Case Report
Treatment Strategy of Metastatic Gastrointestinal Stromal Tumor in Pregnancy
Ikoma N1*, Harvin JA2, Buryanek J3 and Lesslie DP2
1Department of Surgical Oncology, University of Texas MD Anderson Cancer Center, USA
2Department of Surgery, University of Texas Health Science Center at Houston, USA
3Department of Pathology, University of Texas Health Science Center at Houston, USA
*Corresponding author: Naruhiko Ikoma, Department of Surgical Oncology, The University of Texas MD Anderson Cancer Center, 1400 Pressler FCT 17.6055, Houston, Texas, 77030, USA
Published: 19 Sep, 2016
Cite this article as: Ikoma N, Harvin JA, Buryanek J,
Lesslie DP. Treatment Strategy of
Metastatic Gastrointestinal Stromal
Tumor in Pregnancy. Clin Surg. 2016;
1: 1125.
Abstract
Treatment for metastatic gastrointestinal stromal tumor (GIST) is a challenge, particularly in
pregnant female because of the absence of safety data concerning imatinib use during pregnancy. We
report a case of metastatic GIST surgically resected during pregnancy with a favorable outcome. A
treatment strategy for GIST in pregnant patients should be carefully discussed by a multidisciplinary
team, based on the phase of pregnancy, tumor resectability, and patient’s opinion in abortion.
Patient counseling and education regarding the paucity of safety data cannot be overemphasized.
Keywords: Gastrointestinal stromal tumor; Pregnancy; Metastasis
Introduction
Gastrointestinal stromal tumor (GIST) is the most common mesenchymal tumor of the gastrointestinal tract. The diagnostic criteria for GIST were first defined in 1983 with the discovery of tyrosine kinase receptor (CD117) mutations. Since then, the number of reported cases has been increasing [1,2]. GIST treatment was revolutionized by the development of molecular-targeted therapy using tyrosine kinase inhibitors, such as imatinib, which significantly improved the 5-year survival rate [3]. Although the incidence of GIST is increasing, it remains rare in women of childbearing age. There are very few cases of GIST diagnosed or managed during pregnancy; a review of the literature yielded only five such case reports [3,7]. The optimal treatment strategy for metastatic GIST diagnosed during pregnancy is unknown and the absence of safety data concerning imatinib use during pregnancy further confounds the treatment decision [8] . Here, we report a case of metastatic GIST surgically resected during pregnancy with a favorable outcome.
Case Presentation
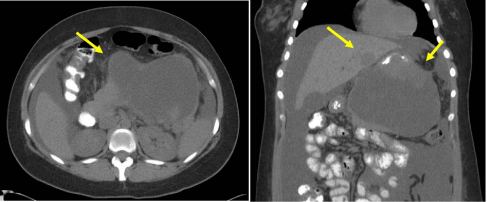
A healthy 20-year-old primigravid at 16 weeks presented to the hospital with severe abdominal pain. Ultrasonography revealed a 15.8 cm mass in the mid-epigastric area. Laboratory data were unremarkable with normal amylase and lipase levels. An MRI was not performed because the patient suffered from claustrophobia. Anon-contrast computed tomography (CT) scan revealed a 15 × 13 × 11cm complex heterogeneous mass which appeared to originate from the body of the pancreas and to protrude into the gastric wall (Figure 1). CT also revealed a 3 cm hypodense lesion on the left side of the liver concerning for metastasis, a moderate volume of ascites, and splenomegaly. Further evaluation with endoscopic retrograde cholangiopancreatography and endoscopic ultrasonography revealed the following: extrinsic compression of the gastric wall from the tumor; a large solid and cystic lesion with septations and well-defined margins; and no clear involvement of the gastric wall. Fine needle aspiration (FNA) was performed on the solid component of the tumor. Seven-hundred cc of thin, reddish-brown, fluid was aspirated from the cystic component. Cytological analyses of the aspirate showed no tumor cells or mucinous material. The tumor markers carcino-embryonic antigen and CA19-9 was not elevated. Pathology indicated a spindle cell tumor consistent with GIST.
Treatment and Follow-up
The following treatment options and risks were discussed with the patient: neoadjuvant imatinib
therapy followed by resection in the late third trimester or immediate resection with adjuvant
imatinib following delivery. The patient opted for surgery. She underwent surgical resection at 18
weeks gestation for a large tumor firmly adherent to both the distal portion of the pancreas and
the posterior portion of the stomach. Upon inspection, the tumor
appeared to originate from the stomach near the esophagogastric
junction. She underwent en-bloc resection of the tumor by total
gastrectomy, distal pancreatectomy, splenectomy, and a Rouxen-
Y esophagojejunostomy. Intraoperative FNA of the liver lesion
confirmed metastatic GIST; therefore, a lateral segment partial
hepatectomy was performed leaving a clear margin. Histological
analyses revealed: a 16 cm, low-grade GIST with a mitotic count < 5/50 high-power field (HPF); mixed spindle and epithelioid cell
types; lymphovascular invasion was detected with 5 out of 9 lymph
nodes being positive for metastasis. Immunohistochemistry revealed
positivity for CD34 and CD117, but no alpha smooth muscle act in
(SMA) (Figure 2). Mitotic figures were counted as less than 5 per
50 high power fields. She was diagnosed with pT4N1M1 stage IV
GIST, and due to its large size (16.0 cm), it was placed in the highrisk
category. Her postoperative course was favorable except for
peri-pancreatic fluid collection that required percutaneous drain
placement. The course of the pregnancy was favorable, and she
delivered a healthy infant at 37 weeks gestation. Follow-up CT scans
were negative for disease recurrence, and she was started on imatinib
400 mg/day for6 weeks after the delivery. At three years after the
surgery she is doing well with no sign of recurrence.
Figure 1
Figure 1
CT scan showed a 15 × 13 × 11 cm complex heterogeneous mass, apparently originating from the body of the pancreas and protruding into the gastric wall, and metastatic lesion in the liver..
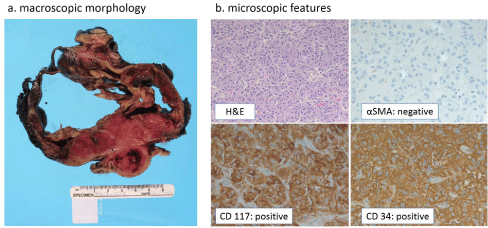
Figure 2:a
Figure 2:A
The gross morphology of the resection of the main tumor showed a mass involving primarily the distal portion of the stomach. The mass had been previously cystic, but had been previously ruptured. b. The H&E section showed the features of the epithelioid variant of GIST. Immunostains showed dual positivity for CD117 and CD34. Alpha SMA was negative in the tumor cells. Mitotic figures were counted as less than 5 per 50 high power fields. However, due to its large size (16.0 cm), it was placed in the high-risk category.
Discussionn
A spigelian hernia is most likely an acquired abdominal wall
Since randomized trials are difficult to perform in pregnant
women because of ethical considerations, consolidation of isolated
case reports of rarely occurring diseases are important. A literature
reviews uncovered 5 previous case reports of GIST in pregnancy
(Table 1). Three patients underwent a simple resection without
adjuvant therapy. One patient waited until 36 weeks and then
underwent simultaneous resection of the tumor and a cesarean
section. One patient was found to be pregnant during treatment with
imatinib for metastatic GIST, which was continued throughout the
pregnancy. None of these case reports indicated a complication of
the pregnancy.
Current consensus recommends adjuvant imatinib use for highrisk
tumors after complete resection of tumors, which has been
shown to improve disease-free and overall survival [9,10]. However,
the safety information concerning imatinib therapy in pregnancy is
limited [4,11] and most of the data are derived from patients with
chronic myeloid leukemia. Pye et al. [5] performed an analysis of 125
pregnancies with imatinib exposure, 50% resulted in a healthy live
infant, 28% in elective termination, and 9.6% of fetuses were born
with malformations. Although most deliveries followed by imatinib
exposure had a successful outcome, there was an increased risk of
fetus malformations; therefore, the current recommendation is that
patients taking imatinib should use effective contraception or should
discontinue the treatment if they conceive inadvertently.
On the basis of a literature review, a reasonable treatment
strategy for GIST in pregnant patients should include the following:
(1) Resection of the primary tumor and metastatic lesions, if feasible,
especially if the tumor was diagnosed in the first or second trimester;
(2) If the tumor is found in the later phase of pregnancy, one should
consider waiting until 36 weeks gestation and then resect the
tumor after or simultaneously with delivery; (3) Adjuvant imatinib
administration should be considered for high-risk tumors, and should
be started after child-birth; (4) For non-resectable metastatic GIST,
continuation of the pregnancy and/or imatinib treatment should
be cautiously discussed with patients with collaboration from the
multidisciplinary team. Patient counseling and education regarding
the paucity of safety data cannot be overemphasized.
Table 1
References
- Mazur MT, Clark HB. Gastric stromal tumors. Reappraisal of histogenesis. Am J Surg Pathol. 1983; 7: 507-519.
- Goettsch WG, Bos SD, Breekveldt-Postma N, Casparie M, Herings RM, Hogendoorn PC. Incidence of gastrointestinal stromal tumours is underestimated: results of a nation-wide study. Eur J Cancer. 2005; 41: 2868-2872.
- Stubbs BM, Desai A, Singh S, Seddon B, Khan F. Gastrointestinal stromal tumour in pregnancy. BMJ Case Rep. 2011; 2011.
- Hensley ML, Ford JM. Imatinib treatment: specific issues related to safety, fertility, and pregnancy. Semin Hematol. 2003; 40: 21-25.
- Goel N, Malik R, Rathi B, Bhaskaran S, Rajaram S, Mehta S, et al. Pregnancy with metastatic gastrointestinal stromal tumor (GIST) on imatinib chemotherapy: an oncologist's nightmare and obstetrician's dilemma. J Gastrointest Cancer. 2013; 44: 115-117.
- Gözükara I, Dilek TU, Durukan H, Düsmez Apa D, Kabil Kucur S, Dilek S. Extragastrointestinal Stromal Tumor during Pregnacy. Case Rep Obstet Gynecol. 2012; 2012: 846747.
- Scherjon S, Lam WF, Gelderblom H, Jansen FW. Gastrointestinal stromal umor in pregnancy: a case report. Case Rep Med. 2009; 2009: 456402.
- Lanzafame S, Minutolo V, Caltabiano R, Minutolo O, Marino B, Gagliano G, et al. About a case of GIST occurring during pregnancy with immunohistochemical expression of epidermal growth factor receptor and progesterone receptor. Pathol Res Pract. 2006; 202: 119-123.
- Dematteo RP, Ballman KV, Antonescu CR, Maki RG, Pisters PW, Demetri GD, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebocontrolled trial. Lancet. 2009; 373: 1097-1104.
- Joensuu H, Eriksson M, Sundby Hall K, Hartmann JT, Pink D, Schütte J, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA. 2012; 307: 1265-1272.
- Pye SM, Cortes J, Ault P, Hatfield A, Kantarjian H, Pilot R, et al. The effects of imatinib on pregnancy outcome. Blood. 2008; 111: 5505-5508.