Review Article
Impact of Skeletal Muscle Loss and Functional Deconditioning on Outcomes in Hepatic Resection and Liver Transplantation: A Review
Randeep Kashyap* and M. Katherine Dokus
Department of Surgery, Division of Solid Organ Transplantation, University of Rochester Medical Center, USA
*Corresponding author: Randeep Kashyap, Department of Surgery, Division of Solid Organ Transplantation, University of Rochester Medical Center, 601 Elmwood Avenue, Box Surg-Txp, Rochester, NY 14642, USA
Published: 23 Dec, 2016
Cite this article as: Kashyap R, Katherine Dokus M. Impact
of Skeletal Muscle Loss and Functional
Deconditioning on Outcomes in Hepatic
Resection and Liver Transplantation: A
Review. Clin Surg. 2016; 1: 1259.
Abstract
A growing number of older and sicker patients are being offered surgical intervention for their
ailments. The effects of frailty and sarcopenia, syndromes characterized by loss of muscle mass
and functional reserve are more prevalent in this population, and operative outcomes are just
beginning to be elucidated. Here, we provide a review of published reports on mortality, morbidity
and resource utilization in liver transplant and hepatectomy on frail and/or sarcopenic adults.
Fourteen studies were selected for review; 82% of analyses reporting described greater resource
utilization (e.g. length of stay, readmissions) in the frail cohort, 80% demonstrated increased rates
of morbidity. The majority of included studies (69%) reported higher mortality in the debilitated
groups undergoing liver transplant or hepatectomy with hazard of death ranging from 1.05-26.4.
Though the preponderance of studies pointed to worse outcomes for frail or sarcopenic patients;
lack of uniformity on the operational definition of these concepts makes generalization and clinical
application difficult. A validated, practicable measure can help surgical teams identify candidates at
greater risk of poor outcomes and implement interventions to reduce such hazards.
Keywords: Frail; Sarcopenia; Liver transplantation; Liver surgery; Outcomes
Introduction
Healthcare providers are amidst a dynamic shift in the demographics of their surgical candidate
population. The benefits of intervention, innovations in anesthesia and surgical technique as well as
improvements in medical care have offered older and sicker patients an operative option for their
ailments. Based on 2010 data, adults over 65 in the United States underwent 37% of all inpatient
procedures- a rate much higher than their 13% share of the population [1,2]. This proportion is only
certain to escalate. By 2030, 72 million Americans (1 in 5) will be an older adult [3]. Likewise, the
global population of older persons is increasing at 2.6% per year, a rate more than twice that of the
population as a whole [4].
Despite progress, research continues to show that older individuals suffer worse outcomes in
terms of 30-day post-operative mortality and greater perioperative complications than their younger
counterparts [5-9]. Arguments for distinctions between numerical and physiological age, however,
are becoming more prevalent. Proponents argue that chronological age has incorrectly been used as
a surrogate for that which has greater influence on outcomes: physiological deconditioning [10,11].
Debility has been operationalized in surgical research in two broad categories: frailty and sarcopenia.
Frailty, generally described as the loss of reserve capacity across organ systems and high
susceptibility to stressors, has been a growing concept in the literature over the past two decades.
Though not fully understood, its pathophysiology is marked by chronic inflammation, compromised
immunity, neuroendocrine and metabolic dysregulations, with contributing psychosocial factors
[12]. The frailty syndrome has been correlated with higher mortality, falls, and hospitalizations [10].
Sarcopenia, first described as the loss of skeletal muscle mass, has evolved to incorporate cellular
processes and outcomes such as decreased muscle strength, diminished mobility and function; it
has also been associated with heightened risk of adverse events [13]. Though not inclusive of the
psychosocial component of the frailty syndrome, phenotypically the two are similar and arguments
to utilize sarcopenia in research and clinically are proliferating in the literature [14,15].
Two leading models to measure frailty are deficit accumulation and the frailty phenotype.
Deficit accumulation, popularized by Mitnitski et al. [16] outlines the consequences of frailty in
terms of illness and impairments in physical function, lifestyle, and
cognition. Fried’s frailty phenotype standardizes the definition via
biometric and self-reported variables including weight loss, grip
strength, and activity level [10]. Akin to frailty, there is no agreed
upon operational definition of sarcopenia; but most research
protocols utilize quantitative methods such as muscle area on MR
or CT, DXA, or bioelectrical impedance. Surgeons typically have
the additional resource of abdominal imaging for pre-operative
planning which can be used to incorporate assessment of muscle
mass into routine evaluation. A recent review of biomarkers by the
International Working Group on Sarcopenia, though, recommends
adopting a comprehensive operational definition of the conceptgauging
physical functioning of muscle above simply measuring mass
[17].
Though more frequent in older adults, deconditioning is
not synonymous with aging, it may also be a consequence of
chronic disease. As seen in patients with cirrhosis awaiting liver
transplantation (LT), the debilitating condition may also be the
reason for operative intervention. Resulting from altered protein
metabolism, poor nutritional intake due to factors including ascites
and encephalopathy, as well as sedentary lifestyle partly attributed
to the rigors of the disease, frailty and sarcopenia are common in
those with end stage liver disease. In a single center study of those
undergoing LT evaluation, 40% were sarcopenic, correlating with
heightened mortality on multivariate analysis (HR 2.18 p= 0.006);
another transplant center determined 17% of their studied evaluations
to be frail and at increased risk of waitlist mortality independent of
their model for end-stage liver disease (MELD) score (HR 1.45, p=
0.03) [18,19]. While disease burden is a significant contributor to
frailty in the liver transplant population, a growing number of older
adults are also undergoing transplant procedures. Between 2002 and
2015 the share of transplanted patients over the age of 64 more than
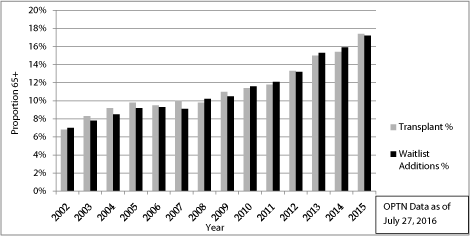
doubled from to 6.8 to 17.4% (Figure 1).
Challenged with organ demand far outstripping supply and
implementation of MELD system in 2002, LT patients tend to
become more deconditioned while waiting for a viable organ to be
offered. Transplant programs must balance equity with selecting
the patients who are going to achieve the best outcomes with this
precious resource.
Transplant is not the only surgical field which has seen growth,
and whose trend is poised to continue in the near future. Attributed
to the aging population, Etzioni et al. [20] project a twenty-year
growth rate of 31% in general surgery (including abdominal and
GI procedures) by 2020. Hepatobiliary specialties are expected to
increase procedure volume by 25% between 2006 and 2020 [21]. The
effects of frailty are more prevalent in this burgeoning population,
and operative results are just beginning to be elucidated. Here, we
provide a review of current research on the outcomes of hepatectomy
and liver transplantation in frail and/or sarcopenic adults.
To qualify for inclusion, studies must have reported on
post-surgical mortality in patients undergoing liver resection
or transplantation (from deceased or living donors). Major
complications and health care resource utilization are reported as
secondary outcomes. Prospective and retrospective cohort studies
and case-control analyses were eligible for inclusion. Studies
measuring sarcopenia or frailty, though acknowledged as separate,
yet intrinsically related clinical entities were included. Articles not
published in English, and studies that did not report on the primary
outcome measure (mortality) were excluded from review.
Table 1
Figure 1
Figure 2
Findings
Characteristics and results of included studies are summarized on Table 1.
Fourteen studies were included. Rates of frailty or sarcopenia
ranged from 2.6% to as high as 68% of included subjects. The selected
papers consisted of eight studies on outcomes in liver transplantation
(two of which were living donor), five in the resection population,
and one paper that included both patients undergoing hepatectomy
and those receiving a liver transplant for HCC.
Resource Utilization
All studies reporting on health care delivery metrics, save one, found at least one measure (overall or ICU length of stay (LOS), readmission, cost, etc.) of heavier utilization in the frailty/sarcopenia group. Length of stay was reported in five studies and ranged from non-significant [22] to 1.2-15 days, or 22-133% longer in the frail/ sarcopenic contingent [6,23-26]. Average ICU LOS was increased by a range of 1-6 days [24,26] The only study to report on readmissions described 90-day post-transplant rates 24% above non-frail [27]. Institutionalization at discharge or inpatient rehab was assessed in two protocols and elevated in the frailty/sarcopenic arm in both by 11.7-18% [23,27].
Morbidity
The majority of studies (9 of 11 reporting) showed a greater
incidence of morbidity in frail or sarcopenic subjects; all liver
transplant articles described more complications in the debilitated
cohort. Complications Clavien-Dondo grade ≥3 were analyzed in
five studies and significantly elevated in three, with the odds of major
complications 3.1-40.6 times greater than the non-frail comparator
group [22,24,25]. Common morbidities included infection,
respiratory failure, biliary complications, renal failure, and universal
reoperation in the frailest LT patients in one study [27].
Mortality
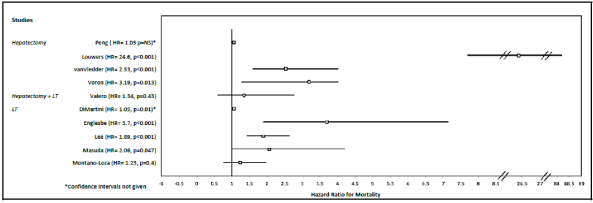
Of the ten studies that provided hazard ratios, 70% found significantly greater likelihood of mortality with frailty/sarcopenia ranging from HR of 1.05 to 26.4 (Figure 2). Survival was assessed at different time points across studies. Peri-operative and one year survival rates were inconsistent between the six published reports with this outcome. Three of studies showed significantly higher 1 year mortality in those considered frail or sarcopenic (2 LT, 1 hepatectomy) [28-30]; whereas three others (2 LT, 1 hepatectomy) reported no short-term survival difference between groups [18,27,31]. Long-term survival ≥3 years were significantly diminished in frail/sarcopenic individuals in the majority of studies reporting this outcome (5/8). Of the four studies exclusively examining the liver transplant population, three reported greater hazard of death in sarcopenic patients.
Discussion
The past few years have seen frailty emerging from the world of
geriatrics into the medical arena at large. As demonstrated by this
review, there has been a recent burgeoning of studies in the surgical
literature- all included papers have been published after 2010 and the
majority in the past three years. This is likely a reflection both of the
population surgeons are serving and the increasing recognition of
frailty as a risk factor in outcomes. Other pressures are also impacting
surgical practice. In an environment of ever-increasing scrutiny on
outcomes and costs, it is essential to balance what is right for our
patients with optimization of resources, be that cost and in the case
of transplant- precious donated organs. Though not uniformly, the
majority of studies on hepatectomy and liver transplant surgeries
reported worse outcomes in morbidity and mortality, as well as
greater health care utilization in frail patients.
Unsurprisingly, all studies that demonstrated greater health care
utilization described statistically higher complication rates in the frail/
sarcopenic group. A wealth of studies has demonstrated that postoperative
complications and infections contribute to greater LOS,
higher readmissions and costs and lower per patient contribution
margins. [32-34] A recent in depth analysis specifically in the
sarcopenic population, by Sheetz et al. [35] quantified the financial
impact of surgery in this population: per 1000mm2 decrease in lean
Unsurprisingly, all studies that demonstrated greater health care
utilization described statistically higher complication rates in the frail/
sarcopenic group. A wealth of studies has demonstrated that postoperative
complications and infections contribute to greater LOS,
higher readmissions and costs and lower per patient contribution
margins. [32-34] A recent in depth analysis specifically in the
sarcopenic population, by Sheetz et al. [35] quantified the financial
impact of surgery in this population: per 1000mm2 decrease in lean
psoas area, there was a $6,989.17 increase in payer cost (p< 0.001).
This figure increased dramatically- to $26,988.41 if complications
arose. Robinson et al. [36] also performed a cost analysis in their
study of veterans undergoing colorectal surgery and observed average
6-month post-operative health care costs to be $77,249 higher in
the frail group. Though complications were not reported, greater
discharge institutionalization and readmission rates were seen in the
frail group.
Little consensus was reached across studies in regards to shortterm
mortality. This finding is especially interesting in the studies of
liver transplant patients, where complication rates were clearly higher
in frail/sarcopenic patients. This may be a result of these patients being
followed closely to assess for and promptly manage any complications
that arise. Long-term mortality in frail individuals was diminished in
more than two-thirds of studies reporting on this outcome. This is
likely not an effect of the intervention, but health status, including
underlying operative indication. However, some recent studies have
demonstrated halting and even reversal of sarcopenia post-transplant
[26 and 37].
In 2002, MELD score was implemented across the United States
to prioritize listing based on severity of liver disease. The utilization of
MELD score has led to the reduction in a number of deaths of patients
on the waiting list [38]. One of the major limitations of the MELD
score, however, is its inability to evaluate and recognize the nutritional
and functional status of patients with cirrhosis. Though both frailty/
sarcopenia and MELD are predictors of pre-transplant mortality,
MELD has not been shown to be associated with sarcopenia and
only weakly linked with frailty [18,19,27,39,40]. It therefore should
not be utilized as a surrogate for a validated measure of physical
deconditioning in LT patients. Future studies should explore whether
the addition of a debility measure to MELD score to prioritize listing
would improve waitlist mortality or post-transplant outcomes.
In a recent systematic review, de Vries, et al. [41] identified 20
unique frailty instruments. Of course, proper identification of frail
and sarcopenic patients is hampered by a paucity of consensus
among disparate definitions, instruments and cutoffs. Eight different
measures (2 frailty, 6 sarcopenia) were used in the studies included in
our review; even in those using the same imaging analysis techniques,
only two pairs used the same cut-off to delineate sarcopenia
[23,30,31,39].
Because of the wide range of available assessments and lack of
uniformity in the studied population, it is difficult for the authors
to recommend a tool to measure debility in the liver resection and
transplant population. What is clear is that the measure should be
validated with additional studies, based upon assessments that are
readily available or easily administered in the clinical setting and
incorporate both physical and psychosocial components of wellbeing.
Along with non-standardized definitions, the indications
for included surgeries and variety of reported endpoints makes
generalizations difficult, an acknowledged limitation of this review.
Conclusion
Based upon a review of the current literature, physical deconditioning and skeletal muscle loss appear to confer higher rates of morbidity and mortality and greater resource utilization in patients undergoing liver transplant or resection. Additional prospective investigations of surgical outcomes using a standard operational definition of frailty or sarcopenia are warranted. A validated, practicable measure can help surgical teams identify candidates at greater risk of poor outcomes and implement pre-habilitative interventions such as nutritional support, physical therapy/exercise [42], and pharmacotherapy to reduce such hazards.
References
- Centers for Disease Control and Prevention NCfHS. National Hospital ischarge Survey, 2010. In: Survey CNNHD. Hyattsville, editor. 2012.
- Werner CA. The Older Population: 2010. 2010 Census Briefs. In: Bureau USDoCEaSAUSC, ed. Washington DC. 2011.
- Prevention CfDCa. The State of Agining and Health in America 2013. Atlanta, GA: US Department of Health and Human Services. 2013./a>
- United Nations. Department of Economic and Social Affairs. Population Division. World population ageing. 2007. New York: United Nations, Dept. of Economic and Social Affairs, Population Division. 2007.
- Al-Refaie WB, Parsons HM, Henderson WG, Jensen EH, Tuttle TM, Vickers SM, et al. Major cancer surgery in the elderly: results from the American College of Surgeons National Surgical Quality Improvement Program. Ann Surg. 2010; 251: 311-318.
- Duron JJ, Duron E, Dugue T, Pujol J, Muscari F, Collet D, et al. Risk factors for mortality in major digestive surgery in the elderly: a multicenter prospective study. Ann Surg. 2011; 254: 375-382.
- Gajdos C, Kile D, Hawn MT, Finlayson E, Henderson WG, Robinson TN. Advancing age and 30-day adverse outcomes after nonemergent general surgeries. J Am Geriatr Soc. 2013; 61: 1608-1614.
- Hamel MB, Henderson WG, Khuri SF, Daley J. Surgical outcomes for patients aged 80 and older: morbidity and mortality from major noncardiac surgery. J Am Geriatr Soc. 2005; 53: 424-429.
- Kurian AA, Wang L, Grunkemeier G, Bhayani NH, Swanstrom LL. Defining "the elderly" undergoing major gastrointestinal resections: receiver operating characteristic analysis of a large ACS-NSQIP cohort. Annals of surgery. 2013; 258: 483-489.
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci ed Sci. 2001; 56: M146-156.
- Rockwood K, Song X, Mitnitski A. Changes in relative fitness and frailty across the adult lifespan: evidence from the Canadian National Population Health Survey. CMAJ. 2011; 183: E487-494.
- Fulop T, Larbi A, Witkowski JM, McElhaney J, Loeb M, Mitnitski A, et al. Aging, frailty and age-related diseases. Biogerontology. 2010; 11: 547-563.
- Lang T, Streeper T, Cawthon P, Baldwin K, Taaffe DR, Harris TB. Sarcopenia: etiology, clinical consequences, intervention, and assessment. Osteoporos Int. 2010; 21: 543-559.
- Cruz-Jentoft AJ, Michel JP. Sarcopenia: A useful paradigm for physical frailty. Eur Geriatr Med. 2013; 4: 102-105.
- Rizzoli R, Reginster JY, Arnal JF, Bautmans I, Beaudart C, Bischoff-Ferrari H, et al. Quality of life in sarcopenia and frailty. Calcif Tissue Int. 2013; 93: 101-120.
- Mitnitski AB, Mogilner AJ, Rockwood K. Accumulation of deficits as a proxy measure of aging. Scientific World Journal. 2001; 1: 323-336.
- Cesari M, Fielding RA, Pahor M, Goodpaster B, Hellerstein M, van Kan GA, et al. Biomarkers of sarcopenia in clinical trials-recommendations from the International Working Group on Sarcopenia. J Cachexia Sarcopenia Muscle. 2012; 3: 181-190.
- Montano-Loza AJ, Meza-Junco J, Prado CM, Lieffers JR, Baracos VE, Bain VG, et al. Muscle wasting is associated with mortality in patients with irrhosis. Clin Gastroenterol Hepatol. 2012; 10: 166-173, 173 e1.
- Lai JC, Feng S, Terrault NA, Lizaola B, Hayssen H, Covinsky K. Frailty predicts waitlist mortality in liver transplant candidates. Am J Transplant. 2014; 14: 1870-1879.
- Etzioni DA, Liu JH, Maggard MA, Ko CY. The aging population and its impact on the surgery workforce. Ann Surg. 2003; 238: 170-177.
- Scarborough JE, Pietrobon R, Bennett KM, Clary BM, Kuo PC, Tyler DS, et al. Workforce projections for hepato-pancreato-biliary surgery. J Am Coll Surg. 2008; 206: 678-684.
- Valero V 3rd, Amini N, Spolverato G, Weiss MJ, Hirose K, Dagher NN, et al. Sarcopenia adversely impacts postoperative complications following resection or transplantation in patients with primary liver tumors. J Gastrointest Surg. 2015; 19: 272-281.
- DiMartini A, Cruz RJ Jr, Dew MA, Myaskovsky L, Goodpaster B, Fox K, et al. Muscle mass predicts outcomes following liver transplantation. Liver Transpl. 2013; 19: 1172-1180.
- Peng PD, van Vledder MG, Tsai S, de Jong MC, Makary M, Ng J, et al. Sarcopenia negatively impacts short-term outcomes in patients undergoing hepatic resection for colorectal liver metastasis. HPB(Oxford). 2011; 13: 439-446.
- Louwers L, Schnickel G, Rubinfeld I. Use of a simplified frailty index to predict Clavien 4 complications and mortality after hepatectomy: analysis of the National Surgical Quality Improvement Project database. Am J Surg. 2016; 211: 1071-1076.
- Montano-Loza AJ, Meza-Junco J, Baracos VE, Prado CM, Ma M, Meeberg G. et al. Severe muscle depletion predicts postoperative length of stay but is not associated with survival after liver transplantation. Liver transplant. 2014; 20: 640-648.
- Sonnenday CJ, Englesbe MJ. Frailty Score Predicts Outcomes Among Liver Transplant Candidates and Recipients. The Liver Meeting. 2013; Washington, D.C.
- Englesbe MJ, Patel SP, He K, Lynch RJ, Schaubel DE, Harbaugh C, et al. Sarcopenia and mortality after liver transplantation. J Am Coll Surg. 2010; 211: 271-278.
- Lee CS, Cron DC, Terjimanian MN, Canvasser LD, Mazurek AA, Vonfoerster E, et al. Dorsal muscle group area and surgical outcomes in liver transplantation. Clin Transplant. 2014; 28: 1092-1098.
- van Vledder MG, Levolger S, Ayez N, Verhoef C, Tran TC, Ijzermans JN. Body composition and outcome in patients undergoing resection of colorectal liver metastases. Br J Surg. 2012; 99: 550-557.
- Voron T, Tselikas L, Pietrasz D, Pigneur F, Laurent A, Compagnon P, et al. Sarcopenia Impacts on Short- and Long-term Results of Hepatectomy for Hepatocellular Carcinoma. Ann Surg. 2015; 261: 1173-1183.
- Eappen S, Lane BH, Rosenberg B, Lipsitz SA, Sadoff D, Matheson D, et al. Relationship Between Occurrence of Surgical Complications and Hospital Finances. Jama-J Am Med Assoc. 2013; 309: 1599-1606.
- Lawson EH, Hall BL, Louie R, Ettner SL, Zingmond DS, Han L, et al. Association between Occurrence of a Postoperative Complication and Readmission Implications for Quality Improvement and Cost Savings. Ann Surgery. 2013; 258: 10-18.
- Zimlichman E, Henderson D, Tamir O, Franz C, Song P, Yamin CK, et al. Health care-associated infections: a meta-analysis of costs and financial mpact on the US health care system. JAMA internal medicine. 2013; 173: 2039-2046.
- Sheetz KH, Waits SA, Terjimanian MN, Sullivan J, Campbell DA, Wang SC, et al. Cost of major surgery in the sarcopenic patient. J Am Coll Surg. 2013; 217: 813-818.
- Robinson TN, Wu DS, Stiegmann GV, Moss M. Frailty predicts increased hospital and six-month healthcare cost following colorectal surgery in older adults. Am J Surg. 2011; 202: 511-514.
- Bergerson JT, Lee JG, Furlan A, Sourianarayanane A, Fetzer DT, Tevar AD, et al. Liver transplantation arrests and reverses muscle wasting. Clin Transplant. 2015; 29: 216-221.
- Kamath PS, Kim WR; Advanced Liver Disease Study G. The model for end-stage liver disease (MELD). Hepatology. 2007; 45: 797-805.
- Harimoto N, Shirabe K, Yamashita YI, Ikegami T, Yoshizumi T, Soejima Y, et al. Sarcopenia as a predictor of prognosis in patients following hepatectomy for hepatocellular carcinoma. Br J Surg. 2013; 100: 1523- 1530.
- Meza-Junco J, Montano-Loza AJ, Baracos VE, Prado CM, Bain VG, Beaumont C, et al. Sarcopenia as a prognostic index of nutritional status in concurrent cirrhosis and hepatocellular carcinoma. J Clinical Gastroenterol. 2013; 47: 861-870.
- de Vries NM, Staal JB, van Ravensberg CD, Hobbelen JS, Olde Rikkert MG, Nijhuis-van der Sanden MW. Outcome instruments to measure frailty: a systematic review. Ageing Res Rev. 2011; 10: 104-114.
- Mathur S, Janaudis-Ferreira T, Wickerson L, Singer LG, Patcai J, Rozenberg D, et al. Meeting report: consensus recommendations for a research agenda in exercise in solid organ transplantation. Am J Transplant. 2014; 14: 2235-2245.
- Velanovich V, Antoine H, Swartz A, Peters D, Rubinfeld I. Accumulating deficits model of frailty and postoperative mortality and morbidity: its application to a national database. J Surg Res. 2013; 183: 104-110
- Kaido T, Ogawa K, Fujimoto Y, Ogura Y, Hata K, Ito T, et al. Impact of sarcopenia on survival in patients undergoing living donor liver transplantation. Am J Transplant. 2013; 13: 1549-1556.
- Krell RW, Kaul DR, Martin AR, Englesbe MJ, Sonnenday CJ, Cai S, et al. Association between Sarcopenia and the Risk of Serious Infection Among Adults Undergoing Liver Transplantation. Liver Transplant. 2013; 19: 1396-1402.
- Masuda T, Shirabe K, Ikegami T, Harimoto N, Yoshizumi T, Soejima Y, et al. Sarcopenia is a prognostic factor in living donor liver transplantation. Liver Transpl. 2014; 20: 401-407.