Case Report
Pancreaticoduodenectomy Following Chemotherapy for Locally Advanced Adenocarcinoma of the Pancreatic Head: Case Report
Amine Chhaidar* and Ali Ben Ali
Department of General Surgery, Sahloul University Hospital, Tunisia
*Corresponding author: Amine Chhaidar, Department of Surgery, Sahloul University Hospital, Sousse 4051, Tunisia
Published: 01 Nov, 2018
Cite this article as: Chhaidar A, Ben Ali A.
Pancreaticoduodenectomy Following
Chemotherapy for Locally Advanced
Adenocarcinoma of the Pancreatic
Head: Case Report. Clin Surg. 2018;
3: 2186.
Abstract
Context: Pancreatic ductal adenocarcinoma is the fourth leading cause of cancer-related mortality.
It has an extremely poor prognosis and prolonged survival is achieved only by resection with
macroscopic tumor clearance. Locally advanced pancreatic adenocarcinoma is characterized by the
abutment or invasion of major vascular structures preventing surgeons from achieving optimal R0
resection.
Aims: We report a case of 52 years old patient with locally advanced, unresectable pancreatic ductal
adenocarcinoma. The patient was treated initially with neoadjuvant chemotherapy and subsequent
radical surgery. An objective response was obtained and the patient’s health improved.
Conclusion: Patients with locally advanced/unresectable tumors should be included in neoadjuvant
protocols and subsequently be reevaluated for resection, which is possible in a relevant number of
patients.
Keywords: Pancreatic adenocarcinoma; Neoadjuvant chemotherapy; SMV
Abbreviations
PDA: Pancreatic Ductal Adenocarcinoma; CT-scan: Computed Tomography Scan; LAPA: Locally Advanced Pancreatic Adenocarcinoma; SMA: Superior Mesenteric Artery; SMV: Superior Mesenteric Vein; NCCN: National Comprehensive Cancer Network
Introduction
Pancreatic Ductal Adenocarcinoma (PDA) remains as one of the most devastating human
cancers, with an overall 5-year survival of <5% [1]. It has an extremely poor prognosis and prolonged
survival is achieved only by resection with macroscopic tumor clearance [2].
Pancreatic tumors can be classified as resectable (stage I or II), locally advanced defined as
an unresectable tumor with no evidence of distant metastasis (stage III), or metastatic (stage IV).
However, with recent advances in pancreatic imaging and surgical techniques, a distinct subset
of tumors is emerging that blurs the distinction between resectable and locally advanced disease:
tumors of ‘borderline resectability [3].
There is a strong rationale for a neoadjuvant approach, since a relevant percentage of pancreatic
cancer patients present with non-metastatic but locally advanced disease and microscopic
incomplete resections are common [2]. Therefore, in patients with locally advanced pancreatic
adenocarcinoma, neoadjuvant treatment has been proposed as a way to decrease tumor burden and
downstage tumors [4].
We report our first locally advanced, unresectable PDA case treated initially with neoadjuvant
chemotherapy and subsequent radical surgery.
Case Presentation
A 52-year-old man presented to us with a history of epigastric pain and weight loss. This pain
was accompanied by a history of dyspepsia and progressively increasing jaundice with clay-colored
stools. There was no history of fever or pruritus.
On physical examination, there was evidence for icterus without any palpable neck
lymphadenopathy. The gallbladder was not palpable and no evidence
was found for ascites or peripheral edema.
Laboratory results demonstrated a cholestatic pattern; bilirubin
86.1 μmol/L (reference: range: 0 μmol/L to 17 μmol/L), alkaline
phosphatase 448 IU/L (reference: range: 40 IU/L to 120 IU/L), and
GGT 552 IU/L (reference: range: 0 IU/L to 150 IU/L). CA19-9 was
17.8 UI/ml (reference: range: 0 U/ml to 35 U/ml), ACE was 15.3 ug/L
(reference: range: 0 ug/L to 4.5 ug/L).
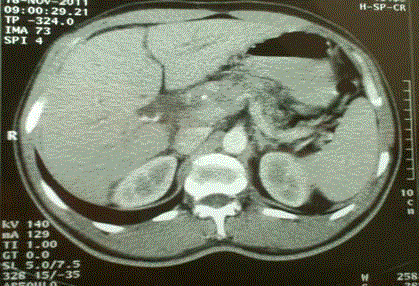
Abdominal and chest computed tomography scan confirmed the
presence of solid tumor at the pancreatic head. The axial diameter of
the tumor was 4 cm. Unfortunately, it was not possible to perform
a needle biopsy due to technical difficulties. However, images were
analyzed by a group of expert radiologists and evaluated with a
multidisciplinary team composed of oncologists, gastroenterologists
and surgeons, who agreed in the diagnosis of pancreatic cancer.
This tumor was classified as resectable on CT-scan in the absence
of contact with the main peripancreatic vessels to a ‘‘significant extent’’
(superior than 180_) and obvious signs for peritoneal carcinomatosis
or other distant metastases (Figure 1).
The patient underwent primary explorative laparotomy which
revealed a pancreatic node compatible with a pancreatic tumor
encasing the porto-splenic confluence (arrow) and attachment to
the HA (arrowhead) Therefore, the tumor was not resected. The
patient was treated by palliative surgical procedures: a biliodigestive
anastomosis with cholecystectomy and relief of jaundice was achieved.
Lymph node biopsy of the hepatic artery was made. Pathology
revealed a well differentiated pancreatic-type adenocarcinoma. The
patient then commenced chemotherapy with gemcitabine (1,000
gm/m2) in combination with cisplatin (50 mg/m2) every two weeks.
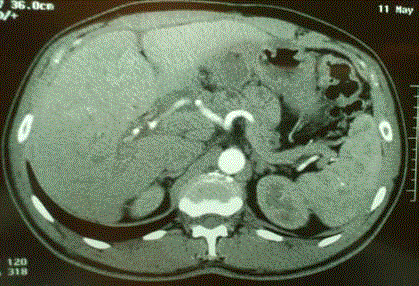
He completed three cycles of chemotherapy. During chemotherapy,
cross sectional imaging suggested partial tumor regression with no
evidence of metastatic disease (Figure 2). Six months after the initial
diagnosis the patient underwent surgery with curative intent in the
form of a Whipple procedure. He had an uneventful recovery period.
The whole pancreatic specimen was sent for pathological
examination. Macroscopically, there was no evidence of tumor.
Histopathological analysis of the specimen demonstrated
moderately differentiated pancreatic adenocarcinoma with extensive
signs of regression, with multiple microscopic infiltrative foci.
Surgical margins were free of disease.
The general condition of the patient remains stable and he is still
alive 19 months after diagnosis. Follow-up CT-scan did not identify
local or distant recurrences.
Figure 1
Figure 2
Discussion
Pancreatic ductal adenocarcinoma is the fourth leading cause
of cancer-related mortality [5], and is associated with an extremely
poor prognosis, reflected by a median survival of 5-8 months and a 5
year survival probability of less than 5% when all stages are combined
[5,6].
At the time of diagnosis, tumors are classified according to
radiological findings: the improvement of helical dual phase scanning
sensitivity and surgeons experience has advanced the classification
from resectable and unresectable to resectable, borderline and
unresectable [3,7]. Locally Advanced Pancreatic Adenocarcinoma
(LAPA), comprising both borderline and unresectable tumors,
is characterized by the abutment or invasion of major vascular
structures preventing surgeons from achieving optimal R0 resection.
The criteria used to define resectability, especially the definitions of
BR and UR disease, seem to differ among practitioners [8,9].
Locally advanced, unresectable PDA was defined as the presence
of PDA with an encasement (tumor involvement, >180° of the
circumference of the vessel) of one or more of the following vessels:
Superior Mesenteric Artery (SMA), celiac trunk, aorta, inferior vena
cava, portal vein, or Superior Mesenteric Vein (SMV); presence of
thrombosis of the portomesenteric venous system was considered as
unresectable disease as well.
Borderline resectable tumor was defined as PDA with an abutment
(tumor involvement, <180° of the circumference vessel) of the SMV
or portal vein and of the SMA or hepatic artery. Short-segment
encasement/occlusion of the SMV or portal vein amenable to vascular
resection and reconstruction was considered as borderline resectable
disease as well [10]. The designation of borderline resectable tumors
has emerged to describe a subpopulation of potentially resectable
tumors. The National Comprehensive Cancer Network (NCCN) has
defined this subgroup as locally advanced, resectable tumors. These
patients are at a high risk for margin positive resection with initial
surgery [11,12].
In fact, patients with borderline resectable tumors treated with
surgical resection alone can be expected to have a higher rate of local
and systemic disease recurrence and worse survival compared with
patients who presented with initially resectable disease [11].
Treatment of LAPA has been challenging. Operation without
preoperative therapy frequently results in explorative laparotomy
alone or pancreatic resection with microscopic or macroscopic
residual disease all of which do not seem to provide any survival
benefit [13].
Surgical resection with negative margins (R0) continues to be the
only opportunity for cure [14]. It is necessary to ensure long-term
survival. Thus, patients diagnosed with LAPA are likely to be spared
from surgery; rapid disease progression and poor clinical status
means patients undergo exclusively medical treatment with poor
survival rates [10,15,16].
However, patients diagnosed with non-metastatic LAPA
with a good clinical status are likely to undergo chemoradiation
[17]. Although it is impossible for patients with arterial adhesion/
invasion to undergo resection, venous involvement proved not to be
a contraindication to resection [18,19]. Thus, despite low resection
rates, several reports showed interesting results on overall survival
after pancreaticoduodenectomy for LAPA [20].
The role of neoadjuvant therapy in LAPA is a highly debated topic
[8,9]. This modality of treatment may allow tumor downsizing, reduce
the incidence of positive resection margins, delivery of treatment
to intact well-vascularized tissues, and higher rates of treatment
completion. Also, it facilitates selection for surgery of patients with
favorable tumor behavior. Patients who do not develop progressive
disease prior to rescue surgery or patients with significant downsize
response may have a better prognosis, and moreover, those with
poor tumor biology are selected out via disease progression, thereby
avoiding the morbidity of futile surgery [21].
A systematic review analyzed the role of neoadjuvant
chemoradiotherapy for the treatment of both resectable and initially
labeled as unresectable pancreatic cancer. This study demonstrated
that patients with unresectable pancreatic cancer who underwent
neoadjuvant chemoradiotherapy achieved comparable 1-year
survival as those with initially resectable disease; 40% of borderline or
unresectable cases were ultimately resected. Also, it was not associated
with a statistically significant increase in the rate of pancreatic fistula
or overall complications in the chemoradiation group [11,22].
Mura Assifi et al. analyzed a total of 14 phase II clinical trials
including 536 patients. Patients were divided into two groups:
patients with initially resectable tumors (Group A), and patients
with borderline/unresectable tumors (Group B). A total of 14 phase
II clinical trials including 536 patients were analyzed. Following
treatment, resectability was 65.8% (95% CI 55.4% to 75.6%) compared
with 31.6% in Group B (95% CI 14.0% to 52.5%). A significant partial
response was observed in patients with borderline/ unresectable
tumors; 31.8 (95% CI 24.2% to 39.8%) in Group B, and 9.5% (95% CI
2.9% to 19.4%) in Group A (p=0.003). Progressive disease was seen in
17.0% (95% CI 11.9% to 22.7%) of patients in Group A vs. 21.8% (95%
CI 10.1% to 36.5%) in Group B (p=0.006). Median survival in resected
patients was 23 months for Group A, and 22.3 months for Group B.
Nearly one-third of tumors initially deemed marginal for operative
intervention were ultimately able to be resected following treatment.
Until more effective targeted chemotherapeutics are developed, the
only groups of patients with pancreatic cancer that may benefit from
neoadjuvant treatment are those with locally advanced disease. From
this data we conclude that patients with borderline/unresectable
disease have increased partial response rates and a survival time
similar to patients with resectable disease after receiving neoadjuvant
treatment plus resection [4].
Gillen S et al. reviewed studies concerning the effects of
neoadjuvant therapy on tumor response, toxicity, resection, and
survival percentages in pancreatic cancer. The most important
findings was that in the group of resectable tumor patients, resection
and survival rates after neoadjuvant therapy were similar to the ones
observed in primarily resected tumor that are treated by adjuvant
therapy. Thus, in this group of patients, the current data did not
point to an obvious advantage of neoadjuvant therapy. In contrast, in
patients initially staged locally advanced/unresectable, approximately
one third of the patients could be resected following neoadjuvant
therapy with comparable survival rates as patients who were staged as
resectable before treatment [2].
As of now, the available data strongly suggest that patients
with locally advanced/unresectable tumors should be included in
neoadjuvant protocols and subsequently be reevaluated for resection,
which is possible in a relevant number of patients [2].
Conclusion
Surgical resection after downstaging of locally advanced and
borderline resectable pancreatic cancer should be offered to all
surgically fit patients without an increased postoperative mortality/
morbidity. Patients resected after neoadjuvant treatment have at least
the same survival rate of patients with resectable disease who undergo
primary resection.
Our initial experience in this case with locally advanced,
unresectable PDA treated with neoadjuvant chemoradiation is
encouraging. In this case, these results are consistent with the
published literature, suggesting a clear benefit in terms of curative
surgical resection. At this time our center is actively enrolling more
patients to be considered for this modality of treatment.
References
- Howlader N, Noone AM, Krapcho M, Neyman N, Aminou R, Altekruse SF, et al, (editors): SEER Cancer Statistics Review, 1975–2008. Bethesda, MD: National Cancer Institute; 2011.
- Gillen S, Schuster T, Meyer zum Bu¨schenfelde C, Friess H, Kleeff J. Preoperative/Neoadjuvant Therapy in Pancreatic Cancer: A Systematic Review and Meta-analysis of Response and Resection Percentages. PLoS Med. 2010;7(4):e1000267.
- Varadhachary GR, Tamm EP, Abbruzzese JL, Xiong HQ, Crane CH, Wang H, et al. Borderline resectable pancreatic cancer: definitions, management, and role of preoperative therapy. Ann Surg Oncol. 2006;13(8):1035-46.
- Assifi MM, Lu X, Eibl G, Reber HA, Li G, Hines OJ. Neoadjuvant therapy in pancreatic adenocarcinoma: a meta-analysis of phase II trials. Surgery. 2011;150(3):466-73.
- Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59(4):225-49.
- Sultana A, Tudur Smith C, Cunningham D, Starling N, Neoptolemos JP, Ghaneh P. Meta-analyses of chemotherapy for locally advanced and metastatic pancreatic cancer: results of secondary end points analyses. Br J Cancer. 2008;99(1):6-13.
- Talamonti M. Borderline resectable pancreatic cancer: a new classification for an old challenge. Ann Surg Oncol. 2006;13(8):1019-20.
- Turrini O, Viret F, Moureau-Zabotto L, Guiramand J, Moutardier V, Lelong B, et al. Neoadjuvant chemoradiation and pancreaticoduodenectomy for initially locally advanced head pancreatic adenocarcinoma. Eur J Surg Oncol. 2009;35(12):1306-11.
- Lee JL, Kim SC, Kim JH, Lee SS, Kim TW, Park DH, et al. Prospective efficacy and safety study of neoadjuvant gemcitabine with capecitabine combination chemotherapy for borderline-resectable or unresectable locally advanced pancreatic adenocarcinoma. Surgery. 2012;152(5):851-62.
- Barugola G, Partelli S, Crippa S, Capelli P, D'Onofrio M, Pederzoli P, et al. Outcomes after resection of locally advanced or borderline resectable pancreatic cancer after neoadjuvant therapy. Am J Surg. 2012;203(2):132-9.
- Galindo J, Gabrielli M, Guerra JF, Cassina JC, Garrido M, Jarufe N, et al. Neoadjuvant chemoradiation therapy for borderline pancreatic adenocarcinoma: report of two cases. World J Surg Oncol. 2013;11:37.
- Crane CH, Varadhachary G, Wolff RA, Pisters PW, Evans DB. Pre-operative chemoradiation for localized, radiographically resectable pancreatic cancer. Best Pract Res Clin Gastroenterol. 2006;20(2):365-82.
- Massucco P, Capussotti L, Magnino A, Sperti E, Gatti M, Muratore A, et al. Pancreatic resections after chemoradiotherapy for locally advanced ductal adenocarcinoma: analysis of perioperative outcome and survival. Ann Surg Oncol. 2006;13(9):1201-8.
- Heinrich S, Pestalozzi BC, Schafer M, Weber A, Bauerfeind P, Knuth A, et al. Prospective phase II trial of neoadjuvant chemotherapy with gemcitabine and cisplatin for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26(15):2526-31.
- Sultana A, Smith CT, Cunningham D, Starling N, Neoptolemos JP, Ghaneh P. Meta-analyses of chemotherapy for locally advanced and metastatic pancreatic cancer. J Clin Oncol. 2007;25(18):2607-15.
- Haddock MG, Swaminathan R, Foster NR, Hauge MD, Martenson JA, Camoriano JK, et al. Gemcitabine, cisplatin, and radiotherapy for patients with locally advanced pancreatic adenocarcinoma: Results of the North Central Cancer Treatment Group Phase II Study N9942. J Clin Oncol. 2007;25(18):2567-72.
- White RR, Reddy S, Tyler DS. The role of chemoradiation therapy in locally advanced pancreatic cancer. HPB (Oxford). 2005;7(2):109-13.
- Wang C, Wu H, Xiong J, Zhou F, Tao J, Liu T, et al. Pancreaticoduodenectomy with vascular resection for local advanced pancreatic head cancer: A single center retrospective study. J Gastrointest Surg. 2008;12(12):2183-90.
- Ishikawa O, Ohigashi H, Imaoka S, Furukawa H, Sasaki Y, Fujita M, et al. Preoperative indications for extended pancreatectomy for locally advanced pancreas cancer involving the portal vein. Ann Surg. 1992;215(3):231-6.
- Katz MH, Pisters PW, Evans DB, Sun CC, Lee JE, Fleming JB, et al. Borderline resectable pancreatic cancer: The importance of this emerging stage of disease. J Am Coll Surg. 2008;206(5):833-46.
- Van Tienhoven G, Gouma DJ, Richel DJ. Neoadjuvant chemoradiotherapy has a potential role in pancreatic carcinoma. Ther Adv Med Oncol. 2011;3(1):27-33.
- Laurence JM, Tran PD, Morarji K, Eslick GD, Lam VW, Sandroussi C. A systematic review and meta-analysis of survival and surgical outcomes following neoadjuvant chemoradiotherapy for pancreatic cancer. J Gastrointest Surg. 2011;15(11):2059-69.