Research Article
Are Previous Experience with Laparoscopy and the Inclusion in a Group of Surgeons with Prior Robotic Experience Required to Perform Robotic Rectal Cancer Surgery?
Rosa M Jiménez-Rodríguez1*, Verónica Pino-Díaz1, José Manuel Díaz Pavón1, Juan Manuel Praena Fernandez2, Javier Padillo1 and Fernando de la Portilla1,3
1Department of General and Digestive Surgery, Colorectal Surgery Unit, University of Seville, Spain
2Department of Statistical analysis, “Virgen del Rocío” University, Spain
3Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas, Instituto de Salud Carlos III, Spain
*Corresponding author: Rosa M Jimenez-Rodriguez, Department of General and Digestive Surgery, Colorectal Surgery Unit, University of Seville, 41013 Sevilla, Spain
Published: 01 Nov, 2018
Cite this article as: Jiménez-Rodríguez RM, Pino-Díaz V,
Díaz Pavón JM, Praena Fernandez JM,
Padillo J, de la Portilla F. Are Previous
Experience with Laparoscopy and the
Inclusion in a Group of Surgeons with
Prior Robotic Experience Required
to Perform Robotic Rectal Cancer
Surgery?. Clin Surg. 2018; 3: 2191.
Abstract
Introduction: Robotic rectal surgery could decrease the learning curve but the necessity of previous
experience with laparoscopy or the importance of being part of a group of surgeons with previous
robotic experience have not been analyzed yet.
Material: A total of 81 patients with rectal cancer were included in the study. Patients were divided
into 3 groups of 27 patients each that were assigned to three surgeons. One had previous laparoscopic
experience, but was included in a group without prior robotic experience. Another surgeon also had
previous experience with laparoscopy and was included in a group with prior robotic experience.
The third surgeon had no prior experience with laparoscopic rectal cancer surgery, but he was
included in a group with robotic experience.
Results: Conversion and intra-operative complication rates were lower in patients treated by the
two surgeons who were included in a group with prior robotic experience. The CUSUMOT curve also
showed that operative time decreased earlier in these two surgeons, the incidence of postoperative
re-interventions was higher among these two groups.
Conclusion: No previous experience with laparoscopic surgery for rectal cancer is required to begin
robotic surgery if it is performed by a group of surgeons with prior robotic experience.
Keywords: Learning curve; Rectal cancer; Robotics
Introduction
Laparoscopic rectal cancer surgery is a complex procedure requiring a high number of cases to
overcome the learning curve. According to several studies, 40-90 procedures must be carried out
to obtain standardized outcomes, although it has been demonstrated that oncologic results do not
change during the learning period [1-6].
Robotic surgery, which provides surgeons with a three-dimensional surgical view, enhanced
dexterity and precision in the use of the surgical instruments and the ability to control the operative
field by manipulating the camera, may reduce the number of cases required to overcome the learning
curve for mesorectal excision. Indeed, several studies report a shortening in the learning curve from
15 to 35 cases with the use of this robotic technique [7-10].
Some studies claim the importance of implementing preclinical training programs in robotic
surgery, but they do not evaluate the necessity of prior experience with laparoscopic rectal cancer
surgery, either in relation or not to these robotic training programs [11].
Consequently, the aim of the present study is to assess whether prior experience with laparoscopic
rectal cancer surgery and the inclusion in a group of surgeons with previous robotic experience
in the treatment of this condition may affect the learning curve and the short-term clinical and
oncologic outcomes after surgery.
Table 1
Material and Methods
A prospective database of 250 patients was analyzed. The
patients underwent surgery for rectal cancer from January 2008
through January 2016 in the Coloproctology Unit of the University
Hospital Virgen del Rocío in Seville. The protocol established by the
multidisciplinary assessment committee of our hospital determined
whether patients should be administered preoperative long-course
chemoradiotherapy.
Of the 250 patients in the database, 81 patients with rectal
cancers located <16 cm from the anal verge were finally included in
our study. They were divided into three groups of 27 patients and
were assigned to three different surgeons who were robotic surgery
novices. Surgeon 1 had a large experience with rectal cancer surgery,
both laparoscopic and open approach (more than 100 cases in each
procedure), but was included in a group without prior experience
in robotic surgery. Surgeon 2 also had extensive experience in both
laparoscopic and open rectal cancer resections (more than 100 cases
in each technique) and was included in a group with experience in
robotic colorectal resections. Surgeon 3 had limited experience with
rectal resection (less than 20 cases in each procedure), but he was part
of a group with a experience of over 150 robotic rectal resections.
None of them had previous robotic experience in rectal cancer or in
other robotic procedures.
The number of cases allocated to each surgeon (27) was established
considering previous publications [7-8,10,12-13], which suggest that
27 is the lowest number of cases necessary to overcome the learning
curve in robotic rectal cancer resections.
Variables
Besides age and sex, the following variables were included in
our study: BMI, tumor location, tumor stage, neoadjuvant therapy,
surgical procedure (anterior resection with or without protective
ileostomy, Hartmann procedure or abdominoperineal resection),
intra and postoperative complications and conversion rates.
Operative time was defined as the time taken from
pneumoperitoneum induction to completion of skin closure.
Console time was considered as the time spanning from the
beginning of the resection using the console to the removal of the
specimen through a Pfannenstiel incision plus the time required to
perform the anastomosis. In the case of Hartmann procedures and
abdominoperineal resections, console time ends when the specimen
section is completed. Specimen length, margins and postoperative
stage were also recorded.
A written consent form signed by the patients was required
before surgery to record all the variables in a database reviewed and
approved by an ethical committee.
Figure 1
Statistical Analysis
Qualitative variables were expressed as absolute frequencies
and percentages and quantitative variables as means and Standard
Deviation (SD) or as median and interquartile range ([p25; p75]) for
data with non-symmetrical distribution. The statistical analysis of
data was performed on contingency tables using Fisher's exact test or
chi square test to analyze qualitative variables and variance analysis or
Student's t-test for quantitative variables. Kruskall-Wallis or Mann-
Whitney U tests were used to examine non-Normal quantitative
variables.
The 81 patients included in our study were divided into 3 groups
and were assigned to three different surgeons. Groups were compared
with one another as follows: a group formed by patients allocated to
surgeons 1 and 2 (with previous experience with laparoscopic rectal
cancer resection) was compared with the group assigned to surgeon
3 and another group formed by patients assigned to surgeons 2 and
3 (included in a group with experience in robotic colorectal surgery)
was compared to the group allocated to surgeon 1. Considering the
earliest outcomes, variables showing a statistically significant value of
p<0.05 underwent multivariate analysis.
Patients in each group were chronologically arranged from
the earliest to the latest date of surgery. CUSUMOT (Operating Time) was
determined for every patient by means of the following equation:
CUSUMOT value=C (i+1) + (Xi-mean Xi)
As no patient died during the study, it was not necessary to
calculate the risk-adjusted CUSUM value (RA-CUSUM).
In order to calculate the CUSUM plot to measure the correlation
between success and learning curve (CUSUMsu), "success" was defined
as a surgical procedure not showing any of the following parameters
or events: conversion to open surgery, intra-operative complications,
postoperative complications and/or mortality. We then applied the
equation:
CUSUMSU value (n) = Σ (xi - T) + (-1)xipi
where xi represents the presence or absence of the event being
studied; T stands for the observed probability of the event, in this
case the probability of "success" (64.2% in this case) and pi is the
individual probability of failure in our population, once we apply to
every value a logistic regression model which included the variables
of age, gender, BMI, tumor location and operating time and which
met the requirements for success.
Data were analyzed using the statistical software IBM SPSS 19.0
(SPSS. Inc., Chicago, IL).
Figure 2
Results
A total of 81 patients were included in the study; 52 males and
29 females with a mean age of 64,62 (SD=10.78) and mean BMI of
27.50 (SD=3.73). The procedures included 32 anterior resections with
ileostomy, 29 anterior resections without ileostomy, 5 Hartmann
procedures and 15 abdominoperineal resections. All patients were
diagnosed of rectal cancers located 9.01 cm (SD=4.38) from the anal
verge. All the characteristics are shown in (Table 1).
Our study population was divided into 3 groups according to the
surgeon in charge of the surgical procedure. Differences between the
three groups can be seen in (Table 1).
The comparative analysis of the three groups revealed no
differences. A new analysis was carried out between the two groups
of patients assigned to the two surgeons with previous laparoscopic
experience and the group assigned to the surgeon without such
experience. Again the results revealed no significant differences. Next,
a combined analysis was performed of the cases assigned to surgeons
2 and 3, who were included in groups with robotic experience, and
the cases assigned to surgeon 1, who was not included in a group
with robotic experience. The univariate analysis yielded significant
differences in conversion rates (5.6 % vs. 25.9%) p=0.014, intraoperative
complications (1.9% vs. 14.8%) p=0.040 and postoperative
complications (27.8% vs. 7.4%) p=0.043 (Table 1).
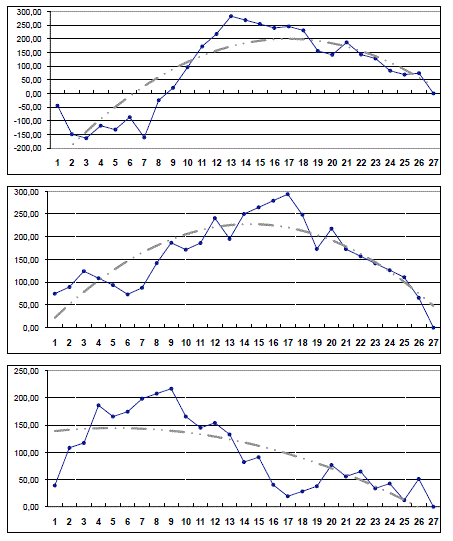
The comparison of the CUSUMOT curves (Figure 1) plotted for
each surgeon showed a decrease in operative time after cases 13, 17
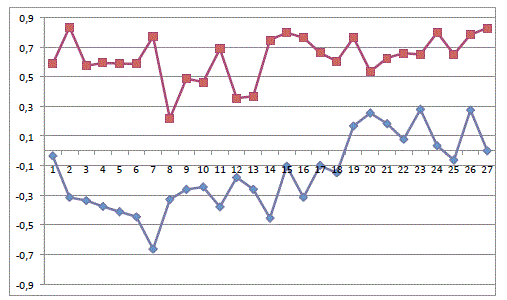
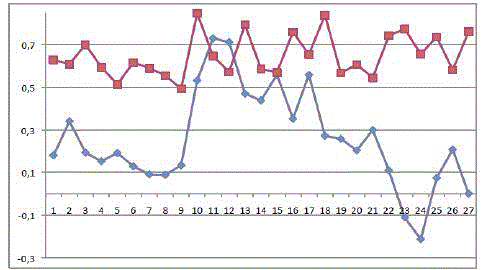
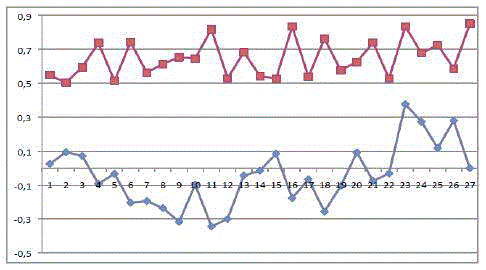
and 9 for each surgeon respectively. CUSUMSU curves (Figures 2-4),
which show the probability of success for each surgeon; reveal how
success rate increases as the number of cases progresses (upward
trend). Moreover, a higher probability of success is observed in the
cases assigned to surgeons 2 and 3 (p>0.5) than in the cases assigned
to surgeon 1.
Figure 3
Figure 4
Table 2
Discussion
Rectal cancer surgery is a complex procedure which involves
total or partial mesorectal excision, a precise and highly complicated
technique. In order to obtain satisfactory oncologic outcomes, the
dissection should proceed through the "Holy plane" described by
Professor Heald [14].
The complexity of this surgical technique is determined by such
factors as the reduced operative field of the pelvic cavity, particularly
in the case of male patients; the tumor diameter which may impair
visualization at this level; the probability of infiltration of adjacent
organs like the prostate or the vagina and the use of neoadjuvant
therapy, which affects tissue elasticity.
The use of the laparoscopic technique for mesorectal excision
introduces a series of factors which increase the complexity of the
procedure: two-dimensional surgical view, loss of the visual depth
perception, limited mobility of straight laparoscopic instruments
with fixed tips and the need for an assistant to move the camera and
other instruments. As a result, laparoscopic rectal cancer surgery has
an estimated learning curve of 65 cases [1-5].
Robotic surgery, on the other hand, provides a three-dimensional
surgical view, a stable camera platform and articulated instruments.
Hence, it offers the possibility of shortening the learning curve to 24-
27 cases [15].
Nevertheless, the analysis of the learning curve of robotic rectal
surgery has not taken into account the previous experience of
surgeons with laparoscopy or their inclusion in a group with prior
robotic experience.
Recently, Guend et al. [11] published a study to determine the
number of cases required for establishing a robotic colorectal cancer
surgery program as well as the impact of this program on the learning
curve of individual surgeons. According to the results, establishing
this program requires 75 cases. Moreover, the authors claim that this
sort of programs may shorten the number of cases in an individual
surgeon's learning curve to 25-30. In our study, we have observed that
the inclusion in a group of surgeons with previous robotic experience
affects both conversion rates and operative time. However, the
study conducted by Guend et al. [11] does not analyze rectal tumors
exclusively, nor does it indicate the previous laparoscopic experience
of the surgeons included in the program or of the novice surgeons.
There are no reports in the literature which analyze the influence
of the correlation between previous laparoscopic experience and the
inclusion in a group of surgeons with robotic prior experience on
both the learning curve and the short-term outcomes of rectal cancer
surgery.
Prior laparoscopic experience has been analyzed by Kim et al.
[16], who describe a one-step transition from open surgery to robotic
surgery. They analyzed 100 consecutive cases performed by the two
surgeons included in the study: surgeon A had no previous experience
with laparoscopic rectal cancer surgery but he had some experience
in open surgery and surgeon B had previous laparoscopic experience.
Outcomes in relation to operative time and distal resection margins
were more satisfactory in the cases performed by the surgeon without
previous laparoscopic experience. However, the authors do not justify
these results. They do not explain whether the surgeon was assisted by
another surgeon or was part of a group of surgeons with previous
robotic experience, which could justify the shorter operative time and
the reduction in conversion rates.
Although some basic laparoscopic experience is necessary in
order to place the trocar and to manage the patient when it is not
possible to continue with the robotic procedure, no prior experience
with laparoscopic surgery for rectal cancer is necessary, according
to the results of our study. The results reported for the surgeon 1,
who had not overcome the learning curve for laparoscopy, and for
the other two surgeons showed no significant differences. As a result,
intra-operative complications were not higher in the cases performed
by surgeon 1. Also, he performed the same number of conservative
surgeries of the sphincter as the other two surgeons but he did not
perform more protective ileostomies.
Unlike to what we might expect from previous surgical
experience, the longest operative time was observed in surgeon 1,
whereas surgeons 2 and 3 obtained similar operative times. Probably,
the inclusion in a group with prior robotic experience facilitated
motion of the patient cart arms and the shortening of operative time.
The surgeon with previous experience with laparoscopic surgery
presented with a lower percentage of postoperative complications in
comparison to surgeons 2 and 3. For surgeon 3, this could be justified
by the lack of experience in rectal cancer surgery, but this is not
applicable to surgeon 2.
In our study, the surgeon without previous laparoscopic
experience performed a lower rate of conversions than the other two
surgeons. Surgeon 1, who was not included in a group with previous
robotic experience, showed the highest incidence of conversions.
Although the results are not significant, in our opinion, these findings
may show that the inclusion in a group of surgeons with previous
robotic experience is important to overcome difficult situations, as
was seen in the first cases of laparoscopic colorectal cancer surgery
[17]. It is likely that the learning curves for success plotted for
surgeons 2 and 3 were superior to that of surgeon 1, as the chances of
conversion or intra-operative complications decreased with the help
of a group of surgeons with robotic experience.
None of the three series of cases in our study showed differences
in relation to the length of the surgical specimen or to the number of
lymph nodes. The distance between the tumor and the distal margin
was slightly longer in the group assigned to surgeon 1, although the
differences were not significant.
Another variable that it is hard to analyze is the variability in the
ability to perform surgery and mainly minimally invasive surgery.
This has not been analyzed in this study due to the high level of
subjectivity when making this analysis since it could include many
other factors (age, place of previous training, etc) [18].
Although previous experience with laparoscopic surgery does
not seem to be necessary to perform robotic rectal cancer surgery,
oncologic outcomes in the mean and short term must be further
analyzed in order to establish whether the learning curve affects
patient’s survival.
In addition, the use of robotic surgical simulators to provide
surgeons with preclinical practice must also affect the learning curve.
The design of charts to quantify the acquisition of basic skills and the
progress in the use of different instruments on the part of trainees
could lead to the necessity to establish some minimum requirements
that should be attained before the preclinical training is completed.
Conclusion
The lack of previous experience with laparoscopy for the treatment of rectal cancer does not seem to affect the short-term outcomes of robotic surgery. Yet, being part of a group of surgeons who have overcome the learning curve for this technique reduces operative times and conversion rates but not the incidence of re-interventions in the immediate postoperative period.
Contribution
All authors were responsible for conceptualizing and designing the study; collecting, analyzing and interpreting the data and drafting the manuscript. All authors gave their approval on the final version for publication.
References
- Park IJ, Choi GS, Lim KH, Kang BM, Jun SH. Multidimensional analysis of the learning curve for laparoscopic resection in rectal cancer. J Gastrointest Surg. 2009;13:275-81.
- Bege T, Lelong B, Esterni B, Turrini O, Guiramand J, Francon D, et al. The learning curve for the laparoscopic approach to conservative mesorectal excision for rectal cancer: lessons drawn from a single institution’s experience. Ann Surg. 2010;251:249-53.
- Kayano H, Okuda J, Tanaka K, Kondo K, Tanigawa N. Evaluation of the learning curve in laparoscopic low anterior resection for rectal cancer. Surg Endosc. 2011;25(9):2972-9.
- Son GM, Kim JG, Lee JC, Suh YJ, Cho HM, Lee YS, et al. Multidimensional analysis of the learning curve for laparoscopic rectal cancer surgery. J Laparoendosc Adv Surg Tech A. 2010;20:609-17.
- Ito M, Sugito M, Kobayashi A, Nishizawa Y, Tsunoda Y, Saito N. Influence of learning curve on short-term results after laparoscopic resection for rectal cancer. Surg Endosc. 2009;23(2):403-8.
- Luján, Cir Esp
- Jimenez-Rodriguez RM, Diaz-Pavon JM, de la Portilla de Juan F, Prendes-Sillero E, Dussort HC, Padillo J. Learning curve for robotic-assisted laparoscopic rectal cancer surgery. Int J Colorectal Dis. 2013;28(6):815-21.
- Park EJ, Kim CW, Cho MS, Baik SH, Kim DW, Min BS, et al. Is the learning curve of robotic low anterior resection shorter than laparoscopic low anterior resection for rectal cancer? A comparative analysis of clinicopathologic outcomes between robotic and laparoscopic surgeries. Medicine (Baltimore). 2014;93(25):e109.
- Melich G, Hong YK, Kim J, Hur H, Baik SH, Kim NK, et al. Simultaneous development of laparoscopy and robotic provides acceptable perioperative outcomes and shows robotics to have a faster learning curve and to be overall faster in rectal cancer surgery: analysis of novice MIS surgeon learning curves. Surg Endosc. 2015;29(3):558-68.
- Foo CC, Law WL. The learning curve of robotic-assisted low rectal resection of a novice rectal surgeon. World J Surg. 2016;40(2):456-62.
- Guend H, Widmar M, Patel S, Nash GM, Paty PB, Guillem JG, et al. Developing a robotic colorrectal cancer surgery program: understanding institutional and individual learning curves. Surg Endosc. 2017;31(7):2820-8.
- Sng KK, Hara M, Shin JW, Yoo BE, Yang KS, Kim SH. The multiphasic learning curve for robot-assisted rectal surgery. Surg Endosc. 2013;27(9):3297-307.
- Yamaguchi T, Kinugasa Y, Shiomi A, Sato S, Yamakawa Y, Kagawa H, et al. Learning curve for robotic-assisted surgery for rectal cancer: use of the cumulative sum metho. Surg Endosc. 2015;29(7):1679-85.
- Carlsen E, Schlichting E, Guldvog I, Johnson E, Heald RJ. Effect of the introduction of total mesorectal escisión for the treatment of rectal cancer. Br J Surg. 1998;85(4):526-9.
- Jimenez-Rodriguez RM, Rubio-Dorado-Manzanares M, Diaz-Pavon JM, Reyes-Díaz ML, Vazquez-Monchul JM, Garcia-Cabrera AM, et al. Learning curve in robotic rectal cancer surgery: current state of affairs. Int J Colorectal Dis. 2016;31(12):1807-15.
- Kim I, Kang J, Park Y, Kim N, Sohn S, Lee K. Is prior laparoscopic experience required for adaptation to robotic rectal surgery? Feasability for one-step transition from open to robotic surgery. Int J Colorectal Dis. 2014;29:693-9.
- Ogiso S, Yamaguchi T, Hata H, Kuroyanagi H, Sakai Y. Introduction of laparoscopic low anterior resection for rectal cancer early during residency: a single institutional study on short term outcomes. Surg Endosc. 2010;24(11):2822-9.
- Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J Chronic Dis. 1987;40(5):373-83.