Case Report
Aortic Dissection and Direct Factor Xa Inhibitors: Uncharted Waters
Igo B Ribeiro1, Sean Dickie2, Donna Nicholson2, Pietro Di Santo3 and Marc Ruel1*
1Department of Cardiac Surgery, University of Ottawa Heart Institute, Canada
2Department of Cardiac Anesthesia, University of Ottawa Heart Institute, Canada
3Department of Cardiology, University of Ottawa Heart Institute, Canada
*Corresponding author: Marc Ruel, Department of Cardiac Surgery, University of Ottawa Heart Institute, 40 Ruskin Street, Ottawa, Ontario K1Y 4W7, Canada
Published: 23 Oct, 2018
Cite this article as: Ribeiro IB, Dickie S, Nicholson D, Di
Santo P, Ruel M. Aortic Dissection and
Direct Factor Xa Inhibitors: Uncharted
Waters. Clin Surg. 2018; 3: 2177.
Abstract
Acute type A aortic dissection is a surgical emergency due to the high mortality in medically
treated patients mainly in the first 48 hrs of presentation. The use of factor Xa inhibitors in patients
presenting with acute aortic dissection is a challenging situation. Surgical delay has been proposed
to mitigate extensive surgical bleeding. We present a case of a patient on Rivaroxaban who could not
tolerate delayed surgery and developed massive bleeding despite the use of thromboelastography
and specific anti-Xa essay to guide optimal surgical time.
Keywords: Aortic dissection; DOAC; Factor Xa Inhibitors
Introduction
The prescription of Direct Oral Anticoagulants (DOACs) has increased annually, as guidelines
favor their use over warfarin in most clinical scenarios. DOAC's include dabigatran, a direct thrombin
inhibitor, and the direct activated factor X inhibitors (FXa-inhibitor): rivaroxaban, apixaban, and
edoxaban. Only dabigatran has a universally approved reversal agent. While a reversal agent for
FXa-inhibitors has been recently approved by the FDA for use [1], it is not widely available and is
not currently approved for clinical use in Canada or the EU. While waiting for these reversal agents,
cardiac surgeons must occasionally deal with the challenges of emergency operations in patients
who take FXa-inhibitors.
Aortic dissection is a surgical emergency where perioperative bleeding is a significant concern.
Delayed surgery for aortic dissection has been proposed as an alternative in selected patients on
DOAC's [2]. However, clinical acuity may not always allow it. Here we present the case of a patient
who underwent aortic dissection repair on Rivaroxaban. Delayed surgery was attempted, but the
patient’s clinical condition deteriorated and warranted operation. Although pre-operative levels
of speficic Rivaroxaban anti-Xa was reassuring, the patient developed extensive post-operative
bleeding.
Case Presentation
A 61-year old male presented to a primary hospital with sharp central chest and abdominal pain
that radiated to his back, associated with nausea and diaphoresis. His past medical history included
hypertension, chronic kidney dysfunction (creatinine 1.4 mg/dL), obstructive sleep apnea and
unprovoked recurrent VTE/PE on life-long 20 mg of rivaroxaban, which he took in the morning
of the day of the presentation. Computed Tomographic Angiography (CTA) confirmed a Stanford
type A aortic dissection, before transfer to our surgical center, the patient was started on a titrated
labetalol infusion and received 50 IU/Kg of Octaplex as part of a standardized DOAC reversal
protocol. At arrival, he had ongoing chest and abdominal pain as well as coffee ground emesis and
hematochezia. Blood pressure was 130/75 and HR 75. IV pantoprazole and nitroprusside infusions
were added.
A Rivaroxaban-specific anti-Xa essay showed the absence of clinically relevant anti-factor Xa
drug effect (<50 ug/ml). However, Rotational Thromboelastometry (ROTEM) demonstrated a
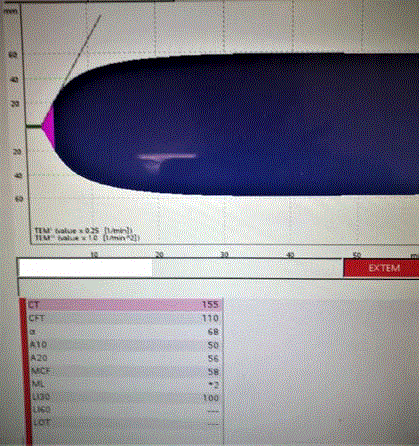
markedly prolonged EXTEM time of 155s (Normal range 43-82) (Figure 1), with normal fibrinogen
and platelet function, which suggested the possibility of residual DOAC activity. As his chest pain
resolved and there was no pericardial effusion or progression of dissection on repeated CTA, delayed
surgery was deemed reasonable. Close ICU monitoring took place.
The following day, patient had recurrent chest and abdominal
pain and increasing creatinine (3.2 mg/dL). Due to persistent
pain and possible signs of hypo perfusion, emergency surgery was
performed slightly after 24 hours of the last dose of rivaroxaban. A
second Rivaroxaban-specific anti-Xa essay was performed just before
the operation in case the first test reflected trough concentrations
and because of increased renal dysfunction [3]. The repeat level
was less than 50 ng/mL. A supra-coronary ascending aorta and
hemiarch replacement were performed under DHCA (22°C).
Bypass, aortic cross-clamp,and circulatory arrest were 181, 34 and
27 min respectively. The patientdeveloped severe coagulopathy
intraoperatively requiring massive blood transfusion- 10 FFPs, 4
adult platelets, 1000 IU of Octaplex® and 100 units of cryoprecipitate.
There was no intraoperative difficulties or surgical bleeding.
Eventually, bleeding was controlled by surgical means and blood
product transfusion and the sternum was closed. Chest tube output
was 550 ml in the first 24 hrs. The patient’s lactate quickly normalized
postoperatively and his clinical abdominal exam was reassuring.
No laparotomy was needed. He developed AKI and fluid overload,
requiring hemodialysis for three days. The patient was extubated on
the postoperative day 3 and was discharged from the ICU on day 7.
He spent a total of 13 days in hospital prior to discharge without the
need for ongoing dialysis.
Four-month follow-up CTA showed near complete remodeling
of the thoracoabdominal aorta. Although this patient is high risk for
recurrent VTE/PE, thrombosis and cardiac surgery have decided to
keep him off anticoagulation until aorta remodeling has been stable.
The patient has returned to his routine activities.
Figure 1
Comment
Type A Aortic dissection is a medical emergency with 50%
mortality in 30 days in medically treated patients [4]. Most of the
death happens during the first two days. Emergency surgery is the
treatment of choice. Operative bleeding is a significant concern
because of fragile tissues and the use of hypothermia for the surgical
repair. Patients on anticoagulation benefit from anticoagulation
reversal before the operation which is not currently available in most
centers, being recently approved for clinical use only in the USA.
Our patient did well despite massive transfusion. However, reviewed
published cases of acute aortic dissection on FXa-inhibitors are not
encouraging. To our knowledge, this is the first report to survive
during the full scope of medication. Three cases have been reported.
One patient died of postoperative complications [5]. Other died of
cardiac tamponade after an attempt of delayed surgery [6]. The third
case who survived had a delayed surgery [2]. However, the later
patient went to the operating room after 60 hrs delay with no likely
residual effect of the anticoagulant based on the calibrated anti-Xa
essay and thromboelastography. Interestingly, the 60 hrs delay is
close to the recommended time off FXa-inhibitors before elective
cardiac operations, which is to have the last dose three days before
the procedure [7]. Hamad et al. [2] have proposed delayed operation
for patients on DOAC's and the use of thromboelastometry and
drug-specific anti-Xa essay for monitoring drug effect [2]. However, a
thorough analysis of risk and benefits of delayed surgery is necessary
due to the deadly natural history of aortic dissection.
We attempted the delayed approach in the hope of full clearance
of the medication. Recurrent abdominal and chest pain and kidney
function deterioration led us to intervene earlier despite an abnormal
ROTEM EXTEM value. Our patient developed severe coagulopathy,
requiring massive transfusion which may have been reflective of
residual DOAC activity despite an anti-Xa value lower than 50 ng/
mL.
Intriguingly, our rivaroxaban-specific anti-Xa test was negative
on two occasions. Arguably, the level might have been between 30 ng/
mL and 50 ng/mL, which may have contributed to the coagulopathy
in addition to other factors associated with coagulopathy post
DHCA. The subcommittee on control of anticoagulation of the
International Society on Thrombosis and Haemostasis currently
recommend DOAC antidote administration if the level is greater than
50 ng/mL and consideration of antidote if greater than 30 ng/mL if
there is a high risk of bleed [3]. Hamad et al. [2] suggest delaying
the operation until the rivaroxaban-specific anti-Xa essay and
thromboelastography are normal. However, FXa-inhibitor-specific
anti-Xa essays have variability and reduced accuracy at upper and
lower limits of quantification, misleading interpretation. Thereforethe
judicious use of them is warranted until accurate and standardized
tests are available [8]. Thromboelastography may be helpful to clarify
coagulation abnormalities in those scenarios.
In the case presented, we tried to delay operation, but had
to change strategies when faced with the possibility of ongoing
dissection and regional hypo perfusion. Surgeons are left to navigate
the delicate balance of delaying surgery and massive bleeding.
Delayed operation is quite concerning in aortic dissection and may
be beneficial only in the seamlessly selected patient. The perioperative
teamshould be prepared for severe coagulopathy and the potential
consequences of massive transfusions. Until anti-Xa reversal agents
are approved for use in all jurisdictions, treating these patients with
require a multidisciplinary approach involving input from cardiac
anesthesia, cardiac surgery, and thrombosis-specialists to optimize
patient outcomes.
References
- FDA clears andexanet alfa for rapid NOAC reversal. 2018.
- Hamad R, Amr G, Demers P. Delayed surgery in patients with acute type A aortic dissection who are receiving novel oral anticoagulants. J Thorac Cardiovasc Surg. 2018:155:e1-4.
- Levy JH, Ageno W, Chan NC, Crowther M, Verhamme P, Weitz JI, et al. When and how to use antidotes for the reversal of direct oral anticoagulants: guidance from the SSC of the ISTH. J Thromb Haemost. 2016;14(3):623-7.
- Hagan PG, Nienaber CA, Isselbacher EM, Bruckman D, Karavite DJ, Russman PL, et al. The International Registry of Acute Aortic Dissection (IRAD): new insights into an old disease. JAMA. 2000;283(7):897-903.
- Dumps C, Halbeck E, Schimpf J, Forst H. [Perioperative coagulation management during ascending aorta replacement on apixaban]. Anaesthesist. 2016;65(8):595-600.
- Béné J, Auffray JL, Auffret M, Caron J, Gautier S. Aortic dissection during rivaroxaban therapy: a challenging care. Acta Anaesthesiol Scand. 2015;59(2):268.
- Sousa-Uva M, Milojevic M, Head SJ, Jeppsson A. The 2017 EACTS guidelines on perioperative medication in adult cardiac surgery and patient blood management. Eur J Cardiothorac Surg. 2018;53:1-2.
- Samuelson BT, Cuker A, Siegal DM, Crowther M, Garcia DA. Laboratory assessment of the anticoagulant activity of direct oral anticoagulants: a systematic review. Chest. 2017;151:127-38.