Research Article
Diagnostic and Prognostic Role of Stromal CD 10 and Ki 67 in Benign and Malignant Phylloides Tumor of Breast
Shailendra Kumar Gupta1, Devendra Singh1, Surender Kumar1* and Singh VK2
1Department of General Surgery, King George’s Medical University, India
2Department of Community Medicine, King George’s Medical University, India
*Corresponding author: Surender Kumar, Department of General Surgery, King George’s Medical University, Lucknow, Pin Code: 226003, India
Published: 05 Oct, 2016
Cite this article as: Gupta SK, Singh D, Kumar S, Singh
VK. Diagnostic and Prognostic Role of
Stromal CD 10 and Ki 67 in Benign and
Malignant Phylloides Tumor of Breast.
Clin Surg. 2016; 1: 1150.
Abstract
Background: Phylloides tumors (PT) are rare fibro epithelial tumor of breast, closely resemble to breast fibroadenoma, ranging from benign to malignant tumor. PT has potential to recur and metastasize due to its stromal characteristics. Differentiation between benign and malignant tumor is not possible with histopathology alone, additional study of stromal CD 10 and Ki 67 antigen expression in PT may be useful to differentiate benign from malignant tumor.
Method: Immunohistochemical study using specific monoclonal antibody to label stromal CD 10 and Ki 67 antigen were done in tissue sample of 35 patients.
Results: Out of total 35 patients histopathologically 18 were benign, 9 borderline and 8 were malignant PT. On immunohistochemistry 16 patients had CD 10 expression labeled as CD 10 positive (7 malignant, 7 borderline and 2 benign), and 19 were CD 10 negative (1 malignant, 2 borderline and 16 benign) (p< 0.000). Overall sensitivity, specificity, PPV and NPV to diagnose malignant PT was 82.35%, 88.88%, 77.77% and 94.11% respectively. 23 patients had Ki 67 labeling index between 1-8 (18 benign, 5 border line) and 12 patients had index > 8 (8 malignant, 4 borderline) i.e. no one malignant PT had Ki 67 labeling index < 8 (p < 0.000). Sensitivity, specificity, PPV and NPV for diagnosing malignant PT were 100%.
Conclusion: Stromal CD 10 and Ki67 expression is higher in malignant PT. Ki 67 labeling index
>8 are strongly suggestive of malignant PT and needs to appropriate treatment and close follow-up
even in histopathologically benign tumor.
Keywords: Phylloides tumor; CD 10, Ki 67; Immunohistochemistry; Histopathology
Introduction
Phylloides tumors (PT) are unique in combining sarcoma with a benign epithelial component,
having varying malignant potential ranging from benign to completely malignant tumor. PT is
a uncommon fibroepithelial tumor of breast, accounting 0.3% -0.5% of all breast neoplasm with
incidence of 2.1 per million. Age of presentation is one decade earlier to breast carcinoma and one
decade later to fibro adenoma with peak age of 45 to 49 years [1,2]. On the basis of histological
behavior WHO classified PT into, benign, borderline malignant and malignant category [3]. Distant
metastasis is rare but local recurrence can occur uncommonly which is not related to histological
behavior of tumor as benign tumors can also recur [4-6].
Diagnosis of phylloides tumor can be difficult on histopathological characteristics alone because
it closely resemble to fibroadenoma [7] therefore an additional study of genetic changes that
characterize the course of Phylloides tumor may have additional prognostic significance. Advances
in immunohistochemical and molecular methods have shed light on the biological nature of the
neoplasm. Molecular markers have been extensively investigated with a view to providing early and
accurate information on long-term outcome and prediction of response to treatment of early breast
cancer.
Ki 67 is a proliferation marker, its expression are considered useful predictor of tumor
progression and cancer prognosis. In this study we also study CD 10 is a member of the family of
matrix metalloproteases which in general, function to degrade matrix adhesions rendering invasive
and metastatic property to any neoplasm. To date very few study correlated the expression of Ki
67 with CD 10 and also correlation of these markers with histopathological grading and with other
clinical parameters.
Aims and objectives
The aim of study is to evaluate the difference of immunophenotype
of the Stromal cells and cells proliferation marker in the Phylloides
tumors, and to evaluate the differential feature between benign and
malignant Phylloides tumors. In this study we compare diagnostic
and prognostic role of Stromal CD10 and Ki67 and expression in
benign and malignant Phylloides tumor of breast and correlate with
known clinical and histopathologically prognostic factors.
Figure 1
Figure 2
Figure 2
Diagrammatic presentation of CD 10 expression in benign,
borderline and malignant phylloides tumor.
Figure 3
Figure 4
Figure 5
Materials and Methods
Paraffin fixed tissue samples of 35 patients were sent for
histopathological and Immunohistochemical study, where
hematoxylin and eosin staining slides prepared and histopathological
study were performed. Histopathologically diagnosed benign,
borderline and malignant PT of breast was taken. Positive tissue
control (Fibroadenoma for CD 10 and Lymphoma for Ki 67) and
negative control (Omission of primary antibody) were taken.
For Immunohistochemistry Streptavidin Biotin
immunoperoxidase method was done. Staining and evaluation using
specific monoclonal antibody to CD 10 and Ki 67 was done as per
standard protocol, the tumor was considered positive for CD 10, if
the Stromal cells were moderate to strong staining intensity in 20% or
more of the Stromal cells.
Ki 67 labeling index is defined as the percentage of cells that
showed a positive nuclear stain. At least 1000 stromal cells were
counted for this analysis, and all Ki 67 indices were determined by
one observer.
Statistical analysis and data Interpretation
For data interpretation appropriate statistical test like chi-square,
ANOVA, Fischer`s `t` Test has been considered and results were
drawn on the basis thorough statistical analysis using SPSS 16.0
(IBM) software.
Observation and results
Following results were obtained from study, out of 35 patients
18 were benign, 9 were borderline malignant and 8 were malignant
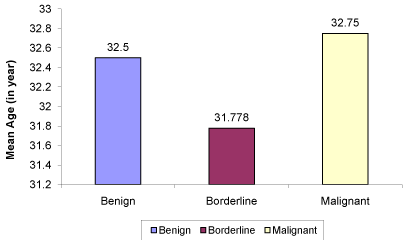
phylloides tumor. . The mean age of patient of phylloides tumor
was 32.37 years, with a range being 13 to 53 years. No significant
differences between mean age (32.5 vs. 31.778 vs. 32.75, p >0.05) of
benign, borderline malignant and malignant Phylloides tumor was
noted.
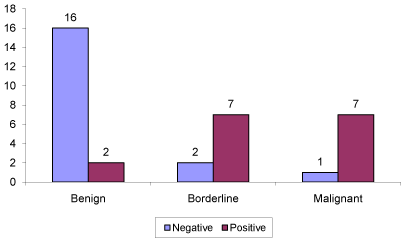
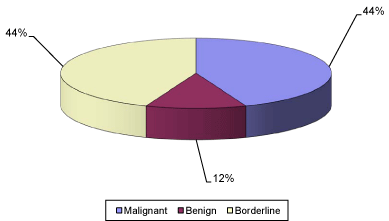
Out of 16 CD 10 positive patients seven were of malignant
histology (7/16, 44%), seven were of borderline (7/16, 44%), and
only 12% of the patients were diagnosed as benign on histopathology
examination (p=0.000). Over all sensitivity and specificity of CD 10
to diagnose malignant potential was 82.35 % and 88.88% respectively,
however independent sensitivity and specificity of CD 10 for assessing
malignant potential of tumor 87.50% and 88.88% respectively.
Diagnostic accuracy of test for combined borderline and malignant
tumor was 85.71%, while only for malignant tumor was 88.46%. PPV
is 77.77% and NPV 94.11 % observed in our study.
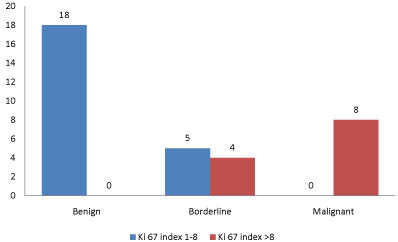
Out of 35 patients 23 patients had Ki 67 index between 1-8
(benign 18, borderline 5), and 12 patients had Ki 67 index >8
(borderline 4, malignant 8). Patients of benign phylloides tumor
had Ki 67 proliferation index < 8 as compared to malignant and
borderline malignant tumor (p = 0.000), as similarly malignant
phylloides tumor had Ki 67 proliferation index >8 as compared to
benign histology (p = 0.000) ). Sensitivity and Specificity of test for
malignant phylloides tumor alone were found 100%. Fisher exact `t`
test did not showed significant difference (p >0.05) in between CD10
and Ki 67 for diagnosing a malignant phylloides tumor.
As the diameter of phylloides tumor increases positivity of CD-10
expression increases. (p< 0.05). Out of 16 CD 10 positive cases only
4 patients had a longest diameter of tumor < 5 cm, while 7 out of 19
CD 10 negative cases have diameter >5 cm. Analytical study did not
show linear association between Ki 67 Index and longest diameter of
phylloides tumor (p >0.05).
No relationship was observed in our study between palpable
lymph nodes and expression of Immunohistochemical marker CD 10
and Ki 67 (p >0.05). No metastasis was found in any of our patients.
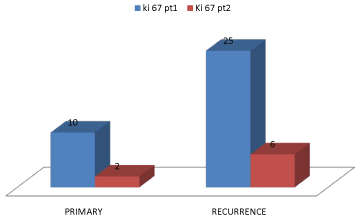
In 3 recurrent cases included in our study the CD 10 expression
remained same in both primary and recurrent diseases, while Ki
67 expression increased in recurrent tumors as compared to their
primary counterpart; however a sample size of two or three patients
is too small to make a universally acceptable hypothesis and to
formulate treatment guidelines.
Those two patients who present with recurrence , primary tumor
is treated by local excision and one patient whose primary record
not available, all these three patients with recurrence are now treated
with modified radical mastectomy. There is neither history, nor
any metastatic work-up like USG ABDOMEN, X-RAY, and LFT
suggestive of distant metastasis present in all 35 patients of phylloides
tumor.
Table 1
Table 1
Descriptive analysis of age of different histopathological groups of phylloides tumor (1 Benign, 2 Borderline malignant, 3 Malignant).
Table 2
Table 2
Expression of CD 10 out of 35 patients 16 patients had CD 10 expression were labeled as CD 10 Positive, 19 patients failed to express CD 10 were labeled as CD10 Negative.
Table 3
Table 4
Table 5
Discussion
Phylloides tumors are rare neoplasm that may poses difficulty
in predicting the clinical outcome on merely histological evaluation
of tumour tissue. WHO [3] and Azzopardi and Salvadori et al. [1,8]
classified phylloides tumor into benign, borderline malignant and
malignant groups on the basis of histological features. Hart et al and
Kim et al. [9-10] preferred benign and malignant without borderline
type. Azzopardi et al. [1] suggested that there is no difference between
borderline and malignant tumor in terms of local and distant relapse
and the same hypothesis was also followed in our study however,
local recurrence may occur in all categories of phylloides tumor [4].
CD 10 is a zinc dependent metalloproteinase that cleave the
protein component of extracellular matrix and play a major role in
tissue remodeling. Being a member of metalloproteinase family it is
possible that increased CD 10 expression may facilitate the metastatic
potential of a tumor and may also potentiate the vascular invasion
[11].
One of the largest Study done by Tse et al. [12] from China in
181patients of phylloides tumor using positive staining of stromal CD
10 as a diagnostic criteria showed 5.88% (6/102) to be benign, 31.37%
(16/32) as borderline and 50% (14/28) as malignant phylloides tumor.
The study overall had a specificity of 95%, positive productive value
(PPV) of 81%, sensitivity of 38%, negative productive value (NPV)
72% and an accuracy of 74%.
Similar results have been found in our study in which only 2 out
of 18 benign phylloides tumor were reactive (11.11%), seven out of
9 borderline patients were CD 10 positive (77.77%), while from 8
malignant phylloides tumor cases seven were CD10 positive (87.5%).
The CD 10 expression as a diagnostic test had a sensitivity of 82.35 %
for borderline malignant and malignant tumor taken together and a
sensitivity of 87.5 % for malignant tumor alone. The Specificity being
88.88% in both borderline malignant and malignant taken combindly
and in considering malignant tumor alone i.e. excluding borderline
tumor. Diagnostic accuracy of test for combined borderline and
malignant tumor was 85.71% and 88.46 % for malignant tumor
alone. The PPV and NPV were 77.77% and 94.11 % respectively. CD
10 expression was significantly high in case of malignant phylloides
tumor as compared to benign (p< 0.000). Similarly CD 10 expression
was significantly high in larger tumors as compare to smaller one (p<
0.05) with eight of ten patient with diameter >10 cm tested positive
for CD 10,while only 4 of 11 patients being positive in tumors less
then 5cms in size. Similar association of CD 10 expression with
increasing age has been observed by Tse et al [12].
Ki 67 is a cell proliferation related protein that can be labeled
with monoclonal antibody MIB-1, also known as MK 167. The Ki 67
labeling index is the number of cells that express immunostaining for
Ki 67. Its expression is present in active phases of cells and absent
in resting cells. The fraction of Ki 67 positive tumor cells i.e. Ki 67
labeling index is often correlated with the clinical course of cancer.
Several published series show that expression of proliferative
antigen Ki 67 when correlated to tumor grading had a higher Ki 67
index in malignant tumor as compared to benign histology. Noronha
et al. [13] of Loma Linda, USA study to evaluate the expression of
CD34, CD117 (c-kit), and Ki-67 in PT of the breast and attempt
to correlate the staining pattern with tumor grade by morphology.
Although most benign PTs (80.6%) showed a Ki-67 of < 2%, a few
cases showed slightly higher proliferation indices.
Similar results have been observed in our study in which out of
35 patients 23 had Ki 67 index between 1-8 (benign 18, borderline 5),
6 patients had Ki 67 index between 9-16 (borderline 2, malignant 4)
and 6 patient also had Ki 67 index >16 (borderline 2, malignant 4),
Histopathologically benign tumor had Ki 67 index < 8. (p< 0.000).
Not even a single patient of malignant phylloides tumor had Ki 67
index < 8(p< 0.000). Sensitivity and specificity for group including
both borderline malignant and malignant phylloides tumor was
70.58% and 100%. While the sensitivity and specificity to diagnose
malignant phylloides tumor alone was 100%. All the subject of our
study were divided into two groups, Ki 67 index less than 8 was
labeled as benign and Ki 67 index more than 8 labeled as malignant,
taking this presumption, the diagnostic accuracy of test for combined
borderline malignant and malignant tumor was 88.46% and for
malignant phylloides tumor alone was 100%. PPV and NPV of test
for combined borderline malignant and malignant tumor and for
malignant phylloides tumor alone was 100%. No significant difference
was observed in the expression of Ki 67 in relation to longest diameter
of tumor (p >0.05). Fisher exact `t` test did not showed significant
difference (p >0.05) in between CD10 and Ki 67 for diagnosing a
malignant phylloides tumor however, in observation it is seen that
Ki 67 had higher sensitivity and specificity as compared to CD 10
to diagnosed malignant phylloides tumor (100%, 100% vs. 87.5%,
88.88%).
Relationship observed between diameter of tumor and increasing
malignant potential, as shown in study out of 18 patients of benign
histology only two had size >10 cm, while out of 8 malignant cases
4 had size >10 cm and only one patient had size < 5 cm. However
analytical study failed to prove this relation (p >0.05). No relationship
was observed in our study between palpable lymph nodes and
expression of Immunohistochemical marker CD 10 and Ki 67 (p
>0.05). No metastasis was found in any of our patients. In 3 recurrent
cases included in our study the CD 10 expression remained same in
both primary and recurrent diseases, while Ki 67 expression increased
in recurrent tumors as compared to their primary counterpart;
however a sample size of two or three patients is too small to make
a universally acceptable hypothesis and to formulate treatment
guidelines.
Conclusion
There is no significant difference between mean age of benign, borderline and malignant phylloides tumors. We investigated CD 10 and Ki 67 and found a significant increase in expression of Stromal cells CD 10 and Ki 67. Ki 67 labeling index of ≤8 suggest benign diseases, a value of ≥8 is strongly suggestive of malignant Phylloides tumor. Expression of CD 10 and Ki 67 is increases in malignant tumor and a higher expression even in benign tumor need a close follow-up for these patients. Tumor size had significant relation with CD 10 expression, as size increases CD 10 expression also increases, but this relation was not observed for Ki 67. Other clinical parameter like lymphadenopathy or locally advanced disease has no such association. Distant metastasis is rare, but local recurrence can occur uncommonly. Tumor size had no significant association with histopahological type, recurrence or lymphadenopathy. Recurrence had no relation with malignancy but associated to surgical management, so excision with adequate margin (>1 cm) has recommended. Lymph nodes dissection is unnecessary if there is no metastasis to lymph nodes, because it has its own complication.
References
- Tavassoli FA. Phyllodes tumor. In Tavassoli FA. Pathology of die breast. 1st ed. Stumford, Connecticut: Appleton and Lange. 1999: 598-613.
- Azzopardi JG, Ahmed A, Millis RR. Sarcoma of the breast. In Bennington JL, ed. Problems in breast and pathology. Philadelphic Saunders. 1979; 11: 346-365.
- World Health Organization: 1981 International histological classification of tumors: histologic typing of breast tumours, ed. sGeneva.1981; WHO.
- Moffat CJ, Pinder SE, Dixon AR, Elston CW, Btancy RW, Ellis IO. Phyllodes tumours of the breast a clinical pathological review of thirty-two cases. Histopathology. 1995; 27: 206-218.
- AK El-Naggar, B Mackay, N Sneige, JG Batsakis. “Stromalneoplasms of the breast: a comparative flow cytometricstudy,” J Surg Oncol. 1990; 44: 151-156.
- Krishnamurthy S, Ashfaq R, Shin HJ, Sneige N. Distinction of phyllodes tumor from fibroadenoma a reappraisal of an old problem. Cancer. 2000; 90: 342-349.
- Salvadori B, Fablo Cusumano, Romualdo Del, Vincenzo Delledonne, Massimo Grassi, Darlo Rovini, et al. Surgical treatment of phyllodes rumors of the breast. Cancer. 1989; 63: 2532-2536.
- Hart WR, Bauer RC, Oberman HA. Cystosarcoma Phylloides; a clinicopathological study of 26 hypercellular periductal stromal tumors of breast. Am J clin pathol. 1978; 70: 211-216.
- Kim CJ, Kim WH. Patterns of p53 expression in phyllodes tumors of the breast. J Korean Med Sci. 1993; 8: 325-328.
- Norris HJ, Taylor HB. Relationship of histologic features to behavior of cystosarcoma phyllodes. Cancer. 1967; 20: 2090-2099.
- K Iwaya, H Ogawa, M Izumi, M Kuroda, K Mukai. "Stromal expression of CD10 in invasive brest carcinoma: a new predictor of clinical outcome". Virchows Archiv. 2002; 440: 589-593.
- Noronha Y, Raza A, Hutchins B, Chase D, Garberoglio C, Chu P, et al. CD 34, CD117, AND Ki 67 expression in Phylloides tumor of breast: an Immunohistochemical study qf 33 cases. Int J Surg Pathol. 2011; 19: 152- 158.
- Gatalica Z, Finkelstein S, Lucio E, Tawfic O, Palazzo J, Hightower B, et al. p 53 protein expression and gene mutation in phyllodes tumor of breast. Pathol Res Pract. 2001; 197: 183-187.
- Celina G Kleer, Thomas J Giordano, Thomas Braun, Harold A Oberman. Pathologic, Immunohistochemical and Molecular Features of Benign and Malignant Phyllodes Tumors of the Breast. Mod Pathol. 2001; 14: 185-190.
- Kocova L, Skalova A, Fakan F, Rousarova M. Phyllodes tumour of the breast: immunohistochemical study of 37 tumours using MIB1 antibody. Pathol Res Pract. 1998; 194: 97-104.
- Kuenen-Bounmeester V, Henzen-Logmans SC, Timmermans MM, Van Staveren I L, Geel AV, Peeterse HJ, et al. Altered expression of p 53 and its regulated proteins in phyllodes tumor of breast. J Pathol. 1999; 189: 169-175.
- Neizabitowski A, Lackowska B, Rys J, Kruezak A, Kowalska T, Mitus J, et al. Prognostic evaluation of proliferative activity and DNA content in the Phyllodes tumor of breast; immunohistochemical and flow cytometric study of 118 cases. Breast cancer Res Treat. 2001; 65: 77-85.