Case Report
One and a Half Syndrome Complicating Resection of Fourth Ventricular Neurenteric Cyst
Toms MC, Gasteiger M, Lal S, Bolinger BD and Toms SA*
Department of Neurosurgery, Geisinger Health System, USA
*Corresponding author: Steven A. Toms, Department of Neurosurgery, Geisinger Health System, MC: 01-28, MH2, 100 North Academy Avenue, Danville, PA 17822, USA
Published: 11 Jul, 2016
Cite this article as: Toms MC, Gasteiger M, Lal S, Bolinger BD, Toms SA. One and a Half
Syndrome Complicating Resection of Fourth Ventricular Neurenteric Cyst. Clin Surg. 2016; 1: 1043.
Abstract
Neuroenteric cysts are rare benign lesions of the central nervous system, typically occurring
anterior to the brainstem. They are thought to arise from disturbances in early gastrulation. A
neuroenteric cyst, appearing wholly within the fourth ventricle without obvious connection to the
ventral brainstem, is described. Removal of the cyst caused a one and one half syndrome, which
slowly resolved after surgery. Reoperation on recurrent cyst showed a midline raphe in the fourth
ventricle, leading the authors to hypothesize that this raphe represented a gliotic band from a split
notochord during development, which may have transmitted vibration during cyst removal, leading
to brainstem injury and the resultant one and one half syndrome.
Keywords: Fourth ventricular neurenteric cyst; Gastrulation
Introduction
Neurenteric cysts are uncommon lesions of the central nervous system. They consist of epithelial
cells and may contain mucosal glands, cilia, and other such features common to the digestive or
respiratory tracts. Their diagnosis can be confusing as they can easily be misdiagnosed as arachnoid
or epidermoid cysts [1-3]. Due to this confusion, they are known by many names throughout
the literature such as endodermal cysts, enterogenous cysts, intestinomas, gastrocytomas,
gastroenterogenous cysts, or archenteric cysts [2,4]. According to Gauden et al. [1], only 140 cases of intracranial cysts have been reported since 1952 [1]. Intracranial cysts are most commonly located in the posterior fossa [3,5,6]. The most common posterior fossa locations are anterior to the brainstem in the cerebellopontine angle and within the fourth ventricle [5-7]. They are rarely positioned supratentorially [1,2].
It has been hypothesized that neuroenteric cysts arise from defects in gastrulation. Since
neuroenteric cysts represent cells of endodermal origin, it has been postulated that herniation of
an endodermal layer during gastrulation through a “split notochord” may allow the adhesion of
cells of endodermal origin into developing neural folds of the ectoderm [8-10]. This hypothesis
would show how cells could be left behind within the fourth ventricle to form neuroenteric cysts
without obvious ventral connections but does fully explain how a “split notochord” may re-join
without the development of a gliotic median raphe along the midline during brain development.
This illustrative case report will suggest that the complications arising from the removal of a dorsally
located neuroenteric cyst might best be explained by a ventral connection to endodermal tissue
along a gliotic midline raphe.
Case Report
A 57-year-old man presented with vertigo when he turned his head to the left for associated
nausea and vomiting. The patient’s physical exam and neurological exam were normal. An MRI,
revealing a cystic lesion dorsal to the brainstem and splitting the cerebellar hemispheres, appeared to
follow CSF on all sequences (Figure 1A-D). A workup for possible neurocysticercosis was negative,
leading to a clinical and radiographic diagnosis of arachnoid cyst.
The patient underwent sub occipital craniotomy with microsurgical resection of the lesion.
After opening the dura, a multiloculated cystic mass was identified located inferior to the cerebellum
and extending into the fourth ventricle resembling a mass of grapes. Cystic masses were dissected
from the fourth ventricle with minimal difficulty with no bleeding nor breach of the pia of the floor
of the fourth ventricle.
After surgery, the patient complained of double vision. An MRI failed to show either diffusion
weighted imaging or FLAIR changes suggestive of stroke or other
injury to the pons (Figure 2A and B). He was diagnosed with having
internuclear ophthalmoplegia and a left sixth nerve palsy. Mild
imbalance rapidly occurred when turning his head. The patient was
prescribed steroids and given an eye patch to help correct the issue.
A multicystic lesion with mucoid material, mucinous epithelium, and
basement membranes was identified on pathological examination
(Figure 3A and B). Epithelial membrane antigen (EMA) was focally
positive, and a diagnosis of neuroectodermal cyst was made.
Although his internuclear ophthalmoplegia (INO) and sixth nerve
palsy improved at seven months postoperatively, he still had a subtle
right INO and a slight left gaze (sixth nerve) paresis, representing the
infrequent diagnosis of one-and-a-half syndrome.
Shortly thereafter, a follow up MRI showed the re-growth of a
cyst in the fourth ventricle, and a second operation was performed
(Figure 4A-D). The lesion, a purplish-hued cyst, was situated at the
floor of the fourth ventricle (Figure 5A and B). It was separated from the
brainstem using micro dissection techniques. A subtle midline raphe
and gliosis were noted on the floor of the fourth ventricle between the
facial colliculi. The patient tolerated the second procedure
well without obvious worsening of his subtle INO and sixth nerve
palsy.
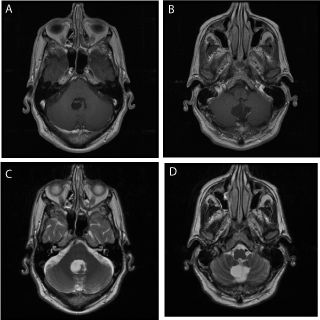
Figure 1
Figure 1
Preoperative axial T1 weighted with gadolinium (A,B) and T2
weighted axial images (C,D) through the lesions. Note the cystic structures
between the tonsils and the cerebellum extending into the fourth ventricle.
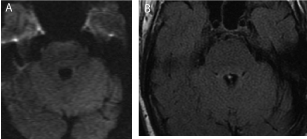
Figure 2
Figure 2
Postoperative diffusion weighted (A) and FLAIR (B) images
through the floor of the fourth ventricle at the level of the PPRF. Note that
there appears to be no swelling or ischemia noted postoperatively at the level
of the expected injury given the clinical findings.
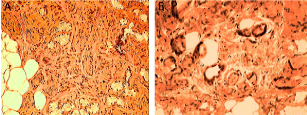
Figure 3
Figure 3
H&E stains (A) and EMA stains (B) of the resectred cyst. Note the
cuboidal epithelium present in (A) and the brown staining of the EMA along
the cuboidal epithelium (B).
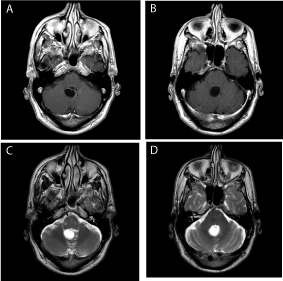
Figure 4
Figure 4
Preoperative axial T1 weighted with gadolinium (A,B) and T2
weighted axial images (C,D) through the recurrent cystic lesion.The cystic
structure now appears to be a single cyst higher in the fourth ventricle than
the prior cysts.
Figure 5
Figure 5
Intraoperative microscopic view of the cyst wall (A) resting on an
underlying cottonoid. After removal of the cyst, the floor of the fourth ventricle
is visible (B).The arrow points to the midline raphe to which the cyst was
attached and is postulated to be the source of the injury to the patient during
the first cyst removal procedure.
Discussion
Neurenteric cysts are benign central nervous system lesions. They
account for just 0. 01% of central nervous system tumors [1]. When
these cysts do occur, they are frequently sited ventral to the spinal
cord with spinal cases occurring about three times more frequently
than intracranial ones [4]. Intracranial neurenteric cysts are generally
traced to the posterior fossa, either anterior to the brainstem in the
cerebellopontine angle or in the fourth ventricle. [5-7]. There have
also been a small number of cases reported supratentorially [1-3].
Although spinal neurenteric cysts are most common in pediatric
patients, intracranial neurenteric cysts happen in all ages with
an approximate median age of 34 [1]. While some studies have
determined a male predominance, [3,5] other studies have found
a female predominance [4]. Therefore, most authors conclude that
neurenteric cysts are independent of sex [1,4,7].
Classification
Neurenteric cysts consist of epithelial cells of a presumed endodermal origin. They may or may not be ciliated, and they lie on a basement membrane, a key feature that differentiates neurenteric cysts from the very similar neuroepithelial cysts [11]. Wilkins and Odom developed a method for classifying neurenteric cysts based on histological features [11]. Type A cysts are lined with pseudo stratified cuboidal or columnar epithelial cells that are similar in appearance to respiratory or gastrointestinal cells. Type B cysts may additionally have complex invaginations and have glands for producing mucinous or serous fluid. These cysts may also contain additional cell types that are associated with gastrointestinal or respiratory tissue such as smooth muscle, lymphoid tissue, and striated muscle. Type C cysts, in addition to the features of type A and B cysts, hold glial elements.
Imaging
There is a great deal of variance in the imaging of these cysts, most likely due to the variable protein makeup of the cysts’ contents [2,12,13]. Cysts most usually appear as hypo dense on CT scans, but they may also appear as hyper dense [2,4,5]. Contrast enhanced CT scan rarely show enhancement, however, some cases do exhibit enhancement [4,5,7]. On MRI, cysts typically appear as is intense to hyper intense to CSF on T1 weighted MRI [1,4,7,12,14]. They usually seem hyper intense to CSF on T2 weighted images [4,7,12]. Since neurenteric cysts have imaging properties very similar to other kinds of intracranial lesions, the diagnosis of a neurenteric cyst by radio imaging alone is quite challenging.
Histopathology
Immuno histochemical analysis is the best way to definitively distinguish a neurenteric cyst from other entities. Due to their endodermal origin, neurenteric cysts are generally negative for both GFAP and S-100 [2,3,6,7]. They are positive for many endodermal markers such as cytokeratin, EMA, and carcinoembryonic antigen [2,3,6,7] and are often positive for acid Schiff staining confirming the presence of goblet cells [1,3,7].
Case Discussion
This case represents a rare occurrence of a neurenteric cyst dorsal
to the brain stem extending into the fourth ventricle. Although the cyst
had an unusual location, what makes this case truly informative is the
visual deficit that the patient experienced after surgery. The patient
complained of double vision developing in the early perioperative
period, and formal examination by a neuro-ophthamologist
diagnosed the patient with a one and a half syndrome. This syndrome
is typically related with damage to the paramedian pontine reticular
formation (PPRF) and the medial longitudinal fasciculus (MLF) on
the same side. Since neither of these structures was within the area
of the cysts, it is puzzling as to why they would have been affected by
the cysts’ removal, as one and a half syndrome is typically caused by
intrinsic brain stem or pontine lesions such as tumor or stroke [15].
Since the postoperative MRI did not reveal bleeding or ischemia
of the brainstem, the authors hypothesize that in the process of cyst
removal, a midline raphe became disturbed. The vibrations of these
fibers, upon the removal of the cysts, resulted in damage more deeply
within the pons in the PPRF and the MLF. This hypothesis is supported
by the observance of abnormal midline raphe during the second
surgery and supports the notion of the “split notochord” hypothesis.
This hypothesis may explain the persistence of endodermal element
dorsally in the fourth ventricle without an anterior cyst component or
persistent of a cleft in the anterior brainstem which connects the cells
to their endodermal origins.
Conclusion
Neurenteric cysts are rarely identified in the fourth ventricle of the brain, as they are a developmental cyst originating from the primitive connections between the nervous system and the endoderm of the gastrointestinal system. Given this origin, it follows that a neurenteric cyst occupying the fourth ventricle would have an intervening connection to the ventral portions of the nervous system. In this case report, a midline raphe was identified. It is speculated that traction to this midline raphe during removal of the cysts led to transmission of a vibrational or traction injury to the pons resulting in a one-and-a-half syndrome. Although both neurenteric cysts and ophthalmoplegic complications of dorsal fourth ventricular surgery are rare, care must be taken in removing dorsal cysts so as not to distort or place traction upon possible connections between the cyst and the brainstem itself.
References
- Gauden AJ, Khurana VG, Tsui AE, Kaye AH. Intracranial neuroenteric cysts: a concise review including an illustrative patient. J Clin Neurosci. 2012; 19: 352-359.
- Teufack S, Campbell P, Moshel YA. Intracranial neuroenteric cysts: two atypical cases and review of the literature. JHN Journal. 2011; 6: 18-21.
- Cheng JS, Cusick JF, Ho KC, Ulmer JL. Lateral supratentorial endodermal cyst: case report and review of literature. Neurosurgery. 2002; 51: 493-499.
- Preece MT, Osborn AG, Chin SS, Smirniotopoulos JG. Intracranial neurenteric cysts: imaging and pathology spectrum. AJNR Am J Neuroradiol. 2006; 27: 1211-1216.
- Bejjani GK, Wright DC, Schessel D, Sekhar LN. Endodermal cysts of the posterior fossa. Report of three cases and review of the literature. J Neurosurg. 1998; 89: 326-335.
- Kulkarni V, Daniel RT, Haran RP. Extradural endodermal cyst of posterior fossa: case report, review of the literature, and embryogenesis. Neurosurgery. 2000; 47: 764-767.
- Wang L, Zhang J, Wu Z, Jia G, Zhang L, Hao S, et al. Diagnosis and management of adult intracranial neurenteric cysts. Neurosurgery. 2011; 68: 44-52.
- Dodds GS. Anterior and posterior rhachischisis. Am J Pathol. 1941; 17: 861-872
- Harris CP, Dias MS, Brockmeyer DL, Townsend JJ, Willis BK, Apfelbaum RI. Neurenteric cysts of the posterior fossa: recognition, management, and embryogenesis. Neurosurgery. 1991; 29: 893-897.
- Macdonald RL, Schwartz ML, Lewis AJ. Neurenteric cyst located dorsal to the cervical spine: case report. Neurosurgery. 1991; 28: 583-587.
- Wilkins RH, Odom GL. Spinal intradural cysts. In: Vinken PJ, Bruyn GW, editors. Tumors of the spine and spinal cord. Handbook of Clinical Neurology. New York: North Holland. 1976; 20(part II): p. 55-102.
- Shakudo M, Inoue Y, Ohata K, Tanaka S. Neurenteric cyst with alteration of signal intensity on follow-up MR images. AJNR Am J Neuroradiol. 2001; 22: 496-498.
- Geremia GK, Russell EJ, Clasen RA. MR imaging characteristics of a neurenteric cyst. AJNR Am J Neuroradiol. 1988; 9: 978-980.
- Brooks BS, Duvall ER, el Gammal T, Garcia JH, Gupta KL, Kapila A. Neuroimaging features of neurenteric cysts: analysis of nine cases and review of the literature. AJNR Am J Neuroradiol. 1993; 14: 735-746.
- Wall M, Wray SH. The one-and-a-half syndrome--a unilateral disorder of the pontine tegmentum: a study of 20 cases and review of the literature. Neurology. 1983; 33: 971-980.