Research Article
Burn Scar Regeneration with the “SUFA” (Subcision and Fat Grafting) Technique - A Prospective Clinical Study
Gargano F1*, Arelli F2, Yfan Guo3, Schmidt S3, Peter Evangelista M4, Robinson-Bostom L5, Harrington DT6 and Liu P3
1Division of Plastic Surgery, St. Joseph’s Medical Center, USA
2Department of Surgery, Azienda Ospedaliera San Camillo-Forlanini, Italy
3Division of Plastic Surgery, Warren Alpert Medical School of Brown University, USA
4Department ofRadiology, Warren Alpert Medical School of Brown University, USA
5Department of Dermatology, Warren Alpert Medical School of Brown University, USA
6Department of Surgery, Warren Alpert Medical School of Brown University, USA
*Corresponding author: Francesco Gargano, Division of Plastic Surgery, St. Joseph’s Medical Center, 703 Main street Paterson, NJ 07503, USA
Published: 13 Oct, 2016
Cite this article as: Gargano F, Arelli F, Guo Y, Schmidt S,
Peter Evangelista M, Robinson-Bostom
L, et al. Burn Scar Regeneration with
the “SUFA” (Subcision and Fat Grafting)
Technique - A Prospective Clinical
Study. Clin Surg. 2016; 1: 1151.
Abstract
Background: Treatment of burn scars with traditional surgical techniques is challenging due to recurrent contractures. The use of fat grafting in thermal injury has been previously reported only
in small clinical series and results are often biased by simultaneous surgical procedures and lack of
scientific methods of validation.
Methods: Our study prospectively evaluates outcomes in 9 patients treated with the “SUFA”
technique (Subcision and Fat Grafting) for debilitating contracted burn scars limiting range of
motion. Results are evaluated clinically with the Vancouver scale and by range of motion through
the affected joints at 1, 3, 6 and 12 months. Scientific validation of the outcomes is performed
evaluating dermal thickening and scar remodeling by high definition ultrasound and histology
examination with hematoxylin-eosin and monoclonal antibodies staining.
Results: Results show clinical improvement, thickening of dermis and redistribution and
reorientation of the collagen fibers within the dermis. Statistical significance (p< 00.5) has been
obtained for all analyzed data. Fat reabsorption occurred with a mean of 40%.
Conclusion: Our study gives scientific validation of the efficacy of subcision and fat grafting in
contracted scar. New surgical and diagnostic techniques are illustrated. Our clinical and diagnostic
outcomes suggest dermis regeneration secondary to the new fat grafting technique.
Keywords: Burn scar; Subcision and fat grafting; Regeneration
Introduction
Reconstruction of burn scars remains one of the most challenging aspects of reconstructive
surgery. The debilitating functional problems, cosmetic deformities and frequent lack of autologous
tissue to replace “like with like” demand that therapeutic efforts are well planned and executed.
Conventional methods of treatment of acute burns include primary excision and grafting of deep
second-degree and full-thickness burns [1,2]. Scar contraction can be decreased by early excision
and grafting, but it is always present to some degree [3].
Current methods of scar contracture releases include non-surgical and surgical methods. Nonsurgical
methods use corticosteroids injection, silicone gels, pressure dressing, physiotherapy, and
laser therapy [4,5]. Surgical correction can be carried out by excision and local tissue rearrangement
such as z-plasties, flaps, tissue expansion and skin grafting [6-8]. With all the above surgical
treatments the final result is to substitute a scar with another scar that will inevitably be prone to a
new contracture.
Fat grafting in burn clinical studies have been conducted on small subsets of patients and
outcomes have been evaluated using functional and aesthetic parameters of assessment that often
lack scientific validation [9-18]. The goal of our study is to advocate fat grafting as an alternative
method to skin graft or flap transfer in burn reconstruction. We prospectively evaluate outcomes
in patients who underwent the “SUFA” technique (Subcision and Fat Grafting) for the treatment of
contracted burn scars limiting range of motion. Clinical and scientific
validated methods are employed to evaluate the effect of fat grafts
on scar remodeling. In particular, a non-invasive and cost-effective
method of evaluation of fat graft take and dermal thickness, namely
ultrasound, is introduced. Dermal “regeneration” is assessed with the
use of monoclonal antibody staining.
Material and Methods
Our IRB approved study prospectively analyzed 9 consecutive
patients affected by secondary burns over a period of 24 months. A
detailed consent form illustrating the procedure risk and benefits was
signed before the surgery. Criteria of inclusion were patients with
contracted scar bands in flexion creases such as the face and neck
(n=6), upper extremity and axilla (n=3), lower extremities (n=2),
perineum (n=1). All analyzed scars limit range of motion of the
affected joints. Other patients with burn scars were non-functional
impeding areas are not included in the study. Patient demographics
showed seven males and two females, mean age 46 years, mean time
since primary burn was 6 years (Table 1).
All patients included in the study have been initially treated
with skin grafts and then with scar release, skin graft or z-plasties for
secondary contractures. The SUFA technique was always performed
after the initial stage of skin grafting or z-plasty. Scars have been
clinically evaluated with the Vancouver scale and range of motion
preoperatively, at 1, 3, 6, and 12 months postoperatively. The
Vancouver scale includes scar pigmentation, vascularity, pliability
and height preoperative and at follow ups (Table 2).
Fat graft survival and skin remodeling have been evaluated by
high frequency 18MHz ultrasound [19] preoperatively, at 1, 3, and
6 months postoperatively. 7.5-13 MHz and 10-20 MHz probes were
used. In selected patients, the dermal remodeling and regeneration
has been demonstrated by histologic examination of scar tissue
biopsies with hematoxylin-eosin and rabbit monoclonal antibodies
staining for collagen type I and III before and after treatment.
Antibodies used were RabMab anti-collagen I antibody (Abcam,
ab138492) and Rabbit Anti-Collagen III antibody (Abcam, ab7778).
Both ultrasound and biopsy have been performed at fixed anatomic
landmarks to facilitate comparison and avoid bias. Operation time,
tumescent infusion, lipoaspirate and injected fat have been recorded.
We have taken pre and postoperative pictures and videos at 1, 3, 6,
and 12 months follow up intervals. We used a goniometer to assess
range of motion of the affected joints. Statistical of data have been
analyzed by Student t-test.
Postoperative protocol included taping of the injected areas for 48
hours, oral antibiotics for three days and sutures removal at 1 week.
Stretching exercises were started at postoperative day 5. Between
surgical sessions, an interval of 2 to 3 months was respected to allow
graft take and remodeling. A maximum of three sessions have been
employed in some patients.
Table 1
Table 2
Figure 1
Figure 2
Figure 3
Figure 4
Figure 5
Table 3
Figure 6
Technique
We have evaluated scar contracture and range of motion in the
flexion creases preoperatively. We marked the scar bands we planned
to be released. All patients were operated on under general anesthesia
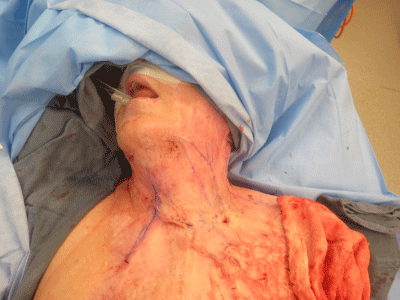
and positioned on the operative table with the contracted cords under
tension to facilitate intra-operative release (Figure1).
The abdomen represented the donor site for fat harvesting in all
patients. Tumescent solution was infiltrated with 2.7-mm, 12 hole
cannula, through intra-umbelical incisions to minimize scarring.
After 15 minutes of waiting time for the vasoconstrictive effect of the
epinephrine, the fat was aspirated with 60 cc syringes under negative
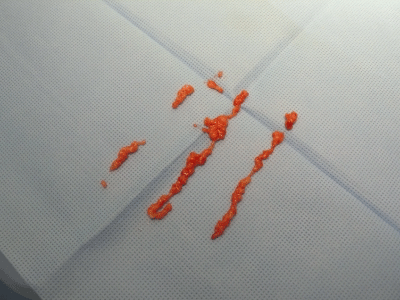
pressure. The lipoaspirate was first washed with ringer lactate in a
proportion of 1:1 and then processed on telfa (Figure 2). Fat graft was
inserted in 10 cc syringe with a traumatic technique: the preparation
of the fat was performed on telfa in longitudinal rows facilitated reinsertion
into the syringe cylinder while exerting a wiggling motion
with the syringe.
The recipient areas were infiltrated with lidocaine 1% with
epinephrine 1:100.000 and vasoconstriction was allowed waiting for
15 minutes. The subcision of the scar cords was performed with18G
Admix needle with cutting tip while the cord is under maximum
tension (BD NokorTM) (Figure 3).
The subcision technique included parallel repetitive longitudinal
passes of the cutting needle through the cords. At the end the
remaining scar adherences were released with 45 to 60-degree
oscillatory motion of the needle blade. The release generates short slit
in the scar, which results in loosing and expansion (Video 1). In this
way a tridimensional donor site scaffold was created. Fat was injected
with 2.5 mm lipofilling cannulas in a three dimensional pattern with
0.3 ml of fat injected at each pass (Figure 4). The total amount of fat
was estimated during the preoperative planning and recorded for
each anatomic subunit. When the face was the area to be treated,
fat was injected mainly at the jaw line to minimize postoperative
disfiguration in case of weight gain. The treatment of burn hands
took into consideration the location of structures to be preserved.
Webspaces were released and fat grafted. The dorsum of the hands
was subcised between dermis and extensor tendons and then fat
grafted in the released space. V-shape cannulas were at times used to
cut more tenacious bands.
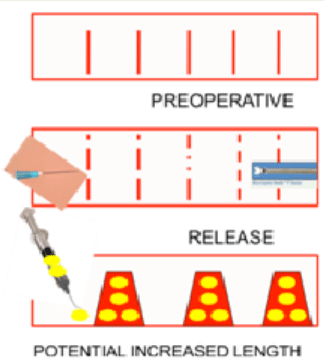
The SUFA technique allowed release and expansion of the scar
tissues with multiple micro-fractures in the offending scar bands
that were filled by fat injection in a three dimensional scaffold. This
elongated the scar of almost 20% at each session (Figure 5).
Results
Mean operative time was 70 minutes (range 45 to 180 minutes),
mean tumescent fluid infiltrated and fat aspirated was 100cc and
mean injected fat grafts 45 cc (range 30cc to 75cc) (Table 3).
All patients showed statistically significant improvements in scar
contractures in the Vancouver scale and for range of motion of the
head, neck, hands, lower extremities, and perineum at 3 and 6 months
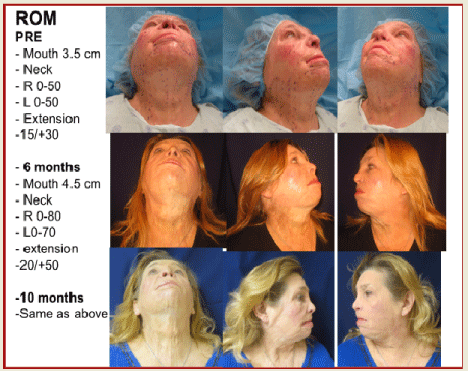
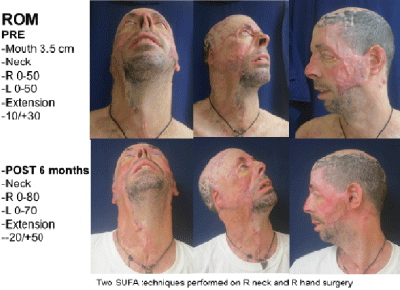
follow ups (P< 0.005) (Table 4, Figure 6 and Videos 2-5).
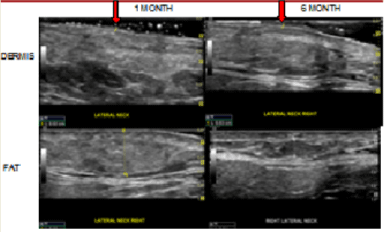
High frequency ultrasound showed mean fat reabsorption of
approximately 40% and increased dermal thickness at three and
six month’s controls (Table 5 and Figure 7). Anatomical bias was
eliminated performing the high frequency ultrasound and biopsies
at fixed anatomic landmarks. Only one radiologist performed the
ultrasounds and one surgeon performed the biopsies.
The histology staining with rabbit monoclonal antibodies for
collagen type I and III showed increased deposition of the collagen
III after the fat grafting and remodeling of the collagen type III fibers
with regular orientation as in mature healing dermis (Figure 8 and 9).
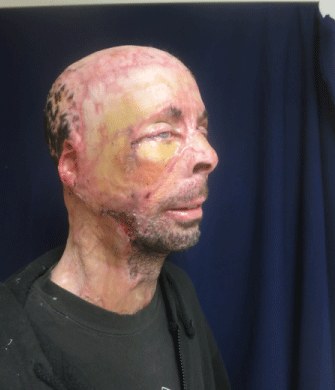
One patient had skin breakdown resulting in uneventful healing
(Figure 10). No donor site morbidity was noticed. All patients were
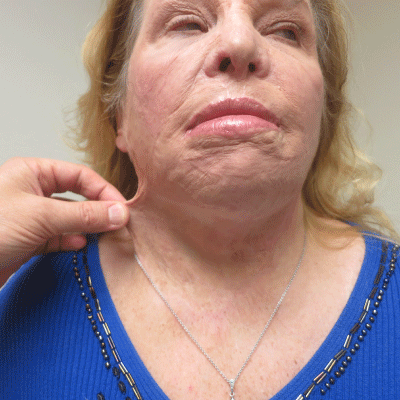
followed up to assess range of motion and functional improvements
were achieved (Figure 11 and 12).
Table 4
Table 5
Table 5
High frequency ultrasound showed mean fat reabsorption of approximately 40% and increased dermal thickness at three and six months controls.
Figure 7
Figure 7
High frequency ultrasound shows fat reabsorption of approximately
40% and increased dermal thickness.
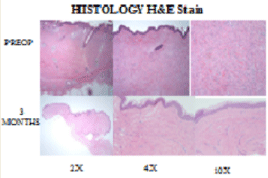
Figure 8
Figure 8
Hematoxilin eosin staining. Preoperative: The lowest power (2x) illustrates dermal thickening and fibrosis, with disruption of hair follicles. On higher power (4x) you can appreciate the fibroblastic proliferation and fibrosis, with focal keloidal collagen bundles (which are highlighted in the 10x picture). Postoperative: 2x and 10x shows scar formation without hypertrophic scar and keloid formation. Collagen fibers have a horizontal orientation wo bundle formation, decreased fibroblasts.
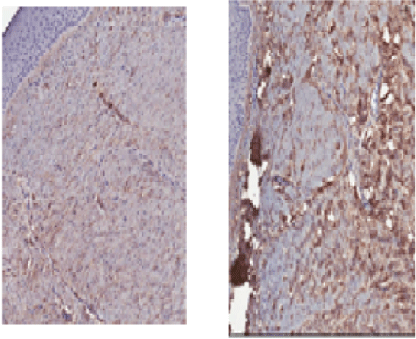
Figure 9
Figure 9
Staining with monoclonal antibody shows increased deposition of the collagen III after the fat grafting. RabMab anti-collagen I antibody (Rabbit monoclonal to Collagen I), Abcam, ab138492. Rabbit Anti-Collagen III antibody (Rabbit polyclonal to Collagen III), Abcam, ab7778.
Figure 10
Discussion
Principles of burn reconstruction include understanding of
wound healing and contracture processes. The effect of time on the
maturation of scars is of pivotal importance and requires expertise on
choosing the correct time for surgery. Multiple operations are the rule
and frequently take place over a long period of time. Over the past
50 years, primary excision and grafting of deep second-degree and
full-thickness burns has become the standard of care in the United
States and in most developed countries [1-2]. All burns of the second
and third degree result in open wounds and heal by contraction and
epithelialization. Contraction is dependent from degree of burn and
time elapsed between injury and surgical intervention. Therefore
contracture can be decreased by early excision and grafting, but it is
always present to some degrees [3]. Contraction leads to tension, and
tension is one of the principal causes of hypertrophic and contracted
scars. Understanding the role of tension and lack of healthy tissues in
the evolution of post-burn deformities is essential to their successful
treatment. Burn reconstruction is fundamentally about the release of
contractures and the correction of contour deformities. It should not
be focused on the excision of burn scars because a scar can only be
traded for another scar of a different variety. In burns the fundamental
problem is the inadequate skin and soft tissues and further excision
of scars, skin graft or Z plasties can easily add to the clinical problem
of worsening contraction. Non-surgical methods of scar release such
as pressure therapy, physical therapy, cortisone injection and laser
therapy are often disappointing. Surgical corrections of contracted
scars can be carried out by excision and reconstruction with local
tissue rearrangement such as Z-plasties, flaps, tissue expansion,
skin grafting. All the above mentioned surgical methods carry the
potential of lack of tissue, “borrowing from Peter to give Paul” and
the event of a new contracture. For the aforementioned reasons, we
think that subcision and fat grafting represents a worthy approach
with remodeling of the scar form inside minimizing the risk of a new
contracture.
The regenerative role of fat grafting in burns has been validated
by in vitro studies. Revascularization decreased scarring and fibrosis,
increased dermis thickening with rearrangement of the collagen fibers
within the dermis has been proven in murine models [20-23]. Despite
the in vitro models positive outcomes, the clinical series reported in
the literature lack of scientific validation or are biased by concurrent
therapies [9-18,24,25-27]. Published data suggest that degree of fat
resorption ranges between 30 and 40% and our data overlaps these
findings [14]. Khouri et al. [25] recently published their technique of
percutaneous aponeurotomy and fat grafting. We describe in detail
the technique learned initially from Dr. Gino Robotti and then refined
by Dr. Khouri R [25]. I personally learned the same technique from
Dr. Khouri [25] and then modified it in some technical details. We
use 18 G needle with sharp pointy blade at its tip because we think this
has more control during the subcision of the scars. Our choice for the
“wash out” fat preparation technique is due to the more long lasting
survival achievable with this technique [26]. Another difference from
previous publications is also that our clinical outcomes have been
evaluated with objective data and measurements with statistically
significant improvement for range of motion. Scientific validation
of the “regenerative effect” of fat grafting has been demonstrated
by high definition ultrasound and histology changes within the
dermis. Our study is the first to validate clinically what has been
previously demonstrated only by in vitro studies [11,17,21,23]. In
fact, ultrasound evaluation with thickening of the dermis at three and
six months and histology findings of rearrangement of the collagen
type III fibers within the dermis confirm what has been previously
demonstrated in the murine models [21].
Lack of soft tissue and burn scar contracture after Z plasties
suggest that we should aim for an “internal remodeling” of the scar
without jeopardizing the already scant and altered skin envelopes
[25]. Our clinical experience suggest that the “SUFA” technique
maintain and improve the range of motion over time when compared
to traditional “external remodeling” techniques.
Our results show that the effects of subcision and fat grafting is
probably a combined and synergic effect of mechanical expansion
(subcision of scar) and fat grafting (dermal regeneration) and
certainly the prevention of scar contracture is due to the fat that could
act as a “spacer” or stem cell induction for regeneration.
The “SUFA technique” cannot be considered effective at 100% in
all cases, especially when the scars are thick and extremely limiting
range of motion. In these instances we use the technique for a
maximum of three times at three months intervals and then release
the tenacious contraction with a “prefabricated Z-plasty” flap. The
prefabrication of the flaps with fat injection softens the scars, gives
adequate padding and limits the scar contracture of the transposed
flaps. The prefabrication of the Z-plasty with fat graft also allows
better viability, lengthening and reduction of contractures.
Our study will aim in the future to include patients in the very
early stages of burns to try to modify the natural history of the
retracting scars.
Figure 11
Figure 12
Conclusion
The “SUFA” technique represents a new promising approach in the era of “regeneration” of the connective tissues. In our study, improvements in clinical outcomes are supported by validating scientific methods such as high definition ultrasounds and histology with monoclonal antibody staining for collagen type I and III. Collagen III regeneration and reorientation is probably promoted by adipose tissue-derived stem cells.
References
- Cope O, Langohr JL, Moore FD, Webster RC. Expeditious care of full thickness burn wounds by surgical excision and grafing. Ann Surg. 1947; 125: 1-22.
- Janzekovic A. A new concept in early excision and immediate grafting of burns. J Trauma. 1970; 10: 1103-1108.
- Burke J, Bondoc CC, Quinby WC. Primary burn excision and immediate grafting: a method for shortening illness. J Trauma. 1974; 14: 389-395.
- Waibel J, Beer K. Fractional laser resurfacing for thermal burns. J Drug Dernatol. 2008; 7: 59-61.
- Waibel J, Beer K. Ablative fractional laser resurfacing for the treatment of a third degree burn. J Drug Dernatol. 2008; 8: 294-297.
- Larson D, Abston S, Evans DB, Dobrkovsky M, Linares HA. Techniques for decreasing scar formation and contractures in the burn patient. J Trauma. 1971; 11: 807-823.
- Ahn ST, Monafo WW, Mustoe TA. Topical silicone gel: a new treatment for hypertrophic scars. Surgery. 1989; 106: 781-786.
- Spence RJ. Management of facial burns. In: Neligan P, editor. Plastic Surgery. Elsevier. 2013: 468-499.
- Ranganathan K, Wong VC, Krebsbach PH, Wang SC, Cederna PS, Levi B. Fat grafting for thermal injury: current state and future directions. J Burn Care Res. 2013; 34: 219-226.
- Brongo S, Nicoletti GF, La Padula S, Mele CM, D′Andrea F. Use of lipofilling for the treatment of severe burn outcomes. Plast Reconstr Surg. 2012; 130: 374e-376e.
- Sultan SM, Barr JS, Butala P, Davidson EH, Weinstein AL, Knobel D, et al. Fat grafting accelerates revascularisation and decreases fibrosis following thermal injury. J Plast Reconstr Aesthet Surg. 2012; 65: 219-227.
- Viard R, Bouguila J, Voulliaume D, Comparin JP, Dionyssopoulos A, Foyatier JL. Fat grafting in facial burns sequelae. Ann Chir Plast Esthet. 2012; 57: 217-229.
- Clauser LC, Tieghi R, Galiè M, Carinci F. Structural fat grafting: facial volumetric restoration in complex reconstructive surgery. J Craniofac Surg. 2011; 22: 1695-1701.
- Klinger M, Marazzi M, Vigo D, Torre M. Fat injection for cases of severe burn outcomes: a new perspective of scar remodeling and reduction. Aesthetic Plast Surg. 2008; 32: 465-469.
- Caviggioli F, Klinger F, Villani F, Fossati C, Vinci V, Klinger M. Correction of cicatricial ectropion by autologous fat graft. Aesthetic Plast Surg. 2008; 32: 555-557.
- Foyatier JL, Comparin JP, Boulos JP, Bichet JC, Jacquin F. Reconstruction of facial burn sequelae. Ann Chir Plast Esthet. 2001; 46: 210-226.
- Wetterau M, Szpalski C, Hazen A, Warren S. Autologous Fat Grafting and Facial Reconstruction, J Craniofac Surg. 2012; 23: 315-318.
- Patel N. Fat injection in severe burn outcomes: a new perspective of scar remodeling and reduction. Aesthetic Plast surg. 2008; 32: 470-472.
- Shung KK. High frequency ultrasonic imaging. J Med Ultrasound. 2009; 17: 25-30
- Rigotti G, Marchi A, Sbarbati A. Adipose-derived mesenchymal stem cells: past, present, and future. Aesthetic Plast Surg. 2009; 33: 271-273.
- Mojallal A, Lequeux C, Shipkov C, Breton P, Foyatier JL, Braye F, et al. Improvement of skin quality after fat grafting: clinical observation and an animal study. Plast Reconstr Surg. 2009; 124: 765-774.
- Mojallal A, Shipkov C, Braye F, Breton P, Foyatier JL. Influence of the Recipient Site on the Outcomes of Fat Grafting in Facial Reconstructive Surgery. Plast Reconstr Surg. 2009; 124: 471-483.
- Thanik VD, Chang CC, Lerman OZ, Allen RJ Jr, Nguyen PD, Saadeh PB, et al. A murine model for studying diffusely injected human fat. Plast Reconstr Surg. 2009; 124: 74-81.
- Klinger M, Caviggioli F, Klinger FM, Gianassi S, Bandi V, Banzatti B, et al. Autologous fat grafting in scar treatment. J Cran Surgery. 2013; 24: 1610- 1615.
- Khouri RK, Smit JM, Cardoso E, Pallua N, Lantieri L, Mathijssen IM, et al. Percutaneous aponeurotomy and lipofilling: a regenerative alternative to flap reconstruction? Plast Reconstr Surg. 2013; 132: 1280- 1290.
- Smith P. Adams WP Jr, Lipschitz AH, Chau B, Sorokin E, Rohrich RJ, et al. Autologous human fat grafting: effect of harvesting and preparation techniques on adipocyte graft survival. Plast Reconstr Surg. 2006; 117: 1836-1844.
- Piccolo NS, Piccolo MS, Piccolo MT. Fat grafting for treatment of burns, burn scars and other difficult wounds”. Clinics in plastic surgery. 2015, 42: 263-283.