Case Report
Treatment Plan for Specific Gastric Varicies: Isolated Gastric Fundal Varicies with Portasystemic Shunt
Garnic JD*, Walters MJ and Ayres SJ
Department of Interventional Radiology (JDG, MJW), Gastrointestinal Medicine (SJA), Benefis Health System, USA
*Corresponding author: J. Daniel Garnic, Department of Interventional Radiology (JDG, MJW), Department of Gastrointestinal Medicine (SJA), Benefis Health System, USA
Published: 30 May, 2016
Cite this article as: Garnic JD, Walters MJ, Ayres SJ.
Treatment Plan for Specific Gastric
Varicies: Isolated Gastric Fundal
Varicies with Portasystemic Shunt. Clin
Surg. 2016; 1: 1023.
Abstract
We present four recent cases of severe gastrointestinal hemorrhage, resulting in profound decrement in hemoglobin. All cases were due to isolated gastric fundal varicies demonstrating a large gastrorenal venous shunt, not amenable to endoscopic sclerotherapy. Each was treated with a combination of TIPS and variceal occlusion with both outflow and inflow occlusion. Follow up for over a year in each case has defined no recurrence of bleeding or variceal development.
Introduction
Gastric varicies represent an extremely difficult disease process in some cases because treatment options have been poor. There are several specific types of gastric varicies. As an extension of
esophogeal varices, (GOE1, SARIN) [1] endoscopic sclerotherapy represents an effective treatment
path. But isolated cardiac, fundal, varicies (IGV1, SARIN) [1] with a large portsystemic outflow
shunt represent a poorly approachable clinical situation by current treatment methods [2]. Not only
are such venous vessels often large and tortuous but a direct high flow portasystemic pathway limits
treatment options and increases complication potential.
Bleeding from isolated fundal gastric varicies fortunately are a relatively rare process compared to esophogeal varicies (10-36%) [2], however bleeding from such varicies is often massive and
therefore more life threatening. This is believed due to lack of treatment options, frequently large
vessel size and often (up to 85% of cases) [3,4] demonstrating a large porta-systemic outflow.
Endoscopic banding is difficult due to poor support in the gastric fundus and large tortuous
vessels. Standard endoscopic sclerotherapy injection has been ineffective because of large vein
size and high flow, injection site massive bleeding a disasterous potential complication [2]. Open
surgical resection of the varicies has proven unacceptable because of poor general patient status
from such a massive bleed limiting patient anesthesia capability and poor anatomic uniformity [2].
Transjugular intrahepatic portasystemic shunt (TIPS) an excellent salvage procedure for esophageal
varicial hemorrhage has proven ineffective for isolated gastric varicies with a large portasystemic
outflow. Although reasoning behind this observation has not been clarified we believe it likely due
to the already low portal pressure in patients with such a large portasystemic outflow. A promising
treatment has been multiply reported by Hirota [5], Akahoshi [6] and Katoh [7] among many reports. This option, Balloon-occluded retrograde transvenous obliteration (B-RTO) has been
undertaken prophylactically in hundreds of patients in Japan with excellent results. However these
patients have, in general been of better Childs classification and have in general not acutely bled.
A study in such acutely compromised patients however indicated extremely poor outcomes in a
vast percentage. We report four patients, accumulated within one year, with massive bleeding from
isolated gastric varicies, treated with a combination of TIPS (despite normal to low corrected portal
pressure) and varix obstruction utilizing a combination of outflow occlusion with an Amplatzer II
occlusion device and inflow obliteration either with gelfoam soaked in sclerosant sodiumtetradecyl
sulfate or an Amplatzer II device if a single large source to the gastric varix is identifiable.
Case Presentation
case 1
A 45 year old female with no known history of hepatitis, cirrhosis or encephalopathy presented
acutely with an initial episode of massive gastrointestinal hemorrhage, hemoglobin decreased to 3
grams. Endoscopy revealed no esophogeal varicies or gastric ulcer. However, isolated gastric fundal
varicies with a probable site of prior hemorrhage was defined (Figure 1). No endoscopic treatment
was deemed appropriate. Consultation for TIPS was requested. As part of the workup for that
consultation we obtained a CT abdomen with arterial and delayed
venous phase imaging. We defined the gastric varicies but also a
large, 10mm, gastrorenal venous shunt. (Figure 2A and B) This large
portasystemic communication certainly resulted in a low corrected
portal vein pressure. Instead of facing a situation in which TIPS is
known to be advantageous, elevated corrected portal vein pressure
with esophageal variceal hemorrhage, we faced a much less favorable
pathophysiological situation. Initial consultation assessment was that
TIPS would likely not alleviate bleeding potential from high flow
gastric varicies and possibly lead to hepatic encephalopathy.
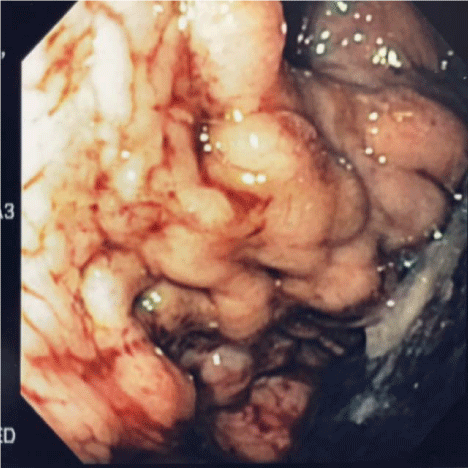
Figure 1
Figure 1
Endoscopic view of isolated gastric varicies, IGV1 (Sarin, 1) which
led to profound hemorrhage in this 45 year old female. Hemoglobin fell to 3
gm at admission.
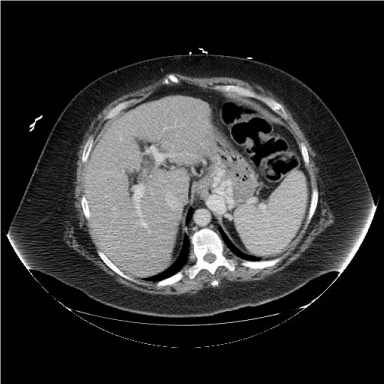
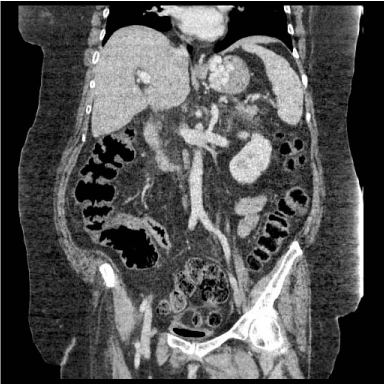
Figure 2
Figure 2
CT transverse image, intravenous contrast but no oral contrast,
defining large isolated gastric variceal set. Inflow seems to come from short
gastric veins from the splenic hilum
After due consideration, discussion with extensively experienced national colleagues and literature review we considered B-RTO but instead decided to initially assess portal pressure directly as the initial portion of a TIPS procedure. After full and thorough informed consent, clarification of a probable stepwise process the patient agreed to initial TIPS procedure, possible later gastric variceal occlusion. Under general endotracheal anesthesia we initiated transjugular access to the hepatic venous system. Transhepatic access to the portal system was slightly more difficult than usual, possibly due to comparatively low corrected portal pressure. Once in the portal system corrected mean portal pressure was measured 13mm Hg, consistent with wedged hepatic vein pressure. Again the treatment options were considered and we decided to proceed with TIPS creation. Once completed, corrected portal pressure fell to approximately 8mm Hg.
Figure 2B
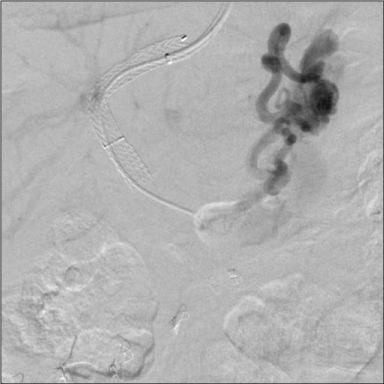
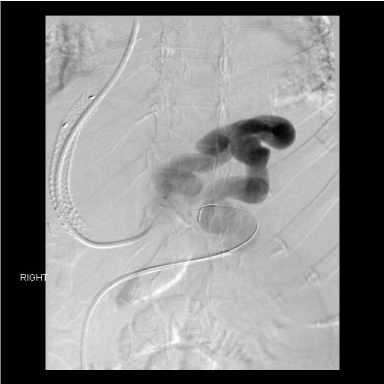
Figure 3
Figure 3
Post TIPS angiographic anatomy defining the large gastro-renal
venous shunt. The TIPS led to profound encephalopathy and coma.
We decided to observe patient outcome; the patient became profoundly encephalopathy, mental status severely impaired, ammonia level 145. We then reevaluated CT and TIPS angiographic anatomy (Figure 3), reconsidering B-RTO. Given lack of experience with B-RTO in the United States, ease of access to the portal system through the TIPS shunt and personal experience with treatment alternatives; we planned an alternative. While confirming reduction of the portasystemic outflow with an Amplatzer ll occluder; we sequentially began to inject gelfoam soaked with sodium tetradecyl sulfate as a sclerosing agent. We injected the sclerosing pledgets into the multiple major primary inflow sources to the gastric varicies. There was no suggestion of pulmonary emboli or fibrosis, encephalopathy altered level of consciousness resolved over 48 hours. Follow up endoscopy and CT at three months showed no evidence of gastric varicies; no gastrointestinal hemorrhage has occurred over two years follow up.
Figure 4
Figure 4
Case three, after TIPS two years prior a large portasystemic shunt
remained a huge, high flow gastrorenal shunt. This shunt remained open
over that interval, ultimately repeat gastrointestinal hemorrhage occurred, A
huge endoscopically confirmed from the isolated gastric varicies.
case 2
Within six months of Case 1 an exactly similar patient presented
acutely with profound gastrointestinal hemorrhage, hemoglobin
reduced to 5 grams and endoscopic demonstration of isolated fundal
gastric varicies. With our experience from Case 1 we proceeded to
CT defining arterial and venous phase, defining the large gastric
varicies and a large gastrorenal portasystemic shunt. We proceeded
in exactly the same sequence; TIPS creation day 1 during which we
found a low corrected portal vein pressure, likely due to the large
portasystemic collateral. Multiple inflow branch occlusions were
accomplished while outflow was diminishing after Amplatzer II plug
placement successful on day 2. The 24 hour interval was useful to
assess inflow anatomy and plan sequential outflow and subsequent
inflow branch occlusion. Again, a three month endoscopic and CT
follow up demonstrated no evidence of gastric varicies. No recurrent
gastrointestinal hemorrhage at one year.
case 3
Our third case developed in a slightly different sequence. Initial
bleeding two years prior was related to large isolated gastric fundal
varicies. This was before we fully understood the importance large
varix diameter and torrential flow in such gastrointestinal hemorrhage
as different from esophageal varicies. At initial presentation a TIPS
was performed although measured corrected portal venous pressure
was low. The large gastric varix with gastrorenal shunt remained
despite the TIPS. Bleeding seemed controlled, the patient did not
develop significant hepatic encephalopathy. She was discharged and
stable for two years. Then a repeat episode of severe gastrointestinal
hemorrhage developed. The CT defined the large gastric varicies
with gastrorenal shunt outflow. The patient was returned to the
interventional laboratory. From a transjugular approach we
investigated the TIPS, it remained patent. Pressure in the portal
system was not elevated. A large, inflow coronary vein to the large
isolated gastric fundal varicies was accompanied by a very small short
gastric vein inflow. A huge gastrorenal outflow was confirmed (Figure
4). We then dilated the TIPS shunt to confirm stability. We proceeded
to both primary inflow and outflow occlusion of the gastric varicies
with Amplatzer II occlusion devices (Figure 5). We did not chase the
small short gastric inflow believing it not significant if outflow totally
occluded. Follow-up at 3 months demonstrated minimal residual
gastric varicies and no further bleeding.
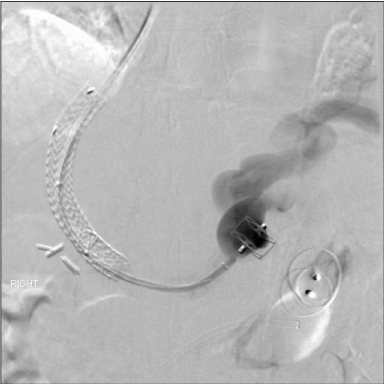
Figure 5
Figure 5
After mapping single inflow from the coronary vein and primary
outflow from the gastrorenal shunt of the isolated gastric varicies, we
occluded outflow initially with a 22mm Amplatzer II occlusion device. We
followed immediately by occluding inflow with a 16mm diameter Amplatzer II
occlusion device. The patient demonstrated no further bleeding to 6 months
and no gastric varicies at 3 month follow up CT.
case 4
51 year old female admitted emergently for upper gastrointestinal
hemorrhage. Admission two years previous for the same diagnosis,
endoscopy defined esophageal varicies; they were successfully
banded. The patient was lost to follow up but reported abstinent from
alcohol. She recently began drinking alcohol, leading to the current
episode. The patient has Type II diabetes and hypertension, but
is otherwise reported in good health. Endoscopy at this admission
defined a large gastric variceal set with a single esophageal varix
defined. It was therefore unclear if the current pattern represented
EOG1 esophagogastric varicies or IGV1 isolated gastric varicies.
Portal and hepatic vein ultrasound defined hepatopetal portal vein
blood flow. There was no thrombus in the portal vein and good
hepatic vein flow. The INR was 1.2; Total bilirubin 0.8; creatinine
1.0; ammonia level 51. This gave a Meld Score of 4, indicating 90
day mortality 3%. We were asked to evaluate the patient for TIPS to
control gastric variceal hemorrhage. A CT abdomen was obtained
with intravenous contrast only. This demonstrated a dense gastric
variceal set, seemingly supplied only from a prominent coronary
vein, no obvious short gastric supply, no obvious esophageal varicies
and a large, approximately 9mm diameter draining gastrorenal vein.
Given this information we suspected IGV1 isolated gastric varicies.
We proceeded to TIPS after fully informed consent obtained. We
decided if corrected portal pressure was significantly elevated
TIPS alone may be sufficient treatment. A normal corrected portal
pressure would not resolve the gastrorenal shunt, high shunt flow
would be maintained with a high likelihood of further variceal
distention and probable massive gastric variceal hemorrhage. In
addition the increasing flow in the varicies bypassing the liver would
lead to progressive encephalopathy. Standard TIPS procedure was
straightforward, first pass entry to the portal venous system; directly
measured corrected portal venous pressure 13mmHg, minimally
elevated. The large coronary vein supply to the isolated gastric varicies
and drainage through the gastrorenal shunt. There was a single
additional small supplying vein branch to the gastric varicies arising
from the renal vein. We completed the TIPS procedure, dilating the
transhepatic course to 8mm, placed a Viator 10mm diameter covered
stent and post dilated to 8mm. Reevaluation of gastric variceal flow
demonstrated persistent flow toward the varicies, even though post
TIPS corrected portal pressure only 5mmHg mean. Venous access
to the left renal vein from the left common femoral vein was then
accomplished. Occlusion of the gastric variceal inflow coronary vein
and gastrorenal vein outflow near simultaneously was accomplished
without difficulty. Amplatzer II plugs, 14 mm into the inflow
coronary vein and 16 mm into the outflow gastrorenal vein were
placed through Boston Scientific curved 7F renal sheaths (Figure 5).
We then reevaluated corrected portal vein pressure, 6mmHg mean.
Follow up angiogram demonstrated essentially no flow to the gastric
varicies. The patient did well, discharged 2 days later without further
bleeding. Abdominal CT at 1month defined no evidence of the gastric
varicies; there was no bleeding in the interval.
Discussion
We have now identified 4 cases of isolated gastric varicies, IGV1
(Sarin, 1) at one institution in 18 months. We believe this may indicate
a genetic bias in our population or more likely under diagnosis of this
condition in the general population. Such possible under diagnosis
may be related to endoscopic difficulty defining the gastric fundus, the
lack of effective treatment of this condition and the massive resultant
bleeding which can be rapidly fatal [2]. Given the torrential blood
flow through the portasystemic shunt even small ulcerations in these
varicies can lead to death before reaching treatment. Endoscopic
treatment of gastric varicies is very challenging and therefore
limited due to gastric fundal size, complexity and interconnection
of varicies which means that placing a hole into such a complex,
high flow vascular system could produce worse hemorrhage and
exsanguinations [2,8,9] even using the latest tissue occlusive material
such as 2-octyl cyanoacrylate. The anatomy of IGV1 has been
clarified and a seemingly completely successful treatment plan, in
the short term, defined. By occluding both inflow and outflow of the
gastric variceal network we believe we have removed the etiology of
gastrointestinal hemorrhage in a more complete but similar manner
to B-RTO only sclerotherapy [10]. The TIPS is essential for accurate
definition of portal pressure and variceal anatomy, especially possible
multiple inflow sources. A non elevated corrected portal vein
pressure indicates that TIPS, a decompressive procedure, alone will
be unlikely to resolve flow through a large, tortuous portasystemic
communication [10]. However, the liver disease underlying this
physiology, hepatitis or cirrhosis produces the increased resistance
to normal portal venous blood flow. This results in various collateral
portasystemic flow patterns. Defining why esophageal (EOG1),
esophagogastric (EOG2) or isolated gastric varicies (IGV1), all of
which can result in life threatening hemorrhage, develop has not
yet been solved. Indeed, understanding the why of development
of Ascities,, likely due to preferential stagnant flow through the
mesenteric veins has yet to be clarified. But understanding this wider
difference may be an easier step than determining why different
variceal collateral patterns develop. Suggested treatment plans for
isolated gastric varicies include TIPS alone. But as cases 1 and 3 have
shown TIPS alone could result in terminal encephalopathy (Case 1)
or continued gastric variceal flow and eventual rebleeding (Case 3).
The presumed goal of the Japanese groups treatment plan, Balloon
occlusion retrograde transvenous injection (B-RTO) of tissues
adhesive is an attempt to completely seal the complex, interconnected
isolated gastric variceal network. Without controlling inflow the
retrograde penetration of cyanoacrylate will be unpredictable.
Additionally the volume and tortuosity of the isolated gastric variceal
network, adhesive injection pressure and time will result in further
variability of retrograde penetration into the network. Insufficient
volume of occlusion could lead to development of alternative outflow
pathways and continued hemorrhage risk. Too vigerous injection
could lead to fatal portal venous occlusion as reported in one of
Morimasas cases. We believe the stepwise treatment plan we have
presented represents a more complete understanding of variceal
anatomy and physiology; a graduated treatment plan to avoid under
treatment or overtreatment. This understanding and the inflow and
outflow pathway occlusion with the Amplatzer II device represents
a more controlled, reproducible, safer treatment option. The range
of pathologic processes possible within the presentation of isolated
gastric varicies is neither a one size fits all treatment plan nor a treat
it and forget it option. Obviously the underlying liver dysfunction
due to portal triad scaring must be monitored and evaluated for
progression, TIPS patency, development of alternative portal flow
pathways and alternate sequels.
Conclusion
We have presented a treatment plan refinement for isolated gastric varicies (IGV1, Sarin). It combines the primary option prevalent in the United States, TIPS only, with the primary treatment plan recommended in Japan, Balloon occlusion retrograde transvenous injection of tissue adhesive (B-RTO). We have added physiologic assessment, defining corrected portal venous pressure as a treatment decision point. We also suggest inflow anatomic mapping of the complex interconnected gastric variceal network to determine the best means of inflow occlusion. We have used the Amplatzer II occlusive plug to seal large primary inflow and outflow veins because of stability of placement in the high flow, tortuous gastrorenal shunt. We have added sclerosant soaked gelfoam sponge injection into smaller multiple inflow sources to gastric variceis after outflow occlusion with the Amplatzer II. This outflow vessel is often difficult to ascend because of severe tortuosity precluding catheter stability adjacent to the left renal vein. The ease of use of the Amplatzer II in high flow positions as well as the ability to remove the device after deployment before release add to procedure safety and successful isolated gastric variceal occlusion.
References
- Sarin SK, Lahoti D, Saxena SP, Murthy NS, Makwana UK. Prevalence,Classification and Natural History of Gastric Varicies: A Long-Term Follow-up Study in 568 Portal Hypertension Patients. Hepatology. 2005; 6: 1343-1349.
- Hashizume M, Akahoshi T, Tomikawa M. Management of Gastric Varicies. Journal of Gastroenterology and Hepatology. 2011; 26: 102-108.
- Ryan BM, Stockbrugger RW, Ryan JM. aPathophsiologic, Gastroenterologic and Radiologic Approach to the Management of Gastric Varicies. Gastroenterology. 2004; 126: 1175-1189.
- Matsumoto A, Kitamoto M, Imamura M. Three-Dimensional Portography Using Multislice Helical CT is Clinically Useful for Management of Gastric FundicVaricies. American Journal of Radiology. 2001; 176: 899-905.
- Hirota S, Matsumoto S, Tomita M, Sako M, Kono M. Retrograde Transvenous Obliteration of Gastric Varicies. Radiology. 1999; 211: 349-356.
- Akahoshi T, Hashizume M, Tomikawa M, Kawanaka H, Yamaguchi S, Konishi K, Kinjo N, et al. Long-Term Results of Balloon- Occluded Retrograde Transvenous Oblireration for Gastric Variceal Bleeding and Risky Gastric Varicies: a 10 Year Experience. Journal of Gastroenterology and Hepatology. 2008; 23: 1702-1709.
- Katoh K, Sone M, Hirose A, Inoue Y, Fujino Y, Onodera M. Balloon Occluded Retrograde Transvenous Obliteration for Gastric Varicies: The Relationship Between the Clinical Outcome and Gastrorenal Shunt Occlusion. BioMed Central Medical Imaging. 2010; 10: 2.
- Wu BU, Carr-Locke DL. The Problem with Gastric Varicies. Medscape General Medicine. 2006; 8: 72.
- Goff JS. Treatment of Gastric Varicies in 2005: Is There a Role for Endoscopy? Visible Human Journal of Endoscopy. 2005; 4: 1-4.
- Ninoi T, Nakamura K, Kaminou T, Nishida N, Sakai Y, Kitayama T, et al. TIPS Versus Transcatheter Sclerotherapy for Gastric Varicies. American Journal of Roentgenology. 2004; 183: 369-376.