Research Article
Paravertebral Continuous Infusion Analgesia in Vats Patients. Single Center Prospective Randomized Study
Federico Raveglia1*, Matilde De Simone3, Alessandro Rizzi1, Piero Di Mauro2, Alessandro Baisi1 and Ugo Cioffi3
1Department of Thoracic Surgery, University of Milan, Italy
2Department of Anesthesiology, University of Milan, Italy
3Department of Surgery, University of Milan, Italy
*Corresponding author: Federico Raveglia, Department of Thoracic Surgery, University of Milan, A.O. St. Paul Via di Rudini '8, 20142, Milan, Italy
Published: 21 Sep, 2016
Cite this article as: Raveglia F, De Simone M, Rizzi A, Di
Mauro P, Baisi A, Cioffi U. Paravertebral
Continuous Infusion Analgesia in Vats
Patients. Single Center Prospective
Randomized Study. Clin Surg. 2016; 1:
1135.
Abstract
Objective: We have previously presented the successful results of paravertebral local analgesia in thoracotomy patients. Therefore, we have developed and described an intraoperative technique for paravertebral catheter (PC) placement and continuous local analgesic infusion in video assisted thoracic surgery (VATS). This innovative technique is different from the “lots of resistance”/blind or the ultrasound-guided placement procedures, besides continuous infusion work differently from paravertebral blocks. We present a randomized study comparing this procedure with opioid intravenous patient controlled analgesia (OIVPCA).
Methods: From October to December 2015, 40 patients submitted to VATS lung resection have been randomized into two homogenous groups for the administration of analgesic drugs, through PC + OIVPCA (morphine 1 mg/1ml bolus, lock out 10 minutes) in “group A” or OIVPCA alone in “group B”. In “group A”, Tuhoy needle was percutaneously introduced below parietal pleura in the same intercostal space as the camera port. Insertion point was detected on the skin, 3 cm by the vertebral column. PC was pushed through the needle until paravertebral space was reached. Postoperative analgesia was performed by continuous infusion of local analgesic (naropine 3,75%, 7 ml/h). The following parameters have been recorded on scheduled postoperative days: a) pain control using visual analogue scale b) respiratory function using FEV1 and ambient air saturation c) morphine-equivalent demanded/administered. Records have been analyzed with Mann-Withney or Student's tests for independent variables.
Results: No complications in PC placement were recorded. Moreover, due to thoracoscopic approach posterior parietal pleura remained intact, unlike thoracotomy, avoiding any drug runoff in the pleural cavity. Significant differences have been found in favor of group A concerning both rest and cough pain control (p< 0.01 and 0.035) and respiratory function in terms of FEV1 (p=0.017). No difference was recorded concerning ambient air saturation (p >0.05). Group A had significant lower mean morphine-equivalent demanded (5.47 mg vs. 9 mg) and consumption (4.76 mg vs. 8.38 mg).
Conclusion: Intra-operative PC placement was safe and easy to perform. Local analgesic
paravertebral continuous infusion was effective and allowed lower morphine consumption. We
strongly recommend this procedure in VATS to further improve thoracoscopy advantages in pain
management.
Keywords: HIPEC; Cytoreductive surgery; Radiotherapy; Rectal carcinoma; Outcomes
Introduction
Postoperative pain in thoracic surgery is often present and associated with severe complications,
such as atelectasis which can develop into pneumonia because of secretions retention. Moreover,
pain prevents effective coughing, deep breathing and patient’s mobility. At present, various
techniques have been proposed and adopted to prevent postoperative pain and epidural anesthesia
(EA) has been considered the gold standard. In a recent study [1] we have compared the efficacy of EA with continues paravertebral block analgesia in thoracotomy patients.
The aim of the study was to find if paravertebral analgesia could avoid some typical EA difficulties.
Among these, EA catheter is usually placed by an anesthesiologist immediately before surgery when
the patient is awake and is contraindicated for patients taking anticoagulant or anti-platelet drugs
that cannot be suspended for the preoperative period or for those who have coagulopathies.
Moreover, this technique may result in dangerous risks during
placement, including dural perforation, spinal cord hematoma, spinal
infection or abscess. During treatment some adverse events may
occur, including hypotension, urinary retention, nausea, vomiting
or itching. Our data showed that paravertebral block analgesia,
administered by a paravertebral catheter (PC) placed thorough
thoracotomy at the end of surgery, was more effective than EA in
terms of pain management and was not affected by any side effects.
Since results were significantly in favor of PC, we decided to
adopt this technique also in VATS, since thoracoscopy covers more
than 50% of thoracic surgery procedures but gold standard analgesia
has not been determined.
Some Authors have already described the effects of paravertebral
[2] or intercostals blocks [3] in thoracoscopy patients. Different
techniques (eg., the loss of resistance technique also known as the
anesthetic/blind approach, the ultrasound-guided or the neuronal
stimulator-guided technique) to pre-operatively place the PC and
administer postoperative single or multiple injections of drugs in the
paravertebral space, have been described. However, these procedures
have been commonly considered demanding, time-consuming and
especially unsafe, because of many difficulties to correctly identify
and reach the paravertebral space.
Based on this evidences, we decided to develop a new technique
for intraoperative PC placement that should have been simple and
moreover feasible in VATS [4].
It’s our belief that this new placement technique combined
with the continuous infusion model could be successful. Moreover,
unlike thoracotomy, the posterior parietal pleura is always kept
intact avoiding any drug runoff in the pleural cavity. This could
make paravertebral analgesia in VATS even more effective than in
thoracotomy patients.
Therefore, we have proposed a prospective randomized study
to compare paravertebral analgesia with opioid intravenous patient
controlled analgesia (OIVPCA), in thoracoscopic patients. OIVPCA
is widely considered the gold standard in the management of
postoperative pain.
The first end point of our study was to show if paravertebral
analgesia is effective. The secondary endpoint was to find out if this
technique guarantees a smaller use of morphine.
Materials and Methods
The study was approved by local ethic committee at San Paolo
Hospital, University of Milan (n° 16490). All recruited patients
provided informed and written consent to the study. We considered
patients who underwent VATS (2 ports and a 4 centimeters minithoracotomy)
for pulmonary anatomic or sub-anatomic resections.
The inclusion and exclusion criteria are reported in Table 1. SAS®
software was used to perform power analysis in order to estimate the
sample size needed (for a power of 80%). The study was prospective,
randomized and double-blind.
Patients enrolled have been randomized into two homogenous
groups (by SAS® software) for the administration of analgesic drugs,
through PC (continuous infusion of naropine 3,75%, 7 ml/h) +
OIVPCA (morphine 1 mg/1ml bolus, lock out 10 minutes) in “group
A” or OIVPCA alone in “group B”.
Each patient in the two groups had simultaneous infusion of
paracetamol 4 times a day. Any other request by patients to be
administered more pain medication were recorded and satisfied.
Our model is based on a new technique of catheter placement
and on the continuous local anesthetic infusion. Catheter (Contiplex®
Tuhoy Set, B-Braun) is placed in the same intercostal space as the
camera port, since some reports [5] and our experience showed that
post-operative pain after VATS, without rib spreading, primarily
originates from the area used for the camera and chest tube. Contrary
to paravertebral block or intercostal nerve block [3-4], continuous
infusion allows to obtain a high and steady concentration of analgesic
at the spinal branches corresponding to the camera intercostal space.
Moreover, drug diffusion through paravertebral space guarantees
also a good anesthetization of the surrounding chest wall.
Catheter is placed 3 cm by the vertebral column. Insertion
point is detected on the skin thanks to the camera light positioned
by the assistant in contact with the parietal pleura of the selected
paravertebral space. A Tuhoy needle is percutaneously introduced by
the surgeon below the parietal pleura, carefully avoiding its damage.
We underline that these maneuvers can be easily performed just
because of the thoracoscope guide. Then a 7 cc bolus of analgesic is
injected through the needle in order to tunnelize the parietal pleural.
The catheter is insert thorough the needle and pushed until the
paravertebral space is reached. If pleural hydro-dissection has been
correctly performed, the catheter easily runs below the parietal pleura
due to its closed tip. A blue marking of the catheter tip, allows to
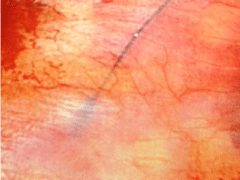
immediately verifying its correct placement (Figure 1). This method
is quicker and safer than the blind or ultrasound-guided approach.
We named the technique “hybrid” since comprises a percutaneous
time coming from the anesthetic approach but at the same time it
is surgically performed by VATS. Local analgesic administration
through the catheter was continues, contrary to standard paravertebral
blocks.
We always placed 1 chest tube at the end of any VATS procedure.
All surgeries were performed by the same equip. In each group
the catheter was removed on the same day that the chest tube was
removed, between the third and fifth day after surgery.
Pain level was measured using a VAS both at rest and while
coughing (where 0 indicates no pain and 10 indicates severe pain)
at 6, 12, 24, 48 and 72 hours after surgery. Moreover, in each group
morphine-equivalent demanded and administered were daily
recorded.
To evaluate pulmonary function at 6, 12, 24, 48 and 72 hours
forced expiratory volume in 1 second (FEV1) and ambient air
saturation were recorded. All data was recorded by the research fellow
(i.e., A.R.). Normally distributed results were compared by Student t
test analysis and non-normally distributed results were compared by
Mann-Whitney analysis.
Table 1
Figure 1
Figure 1
The catheter easily runs below the parietal pleura and its blue marked tip allows to immediately verify the correct placement.
Results
From October to December 2015, 40 patients were enrolled and randomized. The two groups were composed of 20 patients each, 50% and 54% man in the A and B group respectively. The mean patient age was 79 in both groups. No statistical significance was found in the demographic traits (i.e., weight, height and body mass index) of the 2 groups (p >0.47). No patients in either group had a previous history of thoracic surgery or had known allergies. All patients had surgery for lung cancer, except for 2 who underwent lung resection for benign tumor. No complications in PC placement were recorded. No mortality was reported. FEV1 measurements were taken preoperatively and at 6, 12, 24, 48 and 72 hours after surgery with statistical significance (p=0.017) in favor of group A. VAS was recorded at 6, 12, 24, 48 and 72 hours after operation both at rest and while coughing with statistical significance in favor of group A (p< 0.01 and 0.035). Ambient air saturation was taken at 6, 12, 24, 48 and 72 hours after the operation with no statistical significance between two groups (p >0.05). Group A had lower daily mean morphineequivalent demanded (5.47 mg vs. 9 mg) and consumption (4.76 mg vs. 8.38 mg). None of the patients in either groups required additional pain medication.
Discussion
Pain is the most important factor responsible for disorders in
patient’s respiratory mechanism and pulmonary function in the
postoperative period after thoracic surgery [6-11]. It has different
origins, making its etiology very complex. Surgical techniques
influence the nature and intensity of postoperative pain. Standard
thoracotomy is more painful than VATS. In particular, during
thoracotomy, ribs spreading and retraction of the posterior spinal
muscles determine stimuli transmitted by the posterior primary
branches of the spine. The incision pain is mediated by the anterior
branches. On the contrary, during VATS, any ribs spreading is
avoided but the trocar cannulas, when inserted in the intercostal space
(port), make a pressure on surrounding tissue resulting in an injury of
the intercostal nerve. This may determine postoperative neuritis and
increase acute and chronic pain incidence. In particular, camera port
has been described as the port more associated with postoperative
neuritis because of continues camera movements during surgery and
because it is usually used also for chest tube placement.
All these pathways establish stimuli for the origin of pain and
must be blocked.
Following thoracic surgery, a restrictive pathway develops, which
decreases to approximately 40% the baseline values of FEV1 and
functional residual capacity. The relationship between functional
residual lung capacity and closing capacity predicts if atelectasis will
develop; a decreasing functional residual lung capacity to a value less
than closing capacity leads to airway narrowing or closure, which
produces an area with a low ventilation perfusion relationship.
Simultaneously, postoperative pain highly limits voluntary deep
breathing, coughing and patient mobility, and all necessary to
break down atelectasis. All these conditions can be associated with
respiratory infections [12]. Many modalities for the management
of postoperative pain are known. Intravenous opioids may work
adequately but are related to cough suppression, as well as central
nervous system and respiratory depression, leading once again to
retention of secretion and atelectasis. However, intravenous narcotic
analgesia is still considered the most effective treatment in pain
management after surgery and in VATS also.
Our results show that there was always a significant difference
in favor of group A (paravertebral block analgesia) as far as pain
management is concerned, both at rest and while coughing, in the
72 hours after surgery. Data also proves that paravertebral analgesia
group was more successful concerning respiratory outcomes for
pulmonary function in terms of FEV1. However, the most relevant
result is that morphine consumption was lower in group A, avoiding
all the fearsome opioids collateral effects. This allows us to conclude
that the use of paravertebral block analgesia in VATS is effective
in reducing morphine consumption and guaranteeing even more
success in pain management. Concerning PC placement during
thoracoscopy, it is an intraoperative maneuver performed by surgeons
in about 5 minutes, safe and without contraindications.
The strength of our study lies in the prospective randomization
nature and the collection of objective data, such as FEV1, saturation
and morphine consumption. The most important limitations are
represented by the small number of patient’s enrolled, even though
sufficient to reach statistical significance, and the influence of
patient’s personal evaluation of pain. Moreover, the paper shows
that paravertebral analgesia could reduce consumption of opioid, but
does not consider other similar techniques, for example intercostals
continuous analgesia.
References
- Raveglia F, Rizzi A, Leporati A, Di Mauro P, Cioffi U, Baisi A. Analgesia in patients undergoing thoracotomy: epidural versus paravertebral technique. A randomized, double-blind, prospective study. J Thorac Cardiovasc Surg. 2014; 147: 469-473.
- Fibla JJ, Molins L, Mier JM, Sierra A, Carranza D, Vidal G. The efficacy of paravertebral block using a catheter technique for postoperative analgesia in thoracoscopic surgery: a randomized trial. Eur J Cardio-thoracic Surg. 2011; 40: 907-911.
- Forcella D, Pompeo M, Coniglione F, Gatti A, Mineo TC. A new technique for continuous intercostal-intrapleural analgesia in video thoracoscopic surgery. J Thorac Cardiovasc Surg. 2009; 137: e48-e49.
- Cioffi U, Raveglia F, Rizzi A, Di Mauro P, De Simone M, Baisi A. Paravertebral Analgesia in Video-Assisted Thoracic Surgery: A New Hybrid Technique of Catheter Placement for Continuous Anesthetic Infusion. Thorac Cardiovasc Surg. 2015; 63: 533-534.
- Wildgaarda K, Petersenb RH, Hansenb HJ, Møller-Sørensenc H, Ringsteda K, Kehleta H. Multimodal analgesic treatment in video-assisted thoracic surgery lobectomy using an intraoperative intercostal catheter. Eur J Cardio-Thoracic Surg. 2012; 41: 1072–1077.
- Ochroch EA, Gottschalk A, Augostides J, Carson KA, Kent L, Malayaman N, et al. Long-term pain and activity during recovery from major thoracotomy using thoracic epidural analgesia. Anesthesiology. 2002; 97: 1234-1244.
- Senturk M, Ozcan PE, Talu GK, Klyan E, Camci E, Ozyalcin S, et al. The effects of three different analgesia techniques on long-term post thoracotomy pain. Anesth Analg. 2002; 94: 11-15.
- Muehling BM, Halter GL, Schelzig H, Meierhenrich R, Steffen P, Sunder- Plassmann L, et al. Reduction of post-operative pulmonary complications after lung surgery using a fast track clinical pathway. Eur J Cardiothoracic Surg. 2008; 34: 174-180.
- Dowling R, Thielmeier K, Ghaly A, Barber D, Boice T, Dine A. Improved pain control after cardiac surgery: results of a randomized, double-blind, clinical trial. J Thorac Cardiovasc Surg. 2003; 126: 1271-1278.
- Wildgaard K, Ravn J, Kehlet H. Chronic post-thoracotomy pain: a critical review of pathogenic mechanism and strategies for prevention. Eur J Cardiothorac Surg. 2009; 36: 170-180.
- Kehlet H, Dahl JB. Anesthesia, surgery, and challenges in postoperative recovery. Lancet. 2003; 362: 1921-1928.
- Nosotti M, Baisi A, Mendogni P, Palleschi A, Tosi D, Rosso L. Muscle sparing versus posterolateral thoracotomy for pulmonary lobectomy: randomized controlled trial. Interact Cardiovasc Thorac Surg. 2010; 11: 415-419.