Case Report
Operative Repair of Juxtra-Renal Abdominal Aortic Aneurysm with a Horseshoe Kidney Utilizing Cardiopulmonary Bypass
Marvin E. Morris1*, Natasha Saiyed 1, Marc A. Norris1, Neal C. Hadro1and Josesph E. Flack2
1Department of Surgery, Tuft University School of Medicine, USA
2Baystate Heart and Vascular, University of Massachusetts School of Medicine, USA
*Corresponding author: Marvin E. Morris, Department of Surgery, Tuft University School of Medicine, Baystate Vascular Services, 3500 Main St, Suite 201, Springfield, MA 01107, USA
Published: 14 May 2016
Cite this article as: Morris ME, Saiyed N, Norris MA,
Hadro NC, Flack JE. Operative Repair
of Juxtra-Renal Abdominal Aortic
Aneurysm with a Horseshoe Kidney
Utilizing Cardiopulmonary Bypass. Clin
Surg. 2016; 1: 1007.
Abstract
Repair of abdominal aortic aneurysms in the presence of horseshoe kidney (AAA-HSK) can be
technically challenging due to the variable renal arterial blood supply. Precise pre-operative
knowledge of the anomalous renal perfusion is requisite for successful planning and treatment of
AAA-HSK. We describe a case using a left heart cardiopulmonary bypass during operative repair of
an 5.4 cm juxta-renal AAA-HSK to maintain renal perfusion during revascularization.
Keywords: Abdominal aortic aneurysms; Horse shoe kidney (AAA-HSK); Juxta-renal AAAHSK
Introduction
Operative challenges in the repair of HSK-AAA can frustrate even the most experienced surgeons. The variable arterial supply of the HSK-AAA, can involve ectopic or multiple renal arteries arising from the aorta, the iliac vessels or from the aneurysm itself, as described in the Eisendrath classification [1]. The extent of renal parenchyma perfusion by accessory renal arteries is a key determinant if these arteries need to be either bypassed, ligated or incorporated into the operative repair [2,3]. We report a case of a 64-year-old male presenting for elective repair of a 5.4cm abdominal aortic aneurysm in the setting of a congenital pelvic kidney with a Eisendrath V renal perfusion, with two distinct renal arteries originating from the aortic bifurcation. To maintain retrograde perfusion of the HSK, a left heart cardiopulmonary bypass circuit was utilized during operative repair
Case Presentation
This is a 64-year-old gentleman presenting for elective repair of an enlarging 5.4 cm juxtra-renal AAA with a pelvic midline HSK, with a recent increase in size from 4.8 to 5.4 cm. Preoperative
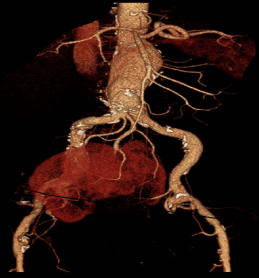
CT angiography revealed two distinct renal arteries originating from the aortic bifurcation (Figure 1).
In our opinion we did not have an endovascular approach suitable to his anatomy therefore
we opted to perform operative repair while utilizing a mechanism to maintain perfusion of the
HSK. In conjunction with our cardiac surgical team, we performed a partial right femoral artery-tovein
cardiopulmonary bypass for retrograde perfusion with a separate circuit in the iliac artery to
maintain renal ostial perfusion during surgical repair.
Operative details include suprarenal aortic exposure for the short proximal aneurysm neck.
Following distal aortic bifurcation exposure, a separate right groin incision was created to expose
the right common femoral artery and vein. Arterial and venous perfusion catheters were placed
for creation of a partial right femoral artery-to-vein cardiopulmonary bypass circuit. After
systemic heparinization with 22,000 units, distal aortic clamping was obtained just above the
aortic bifurcation, followed by a supra-renal aortic cross clamp, just below the superior mesenteric
artery. Distal visceral perfusion during the proximal anastomosis was achieved by means of a right
femoral artery-to-vein circuit. The iliac arteries were surgically controlled for creation of the distal
anastomosis due to the inability to visualize the distal sewing ring. A separate catheter inserted in
the right common iliac artery for ostial perfusion of the renal artery during the distal anastomosis.
Ostial hypothermic renal artery perfusion was performed with cold blood infusion. A mean arterial
pressure of 60 mmHg and average flow of 300-400 ml/min was maintained. Cessation of the
cardiopulmonary bypass circuit occurred following hemostasis. The patient tolerated the operationwell and was discharged home on postoperative day six. He has not experienced any complications following his operation and his renal
function remains unchanged at 1 year follow-up.
Figure 1
Figure 1
3-D CTA reconstruction of a 5.4 juxtra-renal AAA, with pelvic HSK,
with renal arteries originating from the aortic bifurcation (Eisendrath class V).
Discussion
The frequency of HSK is 0.25% in the population and 0.12%
in patients requiring abdominal aneurysm repair [4]. Repair of
abdominal aortic aneurysms in the presence of a horseshoe kidney
can be technically challenging due to the variable renal arterial blood
supply and proportional perfusion of accessory renal arteries. Patients
with horseshoe kidney can have ectopic or multiple renal arteries.
They may arise from the aorta, the iliac vessels or from the aneurysm
wall itself [5,6]. Logistical concerns include the variable renal arterial
blood supply and the extent of renal parenchyma supplied by
accessory renal arteries. Either the transperitoneal or retroperitoneal
approach to open surgical repair is technically feasible, but morbidity
and mortality are high, due to complex anatomy of the aberrant HSK
[5-8]. These anomalous vessels can be challenging to manage in the
operative setting. Therefore, when technically feasible, endovascular
aneurysm repair (EVAR) is treatment of choice. EVAR is well
established for HSK-AAA and is associated with decreased morbidity
and mortality [9-12]. This was not an option in our case due to lack of
requisite anatomy for either standard or fenestrated EVAR.
In our case we felt it was necessary to perfuse the kidney during
operative repair. Both renal arteries originated form the aortic
bifurcation, hence, distal aortic clamping would lead to unnecessary
warm ischemia time. To mitigate the potential renal morbidity we
opted to maintain HSK perfusion during repair. Logistically, partial
cardiopulmonary bypass (CPB) preserves distal end-organ perfusion
during aortic cross-clamping and is aimed to decrease ischemic
time. Partial CPB is used during thoracic aortic surgery to provide
perfusion to the spinal cord and visceral vasculature. During partial
bypass, a portion of the stroke volume is deviated to the descending
aorta. Pulmonary perfusion remains intact, thus maintaining the
cardiac and respiratory functionality [13-17]. A femoral arteryfemoral
vein bypass carries the same functional principles as a left
heart bypass. The circuit is composed of both venous and arterial
cannulas. Following cannulation of the vein and artery, a portion of
the right atrial blood volume is oxygenated and subsequently infused into the femoral artery. A membrane oxygenator is needed due to
drainage from the right atrium and full heparinization is required
[12,14]. Selective visceral perfusion is performed by connecting the
perfusion catheters secured to the Y limb, in our case to the renal
artery ostia for retrograde perfusion.
Conclusions
The therapeutic armamentarium of treatment of HSK-AAA is dynamic and reflects the complexity of arterial perfusion to the HSK-AAA. EVAR for AAA-HSK is the preferred approach and circumvents the operative challenges of surgical exposure, but even evolving technology with fenestrated devices may not be applicable to complex AAA-HSK anatomy. Partial CPB is a safe and effective approach to facilitate HSK perfusion during operative repair. It can serve as a novel addition to the existing repertoire of dynamic adjuncts used to repair this complex congenital anomaly.
References
- Eisendrath DN, Phifer FM, Culver HB. Horseshoe kidney. Ann Surg. 1925; 82: 735-764.
- Bietz DS, Merendino KA. Abdominal aneurysm and horseshoe kidney: a review. Ann Surg. 1975; 181: 333-341.
- Sidell PM, Pairolero PC, Payne WS, Bernatz PE, Spittel JA Jr. Horseshoe kidney associated with the abdominal aorta. Mayo Clin Proc. 1979; 54: 97-103
- Stroosma OB, Koostra G, Shurink GW. Management of aortic aneurysm in the presence of a horseshoe kidney. Br J Surg. 2001; 88: 500-509.
- O’Hara PJ, Hakim AG, Hertzer NR, Krajewski LP, Cox GS, Beven EG. Surgical management of aortic aneurysm and coexistent horseshoe kidney: review of a 31-year experience. J Vasc Surg. 1993; 17: 940-947.
- .DeVirgilio C, Gloviczki P, Cherry KJ, Stanson AW, Bower TC, Hallett JW Jr, et al. Renal Artery Anomalies in Patients with Horseshoe or Ectopic Kidneys: the challenge of aortic reconstruction. Cardiovasc Surg. 1995; 3: 413-420.
- Shortell CK, Welch EL, Ouriel K, Green RM, Deweese JA. Operative management of coexistent aortic disease and horseshoe kidney. Ann Vasc Surg. 1995; 9: 123-128.
- Crawford ES, Coselli JS, Safi HJ, Martin TD, Pool JL. The impact of renal fusion and ectopia on aortic surgery. J Vasc Surg 1988; 8: 375-383.
- Cronwenwett JL, Krupski WC, Rutherford RB. Abdominal aortic and iliac aneurysms. In: Rutherford RB ed. Vascular Surgery. 5th ed. Philadelphia, PA: Saunders, 2000. pp 1272-1273.
- Aquino RV, Rhee RY, Muluk SC, Tzeng EY, Carrol NM, Makaroun MS. Exclusion of accessory renal arteries during endovascular repair of abdominal aortic aneurysms. J Vasc Surg 2001; 34: 878-884.
- Ruppert V, Umscheid T, Reiger J, Schmedt CG, Mussack T, Stelter WJ, et al. Endovascular aneurysm repair: Treatment of choice for abdominal aortic aneurysm coincident with a horseshoe kidney? Three case reports and review of literature. J Vasc Surg. 2004; 40: 367-370.
- Loftus IK, Thompson MM, Fishwick G, Boyle JR, Bell PR. Endovascular repair of aortic aneurysms in the presence of a horseshoe kidney. J Endovasc Surg. 1998; 5: 278-281.
- Kaplan DB, Kwon CC, Marin ML, Hollier LH. Endovascular repair of abdominal aortic aneurysms in patients with congenital renal vascular anomalies. J Vasc Surg. 1999; 30: 407-415.
- Di Eusanio M, Schepens MA, Morshuis WJ, Dossche KM, Pacini D, Kazui T, et al. Brain protection using antegrade selective cerebral perfusion: a multicenter study. Ann Thorac Surg. 2003; 76: 1181-1189.
- Strauch JT, Spielvogel D, Lauten A, Galla JD, Lansman SL, McMurthy K, et al. Technical advances in total aortic arch replacement. Ann Thorac Surg. 2004; 77: 581-589.
- Strauch JT, Spielvogel D, Lauten A, Zhang N, Rinke S, Weisz D, et al. Optimal temperature for selective cerebral perfusion. J Thorac Cardiovasc Surg. 2005; 130: 74-82.
- Halstead JC, Meier M, Wurm M, Zhang N, Spielvogel D, Griepp RB, et al. Optimizing selective cerebral perfusion: deleterious effects of high perfusion pressures. J Thorac Cardiovasc Surg. 2008; 135: 784-791.