally to diethylstilbestrol. Science. 1975; 190: 991-992.
- Teng CT, Walker MP, Bhattacharyya SN, Klapper DG, DiAugustine RP, McLachlan JA. Purification and properties of an oestrogen-stimulated mouse uterine glycoprotein (approx. 70 kDa) Biochem J. 1986; 262: 413- 422.
- Teng CT, Liu Y, Yang N, Walmer D, Panella T. Differential molecular mechanism of the estrogen action that regulates lactoferrin gene in human and mouse. Molec Endoc. 1992; 6: 1969-1981.
- Fouchecourt S, Metayer S, Locatelli A, Dacheux F, Dacheux JL. Stallion epididymal fluid proteome: qualitative and quantitative characterization; secretion and dynamic changes of major proteins. Biol Reprod. 2000; 62; 1790-1803.
- Yu LC, Chen YH. The developmental profile of lactoferrin in mouse epididymis. Biochem J. 1993; 296: 107-111.
- Robaire B, Hinton BT, Orgebin-Crist. The Epididymis. In: Neill JD, editor. Knobil and Neill’s Physiology of Reproduction. , 3rd ed. Philadelphia, Elsevier. 2006.
- Robaire B, Viger RS. Regulation of epididymal epithelial cell functions. Biology of Reproduction. 1995; 52: 226-236.
- Hess RA, Fernandes SA, Gomes GR, Oliveeira CA, Lazari MM, Porto CS. Estrogen and its receptors in efferent ductules and epididymis. J Androl. 2011; 32: 600-613.
- Carreau S, Hess RA. Oestrogens and spermatogenesis. Phil Trans R Soc B. 2010; 365: 1517-1535.
- Sharpe RM. Environmental oestrogens and male infertility. Pure & Appl Chem. 1998; 70: 1685-1701.
- Crews D, McLachlan JA. Epigenetics, evolution, endocrine disruption, health and disease. Endocrin. 2006; 147: S4-S10.