Review Article
Damage Control in Abdominal Surgery
Karamarković A1,2*
1Faculty of Medicine, University of Belgrade, Serbia
2Clinic for Emergency Surgery, Serbia
*Corresponding author: Professor Aleksandar Karamarkovic MD, PhD, FACS, Faculty of Medicine, University of Belgrade, Serbia
Published: 15 Sep, 2016
Cite this article as: Karamarković A. Damage Control in
Abdominal Surgery. Clin Surg. 2016; 1:
1118.
Introduction
Damage Control Surgery (DCS) is established as a life-saving procedure in severely injured patients.
In addition to the trauma, hemorrhage and tissue hypoperfusion, a secondary systemic injury, by
inflammatory mediator release, contributes to acidosis, coagulopathy, and hypothermia and leads to
multi system organ failure. It is necessary to identify patients unable to tolerate a traditional approach
due to the present or impending state of shock. Use of an abbreviated laparotomy is focused only on
control of bleeding and contamination to limit surgical insult and allow for aggressive resuscitation
in an Intensive Care Unit (ICU) to regain physiological reserves. Only after correction of acidosis,
hypothermia and shock are definitive repairs attempted. Closure of the abdominal wound has
developed thanks to a better understanding of the importance of Intraabdominal Hypertension
(IAH) and Abdominal Compartment Syndrome (ACS). A good knowledge of DCS has led to a
significant increase in survival of severely injured patients. The authors provide an overview of the
DCS approach, as well as the indications for DCS and DCS sequence, followed by a discussion of
DCS-associated complications.
Keywords: Severely injured patients; Abdominal trauma; Damage control surgery; Laparotomy.
Introduction
Damage control surgery (DCS) has been established as a life-saving procedure to control
hemorrhage, prevent contamination and protect from further injury in severely traumatized
patients [1-7]. The term “damage control” reportedly originated from the United States Navy and it
represents “the capacity of a ship to absorb damage and maintain mission integrity” [1]. In surgery,
“damage control” refers to those maneuvers designed to ensure patient survival. Although first
described formally in the civilian trauma population, DCS has been used in the military to facilitate
prompt surgical control of bleeding and contamination and early evacuation of injured soldiers, with
resultant improvement in survival rates [1-5]. The concept of abbreviated surgery aimed primarily
at arresting bleeding was first introduced by Pringle in 1908 [8]. Halsted and Schroeder individually
reported their success at arresting bleeding following liver trauma by packing the liver. In 1913
Halsted [9] described modifications and refinements to the then well established practice of packing.
While some discussions of using an abbreviated laparotomy can be found during the American
Civil War and World Wars, after that period it was generally dismissed as poor surgical care. Stone
et al. [10] demonstrated improved survival in 1983 with abdominal packing for the exsanguinating
hypothermic and coagulopathic trauma patient. Once hemodynamic stability was restored and the
coagulopathy corrected, definitive surgical repairs were completed later. This strategy resulted in
the survival of 11/17 patients felt to have a lethal coagulopathy. The application of these techniques
to trauma patients continued to evolve over the next several years [5]. Damage control surgery was
popularized again in the late 1980’s as a method of salvaging critically ill patients with physiologic
compromise due to massive hemorrhage [2,3]. In 1993, Rotondo and Schwab [3] coined the term
‘damage control surgery’, demonstrating the survival benefit with it, and showing a improvement
in mortality (11% to 77%) in patients with combined visceral and major vascular injury using the
three phase approach.
The principles and sequence of damage control include an abbreviated laparotomy for control of
massive bleeding and contamination, secondary correction of abnormal physiological parameters in
an intensive care setting, followed by a planned definitive reexploration for correction of anatomical
derangements.
A review by Shapiro et al. [1] of over 1000 damage control patients showed an overall 50%
survival. The improvement in survival for severe trauma patients comes with under-standing the
fundamental differences of physiology and anatomy between elective surgery patients and emergency trauma patients who have exsanguinating injuries [3,7,11]. The
severe trauma patient can lose physiological reserves due to massive
bleeding and contamination. The exsanguinating trauma patient does
not have time for optimization of medical problems. These essential
differences in presentation lead to overall poor physiological reserves,
incapable of sustaining a prolonged surgical insult. Attempting
to provide a single definitive procedure in these patients leads to
ongoing bleeding from coagulopathy, an unresuscitatable state of
shock, or multiple organ failure [12]. Over the following decades,
refinements were made to the basic steps to produce the current
model in use today [1,3,13,14]. At its core is the identification of
patients unable to tolerate a traditional approach due to present or
impending state of shock, use of an abbreviated laparotomy focused
only on control of bleeding and contamination, to limit surgical
insult and allow for aggressive resuscitation in an Intensive Care
Unit (ICU) to regain physiological reserves. Only after correction
of acidosis, hypothermia and shock are definitive repairs attempted.
Closure of the abdominal wound has further been separated in the
sequence, and developed thanks to the better understanding of the
importance of Intraabdominal Hypertension (IAH) and Abdominal
Compartment Syndrome (ACS).
Table 1
Indications for Damage Control Surgery
One of the most challenging aspects of DC strategy remains
identifying which patients should be “damage controlled.” The lethal
nature of exsanguinations and profound shock causing the “lethal
triad” of hypothermia/ acidosis/coagulopathy has been well described,
but it is clinically difficult to recognize in the dynamics of patient
arrival, resuscitation and diagnosis. Perhaps the most critical factor is
that an early decision needs to be made in these first few minutes. The
patient’s physiology should be the primary determinant in the need
for damage control surgery and open abdomen techniques (Table 1).
Hypothermia begins at the time of insult, due to shock, prolonged
exposure and injury severity [14]. Several studies have acknowledged
the significant relationship between hypothermia and death [1,12].
Hypothermic patients are predisposed to arrhythmia, have reduced
cardiac output, increased systemic vascular resistance, and a left
shift in their oxygen hemoglobin saturation curves. Temperatures
below 35oC cause platelet dysfunction as well as a dysfunction of the
intrinsic and extrinsic clotting cascades.
The lethal nature is well known of persistent metabolic acidosis
due to hypoperfusion of tissue in the traumatized patient. Placement
of a pulmonary artery catheter and an arterial line are essential to help
guide therapy. Traditional endpoints of resuscitation must be tracked:
urine output, lactate clearance, and measuring mixed venous oxygen
saturation. Aggressive measures include control of hemorrhage,
stabilizing systolic blood pressure, elevation of temperature,
optimization of oxygen delivery via aggressive resuscitation with
blood products and the use of inotrops when needed.
The clinically observed coagulopathy in severe injury patients
is not always confirmed by lab tests, suggesting that mechanisms
other than concentration of clotting factors or number of platelets
are involved. The coagulopathic state of the exsanguinating trauma
patient is dilutional in nature, but both the coagulation cascades,
as well as the platelets are affected with platelet dysfunction. The
fibrinolytic system is also activated following massive tissue damage,
shock, and hypothermia. As part of its multifactorial nature, the
essences of this phenomenon are hypothermia as well as acidosis.
The injured patients may have prolongation of the PT, elevation of
Ddimer levels, and reduction of both fibrinogen and antithrombin III
levels. Early aggressive resuscitation with blood products is necessary
to correct the coagulopathy and prevent further physiological
deterioration, using packed red blood cells, plasma and platelets.
Complex blunt injury patterns, multiple penetrating injuries,
injuries across multiple compartments, or combined vascular and
visceral injuries, also lend themselves to damage control approach
(Table 1). Additionally, open abdomen techniques with damage
control surgery, lend themselves to improved effluent control, while
providing ease of peritoneal cavity accessibility in those trauma
patients with a septic abdomen, and reducing the potential for ACS
[15,16].
We should always keep in mind that there are significant variations
in physiological reserves across the patient populations. The elderly
with multiple comorbidities tend to have less tolerance for surgical
procedures due to poor preexisting reserves. Young patients may hide
progression to physiological exhaustion until hemodynamic collapse.
Damage Control Sequence
In the beginning, damage control surgery was described by the
three main steps: abbreviated laparotomy, ICU resuscitation, and
planned re-operation with definitive repair. Addition of a prehospital
initial evaluation stage (Ground Zero) and separation of definitive
abdominal wall closure occurred as their importance became more evident. During the initial evaluation period the exact endpoint
of resuscitation is debated. Rapid control of the airway and
placement of large IV access devices and immediate resuscitation
with RBC’s and plasma are the therapeutic procedures. Traditionally,
crystalloids have been given to restore normal vital parameters,
but permissive hypotension, resuscitating patients to goal systolic
pressure of approximately 90 mmHg with concomitant signs of end
organ perfusion, is gaining favor, especially in patients with long
transport times to definitive care [17-20]. Prompt transport to the
hospital is essential. Upon arrival, a team effort occurs where the
best method is to perform horizontal resuscitation (as opposed to
vertical resuscitation performed with limited personnel) ac-cording
to the recommendations made by the Advanced Trauma Life Support
(ATLS) program supported by the American College of Surgeons.
Damage control resuscitation continues until surgical control of the
bleeding can be performed. Furthermore, correction of hypothermia,
acidosis, and coagulopathy should be started. The use of blood
product allows volume expansion with oxygen carrying capacity and
reversal of coagulopathy.
The aggressive transfusion policy of Red Blood Cells (RBC), Fresh
Frozen Plasma (FFP), and platelets, applied in a proportion of 1:1:1
(pRBC: FFP: platelets), can be combined with permissive hypotension
[21,22].
Stage I
Initial laparotomy and temporary
Abdominal closures: The initial laparotomy focuses on control
of hemorrhage and visceral contamination before the depletion of
the patient’s physiological reserves and initiation of the acquired
coagulopathy that develops. A different technique exists for
emergency control bleeding sources, to allow restoration during the
ICU resuscitation stage before definitive repair later [23-26]. Visceral
contamination control can often be attained using simple suturing
or stapling techniques to control defects or rapidly remove injured
segments and often require external drainage with closed suction
systems.
Liver injuries
Major bleeding from the liver and complex hepatic injuries,
grades IV and V (American Association for the Surgery of
Trauma Organ Injury Scale, AAST-OIS) continue to challenge
even experienced trauma surgeons [27-29]. The close anatomical
relationship with the vena cava and the triple system of blood
vessels (hepatic inflow and outflow) places control of bleeding in the
foreground in liver injury. In addition, it should be noted that the liver
represents a place of synthesis of all coagulation factors except factor
VIII. In a situation of severe bleeding, coagulation factors are quickly
disrupted by consumption, reduced synthesis and degradation, with
the threatening coagulopathy. In severe liver injury (AAST III-VI)
under conditions of coagulopathy, hypothermia and acidosis, the
DCS concept should be applied to stop the bleeding. This approach
does not allow extensive and complex surgical procedures during the
initial laparotomy, but they can be performed after resuscitation in
the ICU, during the planned re-operation (“staged repair”).
Initial surgery involves quick direct and indirect bleeding control
procedures and/or perihepatic tamponade. Depending on the site of
the injury, mobilization of the right lobe may be necessary, which
can be achieved by dividing the triangular and coronary ligaments
that attach the liver to the diaphragm. If the retrohepatic vena cava
is injured, mobilizing the right lobe can lead to massive hemorrhage
that may be difficult to control [27-29].
Techniques for liver reparation
Large liver lacerations may be managed using either continuous
suture or interrupted horizontal mattress sutures (extensive
hepatorrhaphy). These sutures are passed through the hepatic
capsule, traverse the parenchyma at a depth of about 2 cm from the
lacerated surface, and exit at the opposite side through the capsule.
However, this approach is burdened by complications such as
ischemic necrosis, “dead space” with an accumulation of blood and
bile, lack of effective bleeding control of the deep blood vessels and
the high incidence of haemobilia. It is optimal to use the techniques
of direct control, such as hepatotomy or resectional debridement with
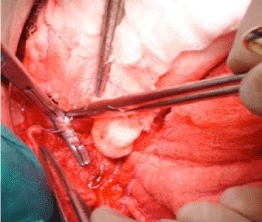
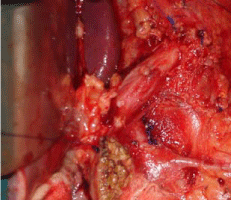
selective vascular ligation (Figure 1).
A tamponade for deep cleft in the parenchyma, after selective
ligation of the blood vessels, can use the omental flap. Finally the
management of complex hepatic injuries may require the use of some
of the most complex surgical techniques in the trauma surgeon’s
armamentarium, including extensive hepatotomy and hepatorrhaphy
with selective deep vessel ligation, formal lobectomy, non-anatomic
resection and debridement [27-29].
Pringle maneuver
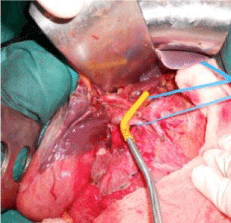
The Pringle Maneuver (PM) is very helpful in controlling bleeding until definitive control is achieved [27,28]. The technique of PM by
clamping the hepatoduodenal ligament with a vascular clamp (Figure
2) or using tape is a very fast and simple procedure for the initial
control of bleeding in severe liver injury. The procedure is safe and
does not impair hemodynamics, but because of the threatening liver
ischemia, PM should be intermittent in character, with a clamping
period of up to 10 minutes. It is also important to distinguish bleeding
from hepatic inflow vessels (hepatic artery and portal vein) and
bleeding from hepatic outflow vessels. PM can re-duce or stop the
bleeding from hepatic inflow.
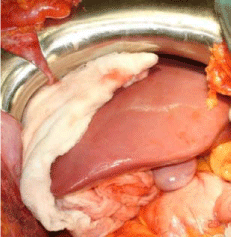
Perihepatic packing
In centrally placed lacerations, after control of the major vessels,
ooze may be managed by cavity packing with viable omentum [27].
In other situations tamponade may be the most expeditious DC
technique. Tamponade can be achieved by perihepatic gauze packing
(“perihepatic packing”), placing a balloon catheter within the tract
where the inflated balloon acts as tamponade, or mobilizing the
injured lobe and circumferentially wrapping it with absorbable mesh
[27-31]. Also in a patient with limited retrohepatic caval injuries,
perihepatic packing may initially control bleeding [27]. Perihepatic
packing is a basic DC technique in establishing control of bleeding in
liver injury. The process is based on the compression of the liver in
superior and posterior directions. Abdominal compresses are placed
around the liver, but not in the lesion itself, in order to compress
the site of the injury and maintain pressure against the diaphragm
(Figure 3). It is very important to avoid cutting the ligaments and
liver mobilization, and also excessive compression between the liver
and diaphragm. Adequately implemented, this maneuver successfully
stops the bleeding, except in major arterial hemorrhage and severe
injuries of juxtahepatic veins (AAST-VI). In such cases, there may be
a repeated attempt at re-tamponade, or using some of the techniques
of vascular control in terms of selective vascular occlusion (SVO) or
total vascular occlusion (TVO) of the liver. Perihepatic tamponade is
used as the main method of hemostasis in 5-6% of cases of severe liver
trauma, where the application of conventional techniques to control
bleeding have been unsuccessful, while in the additional 33% of
patients it is necessary as a procedure, within the concept of DCS, to
stop non-surgical bleeding. This method may be salutary in patients
with acidosis, hypothermia and coagulopathy. In patients with minor
retrohepatic vena cava injuries, tamponade may initially control the
bleeding during the first 24-48h, when reoperation is usually planned.
Other indications for perihepatic packing are listed in (Table 2).
Perihepatic tamponade complications are related to excessive
compression of the liver and vena cava, which may result in ischemia
and necrosis of the liver parenchyma, reducing the flow through
the vena cava, with a decrease in cardiac inflow and deterioration
of circulatory dynamics. Other complications may be portal vein
thrombosis, pulmonary atelectasis and hypoventilation. Septic
complications should not be neglected. However the most important
complications are related to the increase in abdominal pressure and
the high risk of Abdominal Compartment Syndrome (ACS). For this
reason it is very important to regularly control abdominal pressure
by manometry, and timely and adequate correction of abdominal
hypertension. Unfortunately, the mortality from severe liver injury
remains very high, and exsanguinations is the leading cause of death
in these patients [30].
Splenic injuries
In patients with splenic injuries AAST grades III, IV and V,
splenectomy is the procedure of choice for damage control. In lower
grade injuries, simple hemostatic measures may be useful, such as
topical hemostatic agents, suture or mesh wrapping [32].
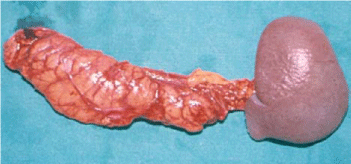
Pancreas injuries
Major pancreas injuries are uncommon, but may result in
considerable morbidity and mortality because of the magnitude of
associated vascular and visceral injuries or underestimation of the
extent of the pancreatic injury (Figure 4). The integrity of the main
pancreatic duct is the crucial point in the management and outcome
of patients with pancreatic trauma [33,34]. Pancreas injuries that
do not involve the duct require external drainage with closed
suction systems. If the pancreas injury is to the left of the mesenteric
vessels, distal pancreactomay is indicated. In massive destruction
of the pancreatic-duodenal complex, pancreaticoduodenectomy is
indicated.
Gastrointestinal tract
If the sites of perforations are the stomach, duodenum,
jejunum/ileum and colon, rapid control of contamination
is an essential part of DCS [35]. Definitive repair with anastomosis
to reestablish intestinal continuity are avoided at this time. Stomach
injuries are sutured closed, with continuous stitch, running stitch
and hemostasis. Smaller perforations of the small and large bowel are closed with a running stitch, but bowels with multiple injuries
or devascularized segments are resected. Staplers allow rapid and
safe resection. Stoma formation and feeding ostomies are sometimes
necessary at this point. The morbidity and mortality following
abdominal trauma and bowel perforation are still high because of
peritonitis and sepsis [35].
Major abdominal vessels injuries
Large abdominal, retroperitoenal and pelvic hematomas with
pelvic fractures, should be carefully explored [36,37]. In patients
who have developed coagulopathy and resultant diffuse nonsurgical
bleeding, packing may be lifesaving. Injury of the major abdominal or
pelvic veins should be managed by ligation. Injury to the abdominal
aorta requires rapid repair using an arterial substitute for wall loss.
The inferior mesenteric artery can be ligated safely in trauma patients.
Injury of the superior inferior mesenteric artery and external iliac
artery should be controlled by repair or by placing an intraluminal
shunt. Bleeding from the internal iliac artery deep in the pelvis is
difficult to control and this may be achieved by ligation, packing and
placing hemostatic agents. Injuries to the renal artery in unstable
patients should be treated with ligation and nephrectomy, after
confirming the presence of a normal contralateral kidney. Although
the adoption of damage control has been associated with reduced
mortality from abdominal vascular injuries due to coagulopathy,
patients have continued to die of exsanguinations and represent a
persistent challenge.
Renal injuries, urinaly collecting system injuries and
internal reproductive organ injuries
Blunt renal trauma managed conservatively is associated with
few complications in the hemodynamically stable patient [38].
Nephrectomy, after confirming the presence of the contralateral
kidney, is the procedure of choice in patients who require damage
control and have massive bleeding from an injured kidney [38]. In
other cases, gauze packing and renal preservation may be possible. In
the damage control situation, urethral repairs or complex procedures
for urethral injuries are not indicated, temporary urinary diversion
techniques are employed. Intraperitoneal bladder injuries should
be managed with a running stitch. In major pelvic trauma, internal
reproductive organs can be injured in association with pelvic
fractures. In very rare situations, the fastest way to control bleeding
would be a hysterectomy.
Temporary abdominal closure
Temporary Abdominal Closure (TAC) has increasingly been
employed, as part of damage control surgery in severely injured
patients, to reduce tension and avoid subsequent ACS during recovery
in the restoration period in the ICU. Abdominal compartment
syndrome is common in these patients who have undergone aggressive
resuscitation. In the intensive care unit, continued attention to
Intraabdominal Hypertension (IAH) and abdominal compartment
syndrome and measures to prevent or treat these conditions is
imperative. Use of this dressing type reduces this risk, and is probably
a major factor in the improvement in mortality seen in this patient
population. TAC has many other useful properties. Besides being
quick, it allows for rapid reentry into the abdominal cavity while
preserving fascial integrity for latter definitive closure. The major goal
of TAC techniques is no longer abdominal coverage alone, but also
fluid control and facilitation of early fascial closure, including helping
in septic source control with wide drainage as important aspects.
Various methods of TAC are available, but negative pressure therapy
seems to be best suited to achieve these goals. Fascial approximation
techniques prevent lateral retraction of the abdominal muscles and can
be combined with TAC techniques. Mesh mediated vacuum assisted
wound closure is emerging as one of the most promising approaches.
Vacuum assisted abdominal dressing (Barker technique abdominal
dressings) is now used for a multitude of reasons [15]. The Barker
style closure can be created from common material, but commercial
kits have been developed which may improve effluent evacuation and
control [39-41]. Given the versatility of this abdominal closure, it has
generally replaced the other temporary closures, such as towel clip
closures, ETHIZIP Temporary Abdominal Wound Closure Device
or Bogota bag. The vacuum-pack technique (Figure 5) is the method
of choice for patients requiring open abdomen management [41].
On completion of abdominal exploration, a perforated polyethylene
sheet is placed over the intraperitoneal viscera and beneath the
peritoneum of the anterior and lateral abdominal wall. Next, a layer
consisting of compressible material, either sterile surgical towels or
a sterile sponge, is placed over the polyethylene sheet. Two silicone
drains are then placed above the towel/sponge and connected to a
vacuum source at 100 to 150 mmHg continuous negative pressure.
The skin surrounding the wound is dried and painted with tincture
of benzoin and kept dry until covered with the final layer, a plastic
polyester drape [41].
Figure 1
Figure 2
Figure 3
Figure 4a
Figure 4a
Gunshot wound with lesion of the pancreas, portal vein and liver:
left splenohemy pancreatectomy.
Figure 4b
Figure 4b
Gunshot wound with lesion of the pancreas, portal vein and liver:
reconstruction of the portal vein.
Table 2
Stage II
ICU resuscitation
After the initial laparotomy, with surgical control of bleeding, the
focus should be on aggressive resuscitation in the ICU. With more
understanding of appropriate resuscitation by support of the patient’s
physiology, the acidosis, hypothermia, and coagulopathy associated with trauma should reverse. Currently, the goal should be as close to
euvolemia as possible, with end organ perfusion, often with liberal
use of blood products [17,19,21].
Secondly, since no single endpoint of resuscitation is capable
of determining the resolution of the shock state, it should continue
until multiple methods of evaluation indicate its resolution. Care
should be taken during the resuscitation to support the patient’s core
temperature, especially with the use of blood products to correct
the patient’s coagulopathy and anemia. A host of products has been
developed in the last two decades to help attain better resuscitation.
These include a multitude of devices to monitor the patient’s
volume status during resuscitation, like the volumetric pulmonary
artery catheter and arterial pulse contour analysis, and to rewarm
the patient, like both external and internal heat exchange devices.
Additionally, a multitude of products has been developed for both
localized hemostasis and the global reversal of coagulopathy to various
levels of success. During this time period, a multitude of ventilator
modes have been developed and commercialized to provide better
lung protective capabilities in the ICU. Mechanical ventilation is an
essential component of the care of patients with Acute Respiratory
Distress Syndrome (ARDS), and a large number of randomized
controlled clinical trials have now been conducted evaluating the
efficacy and safety of various methods of mechanical ventilation for
the treatment of ARDS [42]. Sedation and paralytic use has declined,
to reduce the incidence of ICU polyneuropathy [43-45]. Glycemic
control has become common place in the last decade, but even this
concept has evolved since its inception [46]. Finally, monitoring for
ACS development needs to be performed. Failure to recognize this
clinical entity is often lethal. A better under-standing of this clinical
entity has grown in the last two decades, leading to improved survival
of both trauma and septic patients receiving aggressive resuscitation
[11,15,16].
Stage III
Planned reoperation, definitive repair, abdominal wall
closure
After resuscitation in the ICU has allowed the patient to regain
physiological reserves, generally in 24 to 48 hours, definitive repair
can be undertaken. Since its initial presentation, a more regimented
approach has been developed for guiding the subsequent laparotomy.
In the severe abdominal trauma with septic abdomen, the planned
reoperation focuses on definitive control of the septic source [47-53].
This phase involves the following procedures: careful removal of packs,
inspection and identification of all injuries, control of remaining
bleeding points, definitive gastrointestinal repair, nasoenteric feeding
tube placement, closed suction drainage if needed, temporary or
definitive abdominal wound closure, and tracheostomy if needed.
One advantage of damage control surgery over the traditional
approach is the possibility of regaining intestinal continuity in bowel
injuries and avoiding stomas if possible [47].
Abdominal wall closure
Closure of the abdominal wall, which was initially considered
part of the subsequent laparotomy, has evolved over the past two
decades into part of damage control surgery for trauma patients with
septic abdomen. In these patients there is always a question and the
dilemma of whether to use temporary or definitive abdominal wound
closure. This part of DCS needs to be developed since only 40-70%
of patients can be closed immediately after definitive repair (skin
closure only, silo placement/Bogota bag, vacuum assisted abdominal
dressing). The optimal TAC should control the abdominal viscera
while preventing additional contamination or visceral injury and
control the effluent to preserve skin and soft tissue integrity. They
should be simple to deploy without causing a radiographic artefact.
Unnecessary tension should be avoided to prevent subsequent
abdominal compartment syndrome. Additionally, fusion between
the visceral block and abdominal wall should be prevented. They
should not be costly and actively promote closure of the abdominal
wall. Lastly, fascial integrity should be preserved for later use. In
trauma patients, the majority of patients can achieve definitive
closure; however, fascial closure rates may be lower in cases of septic
abdomen [54]. Open abdominal wounds can be temporized utilizing
skin-only closure, sterile silastic membrane coverage, absorbable or
non-absorbable mesh materials, Negative Pressure Wound Therapy
(NPWT), and the Velcro like Wittmann patch [6]. Immediate use
TAC’s have evolved over the last few decades from simple skin
closures with suture or towel clips. Vacuum assisted abdominal wall
dressings have become the predominate TAC, as they have the most
characteristics of the optimal TAC to date. Additionally, prevention
of fusion of the visceral block to the abdominal wall can be achieved
using vacuum assisted closures, extending the time of primary fascial
closure from 10 to 14 days to up to one month [55].
For patients that will have longer-term closure needs,
interpositional mesh techniques have been developed. The meshes
are attached to the fascial edges and can be tightened over time to
help provide medial traction. Vacuum assisted abdominal dressings
can be used in conjunction with interpositional meshes, though with
more difficulty.
Definitive fascial closure should be pursued whenever possible
[50]. Various wound care adjuncts may help facilitate fascial
approximation/abdominal closure. While some authors suggest that
the Wittmann Patch and NPWT may be associated with improved
rates of fascial closure, others utilize the “Planned Ventral Hernia”
(PVH) as the default pathway in cases where prompt primary
fascial closure is not possible. Additionally, the absorbable meshes
can be left in place to fuse with the visceral block, to provide a bed
for a split thickness skin graft and creation of a “Planned Ventral
Hernia“(PVH), if definitive closure cannot be achieved. Such PVHs
are covered by split thickness skin grafts, with delayed fascial closure
performed after the patient recovers from the acute illness [6]. This
planned ventral hernia can be reversed in six to twelve months, once the visceral block has separated from the surface tissues. Occasionally,
large hernia defects require extensive abdominal wall reconstructions,
utilizing abdominal component-separation techniques. Abdominal
wall reconstruction is especially challenging in the presence of a
fistula or stoma. For this reason, ostomy creation should be avoided
in DCS patients, and enteral anastomosis should be attempted during
definitive repair of DCS (stage III) [1].
Outcomes from DCS
Severe trauma is accompanied by significant morbidity and
mortality. Damage control surgery attempts to identify those
trauma patients incapable of undergoing definitive surgery due
to loss of physiological reserves, and exchange an improvement in
survival for increased morbidity. This approach has shown a survival
rate of approximately 60%, compared to the 11% survival rate of
conventionally treated patients in Rotondo and Schwab’s initial study
[2,3]. In patients with lower energy mechanisms of trauma, such
as stabbings, rates as high as 90% have been reported. Duchesne et
al. [56] reported improved outcomes with the addition of damage
control resuscitation to damage control surgery (74% vs. 55%).
However, damage control surgery is not without its own morbidity
and DCS-associated mortality. Intraabdominal Hypertension (IAH)
and the ACS manifest clinically with tense, distended abdomen,
progressive hypotension, oliguria, and increased airway pressures
[16]. Early recognition of IAH and ACS is essential, by sustained or
repeated elevation with intraabdominal pressure of >12 mmHg.
Abdominal compartment syndrome can be common place in
traumatic injury patients, given the aggressive resuscitation receive.
However, with the rise in its incidence, alternative treatment
modalities have been developed to combat it. Additionally, actively
seeking prevention by using open abdomen techniques such as
vacuum assisted dressings is probably the main reason damage
control surgery improves outcomes [15,16].
Surgical site infections and intraabdominal abscesses associated
with DCS occur in as many as 83% of cases [1]. Major factors to
consider include bile leak (incidence of 8-33%) and enterocutaneous
fistula (incidence of 2-25%) [1]. Enterocutaneous fistulae are more
common in patients treated by DCS, due to increased manipulation
of the viscera. These fistulae tend to have lower spontaneous closure
rates. Up to 15% of trauma patients may experience this complication
[53,54,57].
Intraabdominal Abscess (IAA) rates vary considerably in the
trauma literature (from 10 to 70%), and appear to largely correlate
with the use of intraabdominal packing, especially when the duration
of packing exceeds 72 hours. While more frequent washouts of
the peritoneal cavity may decrease IAA rates, increased bowel
manipulation leads to increased enterocutaneous fistula rates. In
the septic abdomen patient, the development of tertiary peritonitis
(a persistent or recurrent intraabdominal infection despite adequate
initial surgical source control) appears to be approximately 20%;
however, even this rate has considerable variability in the literature
due to numerous definitions in use [58-60]. The advancement of
interventional radiology allows for relatively easier control of this
complication using percutaneous drains, compared to surgical
drainage procedures. Surgical site infections and abdominal abscesses
may also contribute to postoperative fascial dehiscence, reported in
up to 25% of DCS patients [1].
Acute and subacute bowel obstruction in the setting of DCS with
reported incidence, 2-21% is most likely related to surgical adhesions
[41]. Regardless of the timing of post DCS bowel obstruction,
the initial therapy consists of bowel rest, fluid resuscitation with
electrolyte replacement, and nasogastric suctioning. However,
when signs of clinical deterioration develop, operative intervention
has to be undertaken regardless of the anticipated difficulty of
adhesiolysis or the presence of “frozen abdomen”. Chronic ventral
hernia is very common in patients undergoing DCS, with a wide
incidence range (13%-80%) depending on patient-specific factors and
institutional patterns of practice [6,55]. Large ventral hernias may
be associated with prolonged recovery, due to physical discomfort
or loss of function. Definitive abdominal wall closure is associated
with recurrent herniation in 5-10% of cases, depending on the
reconstructive method and patient factors [55]. DCS associated
mortality rates are 17-31% [12,47]. Excluding the primary etiology
that led to the DCS, common factors that cumulatively contribute to
DCS associated morality include Multi System Organ Failure (MSOF),
Systemic Inflammatory Response Syndrome (SIRS), severe infection/
sepsis from a variety of sources, enterocutaneous/enteroatmospheric
fistulae, preexisting malnutrition, chronic comorbid conditions and
advanced age [12,42,47].
Conclusion
Severe trauma with massive hemorrhage may lead to acidosis, coagulopathy, and hypothermia. The lethal nature of exsanguinations and profound shock, causing the “lethal triad” synergistically contributes to further physiological derangement and, if uncorrected, patient death. The concept of damage control surgery has evolved into a lifesaving strategy to improve outcome in selected patients with exsanguinating trauma and life threatening conditions incapable of tolerating traditional methods. Establishment of clearly defined indications is necessary for appropriate use of this approach by performing three basic stages of DCS. During the initial laparotomy, haemorrhage and abdominal contamination are controlled, and temporary abdominal closure is performed (Stage I). The patient then enters Stage II– physiological restoration in ICU. This is followed by planned re-operation and definitive management of injuries and abdominal closure (Stage III). Improved understandings of IAH and ACS have led to the development of DCS as a surgical decompressive strategy. Although DCS may be associated with specific morbidity, it has proven itself clinically as the most successful approach to severely injured patients, with a significant increase in survival.
References
- Shapiro MB, Jenkins DH, Schwab CW, Rotondo MF. Damage control: collective review. J Trauma. 2000; 49: 969-978.
- Rotondo MF, Schwab CW, McGonigal MD, Phillips GR 3rd, Fruchterman TM, Kauder DR, et al. ‘Damage control’: an approach for improved survival in exsanguinating penetrating abdominal injury. J Trauma. 1993; 35: 375-382.
- Rotondo MF, Zonies DH. The damage control sequence and underlying logic. Surg Clin North Am. 1997; 77: 761-777.
- Arthurs Z, Kjorstad R, Mullenix P, Rush RM Jr, Sebesta J, Beekley A. The use of damage-control principles for penetrating pelvic battlefield trauma. Am J Surg. 2006; 191: 604-609.
- Burch JM, Ortiz VB, Richardson RJ, Martin RR, Mattox KL, Jordan GL Jr. Abbreviated laparotomy and planned reoperation for critically injured patients. Ann Surg. 1992; 215: 476-483.
- Stawicki SP, Cipolla J, Bria C. Comparison of open abdomens in non-trauma and trauma patients: A retrospective study. OPUS 12 Scientist. 2007; 1: 1-8.
- Stawicki SP, Brooks A, Bilski T, Scaff D, Gupta R, Schwab CW, et al. The concept of damage control: extending the paradigm to emergency general surgery. Injury. 2008; 39: 93-101.
- Pringle J. Notes on the arrest of hepatic hemorrhage due to trauma. Ann Surg. 1908; 48: 541-549.
- Halsted WS. The employment of fine silk in preference to catgut and the advantages of transfixing tissues and vessels in controlling hemorrhage. Also an account of the introduction of gloves, gutta-percha tissue and silver foil. JAMA. 1913; 60: 1119.
- Stone HH, Strom PR, Mullins RJ. Management of the major coagulopathy with onset during laparotomy. Ann Surg. 1983; 197: 532-535.
- Karamarković A, Doklestić K, Đukić V, Stefanović B, Radenković D, Gregorić P, et al. Povrede jetre. Acta Chir Iugosl. 2010; 57: 57-67.
- Ciesla DJ, Moore EE, Johnson JL, Burch JM, Cothren CC, Sauaia A. A 12-year prospective study of postinjury multiple organ failure: has anything changed? Arch Surg. 2005; 140: 432-438.
- Hoey BA, Schwab CW. Damage control surgery. Scand J Surg. 2002; 91: 92-103.
- Johnson JW, Gracias VH, Schwab CW, Reilly PM, Kauder DR, Shapiro MB, et al. Evolution in damage control for exsanguinating penetrating abdominal injury. J Trauma. 2001; 51: 261-269.
- Schecter WP, Ivatury RR, Rotondo MF, Hirshberg A. Open abdomen after trauma and abdominal sepsis: a strategy for management. J Am Coll Surg. 2006; 203: 390-396.
- Cheatham ML, Safcsak K. Is the evolving management of intraabdominal hypertension and abdominal compartment syndrome improving survival? Crit Care Med. 2010; 38: 402-407.
- Holcomb JB. Damage control resuscitation. J Trauma. 2007; 62: S36-S37.
- Soreide E, Deakin CD. Pre-hospital fluid therapy in the critically injured patient - a clinical update. Injury. 2005; 36: 1001-1010.
- Rhee P, Koustova E, Alam HB. Searching for the optimal resuscitation method: recommendations for the initial fluid resuscitation of combat casualties. J Trauma. 2003; 54: S52-S62.
- Dawes R, Thomas GO. Battlefield resuscitation. Curr Opin Crit Care. 2009; 15: 527-535.
- Holcomb JB. Damage control resuscitation. J Trauma. 2007; 62: S36-S37.
- Beekley AC. Damage control resuscitation: a sensible approach to the exsanguinating surgical patient. Crit Care Med. 2008; 36: S267-S274.
- Chambers LW, Green DJ, Sample K, Gillingham BL, Rhee P, Brown C, et al. Tactical surgical intervention with temporary shunting of peripheral vascular trauma sustained during Operation Iraqi Freedom: one unit’s experience. J Trauma. 2006; 61: 824-830.
- Reilly PM, Rotondo MF, Carpenter JP, Sherr SA, Schwab CW. Temporary vascular continuity during damage control: intraluminal shunting for proximal superior mesenteric artery injury. J Trauma. 1995; 39: 757-760.
- Rasmussen TE, Clouse WD, Jenkins DH, Peck MA, Eliason JL, Smith DL. The use of temporary vascular shunts as a damage control adjunct in the management of wartime vascular injury. J Trauma. 2006; 61: 8-12.
- Glass GE, Pearse MF, Nanchahal J. Improving lower limb salvage following fractures with vascular injury: a systematic review and new management algorithm. J Plast Reconstr Aesthet Surg. 2009; 62: 571-579.
- Trunkey DD. Hepatic trauma: contemporary management. Surg Clin North Am. 2004; 84: 437-450.
- Doklestić K, Karamarković A, Stefanović B, Stefanović B, Milić N, Gregorić P, et al. The Efficacy of Three Transection Techniques of the Liver Resection: A Randomized Clinical Trial. Hepatogastroenterology. 2012; 59: 1501-1506.
- Asensio JA, Petrone P, García-Núñez L, Kimbrell B, Kuncir E. Multidisciplinary approach for the management of complex hepatic injuries AASTOIS grades IV-V: a prospective study. Scand J Surg. 2007; 96: 214-220.
- Karamarković A, Doklestić K, Milić N, Djukić V, Bumbasirević V, Šijački A, et al. Glissonean Pedicle approach in Major Liver resections. Hepatogastroenterology. 2012; 59: 1896-1901.
- Sriussadaporn S, Pak-art R, Tharavej C, Sirichindakul B, Chiamananthapong S. A multidisciplinary approach in the management of hepatic injuries. Injury. 2002; 33: 309-315.
- Peitzman AB, Heil B, Rivera L, Federle MB, Harbrecht BG, Clancy KD, et al. Blunt splenic injury in adults: Multi-institutional Study of the Eastern Association for the Surgery of Trauma. J Trauma. 2000; 49: 177-187.
- Degiannis E, Glapa M, Loukogeorgakis SP, Smith MD. Management of pancreatic trauma. Injury. 2008; 39: 21-29.
- Ahmed N, Vernick JJ. Pancreatic injury. South Med J. 2009; 102: 1253-1256.
- Sule AZ, Kidmas AT, Awani K, Uba F, Misauno M. Gastrointestinal perforation following blunt abdominal trauma. East Afr Med J. 2007; 84: 429-433.
- Sorrentino TA, Moore EE, Wohlauer MV, Biffl WL, Pieracci FM, Johnson JL, et al. Effect of damage control surgery on major abdominal vascular trauma. J Surg Res. 2012; 177: 320-325.
- Paul JS, Webb TP, Aprahamian C, Weigelt JA. Intraabdominal vascular injury: are we getting any better? J Trauma. 2010; 69: 1393.
- Baverstock R, Simons R, McLoughlin M. Severe blunt renal trauma: a 7-year retrospective review from a provincial trauma centre. Can J Urol. 2001; 8: 1372- 1376.
- Schein M, Saadia R, Jamieson JR, Decker GA. The ‘sandwich technique’ in the management of the open abdomen. Br J Surg. 1986; 73: 69-70.
- Brock WB, Barker DE, Burns RP. Temporary closure of open abdominal wounds: the vacuum pack. Am Surg. 1995; 61: 30-35.
- Barker DE, Green JM, Maxwell RA, Smith PW, Mejia VA, Dart BW, et al. Experience with vacuumpack temporary abdominal wound closure in 258 trauma and general and vascular surgical patients. J Am Coll Surg. 2007; 204: 784-792.
- Girard TD, Bernard GR. Mechanical ventilation in ARDS: a state-of-the-art review. Chest. 2007; 131: 921-929.
- Hall JB, Schweickert W, Kress JP. Role of analgesics, sedatives, neuromuscular blockers, and delirium. Crit Care Med. 2009; 37: S416-S421.
- de Jonghe B, Lacherade JC, Sharshar T, Outin H. Intensive care unitacquired weakness: risk factors and prevention. Crit Care Med. 2009; 37: S309-S315.
- Murray MJ, Cowen J, DeBlock H, Erstad B, Gray AW Jr, Tescher AN, et al. Clinical practice guidelines for sustained neuromuscular blockade in the adult critically ill patient. Crit Care Med. 2002; 30: 142-156.
- Van den Berghe G, Schetz M, Vlasselaers D, Hermans G, Wilmer A, Bouillon R, et al. Clinical review: Intensive insulin therapy in critically ill patients: NICE-SUGAR or Leuven blood glucose target? J Clin Endocrinol Metab. 2009; 94: 3163-3170.
- Miller PR, Chang MC, Hoth JJ, Holmes JH 4th, Meredith JW. Colonic resection in the setting of damage control laparotomy: is delayed anastomosis safe? Am Surg. 2007; 73: 606-609.
- Adkins AL, Robbins J, Villalba M, Bendick P, Shanley CJ. Open abdomen management of intra-abdominal sepsis. Am Surg. 2004; 70: 137-140.
- Schein M. Planned reoperations and open management in critical intra-abdominal infections: prospective experience in 52 cases. World J Surg. 1991; 15: 537-545.
- Cipolla J, Stawicki SP, Hoff WS, McQuay N, Hoey BA, Wainwright G, et al. A proposed algorithm for managing the open abdomen. Am Surg. 2005; 71: 202-207.
- Finlay IG, Edwards TJ, Lambert AW. Damage control laparotomy. Br J Surg. 2004; 91: 83-85.
- Horwood J, Akbar F, Maw A. Initial experience of laparostomy with immediate vacuum therapy in patients with severe peritonitis. Ann R Coll Surg Engl. 2009; 91: 681-687.
- Christou NV, Barie PS, Dellinger EP, Waymack JP, Stone HH. Surgical Infection Society intra-abdominal infection study. Prospective evaluation of management techniques and outcome. Arch Surg. 1993; 128: 193-198.
- Tsuei BJ, Skinner JC, Bernard AC, Kearney PA, Boulanger BR. The open peritoneal cavity: etiology correlates with the likelihood of fascial closure. Am Surg. 2004; 70: 652-656.
- Miller PR, Thompson JT, Faler BJ, Meredith JW, Chang MC. Late fascial closure in lieu of ventral 17 hernia: the next step in open abdomen management. J Trauma. 2002; 53: 843-849.
- Duchesne JC, Kimonis K, Marr AB, Rennie KV, Wahl G, Wells JE, et al. Damage control resuscitation in combination with damage control laparotomy: a survival advantage. J Trauma. 2010; 69: 46-52.
- Anderson O, Putnis A, Bhardwaj R, Ho-Asjoe M, Carapeti E, Williams AB, et al. Short- and long-term outcome of laparostomy following intra-abdominal sepsis. Colorectal Disease. 2011; 13: e20-e32.
- Calandra T, Cohen J. International Sepsis Forum Definition of Infection in the ICUCC. The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Crit Care Med. 2005; 33: 1538-1548.
- Buijk SE, Bruining HA. Future directions in the management of tertiary peritonitis. Intensive Care Med. 2002; 28: 1024-1029.
- Evans HL, Raymond DP, Pelletier SJ, Crabtree TD, Pruett TL, Sawyer RG. Diagnosis of intraabdominal infection in the critically ill patient. Curr Opin Crit Care. 2001; 7: 117-121.