Research Article
Head & Neck Spindle Cell Melanoma
Cui Ye1,4, Feiluore Yibulaying3, Lei Feng2,4, Zhi-cheng Yang2,4* and Alimujiang Wushou2,4*
1Department of Orthodontics, Shanghai Stomatological Hospital, Fudan University, China
2Department of Oral and Maxillofacial Surgery, Shanghai Stomatological Hospital, Fudan University, China
3Department of Preventive Medicine, School of Public Health, Fudan University, China
4Oral Biomedical Engineering Laboratory, Shanghai Stomatological Hospital, Fudan University, China
*Corresponding author: Alimujiang Wushou, Department of
Oral and Maxillofacial Surgery & Oral
Biomedical Engineering Laboratory,
Shanghai Stomatological Hospital,
Fudan University, China
Zhi-cheng Yang, Department of Oral
and Maxillofacial Surgery & Oral
Biomedical Engineering Laboratory,
Shanghai Stomatological Hospital,
Fudan University, China
Published: 22 Oct, 2018
Cite this article as: Ye C, Yibulaying F, Feng L, Yang Z-C,
Wushou A. Head & Neck Spindle Cell
Melanoma. Clin Surg. 2018; 3: 2167.
Abstract
Purpose: Head Neck Spindle Cell Melanoma (HNSCM) is very rare morphologic subtype of
melanoma and unexplored comprehensively. The aim of this study was to investigate HNSCM’s
incidence, general demographics, basic clinico-pathologic features, and treatment outcome and
disease specific prognostic factors.
Material and Methods: HNSCM cases were identified in Surveillance, Epidemiology, and End
Results database (1973-2018).
Results: A total of 2500 HNSCM cases with a median age of 66 years were found in the database.
The Male-to-female ratio is 2:1. Statistically significant Overall Survival (OS) and Disease Specific
Survival (DSS) differences were found were depending on age, gender, race, age, tumor location,
SEER historic stage, T stage, N stage, M stage and pathological differentiation (p<0.05). In the
multivariate Cox regression analysis, age >66 years [Hazard ratio (HR) (95%, Confidence Interval,
CI) =2.785 (2.224-3.488), p=0.000, for OS; HR (95% CI) =2.140 (1.535-2.983), p=0.000, for DSS,
age ≤ 66 years as reference], unmarried status [HR (95% CI) =1.254 (1.031-1.525), p=0.023, for OS,
married as reference], T3+T4 stage [HR (95% CI) =1.330 (1.080-1.639), p=0.007, for OS, HR (95%
CI) =1.577 (1.106-2.247), p=0.012, for DSS, TX+T1+T2, as reference] and M1 stage [HR (95% CI)
=2.658 (1.385-5.101), p=0.003, for OS, HR (95% CI) =6.894 (2.122-22.39), p=0.003, for DSS, M0
as reference] were associated with worse DSS and OS. Besides, the non-skin tumor [HR (95% CI)
=0.511 (0.366-0.714), p=0.000, for OS, HR (95% CI) =0.603 (0.366-0.994), for DSS, p=0.047 skin
tumor as reference] was associated with favorable DSS.
Conclusion: HNSCM mostly occurred in white people at 60~80 years old with predominance in
males. The patient’s age, tumor location, T stage, M stage and marital status were independent
prognostic factors for DSS and OS.
Keywords: Spindle cell melanoma; Head neck; SEER analysis
Introduction
Spindle cell melanoma (SCM) is a morphologic variant of melanoma [1]. SCM is a diagnostic
challenge for pathologists, because there are many lesions which can be spindle cell rich and SCM lack
of classic conventional melanoma characteristic [2]. Histologically, SCM is often confused with other
(e.g. mesenchymal or neural) tumors. SCM often leads misdiagnosis and immunohistochemistry is
helpful tool in distinguishing SCM from other sarcoma and carcinoma. Specific immunophenotypes
is decisive in diagnosis to differentiate SCM from desmoplastic melanoma [2,3].
Melanoma is a common malignancy; however, SCM is very rare subtype [4]. SCM is frequently
amelanotic, can occur anywhere on the body [5,6]. The clinical presentation of SCM is non-specific,
and the early diagnosis is often delayed. Plus, it has an aggressive biological behavior and presents
typically with widespread metastatic disease and ended up worse treatment outcome [7]. Since the
majority of cases were found at the advanced stage of tumor, the prognosis is poor despite the
availability of common therapeutic option like surgical therapy, immunotherapy, chemotherapy or
radiotherapy [8].
To our knowledge, except several sporadic case reports and small incomplete retrospective case
series, most of the previous studies have been published on the SCM diagnosis and differential
diagnostic viewpoint [9-12]. Due to the rarity of the SCM, especially the head neck region, the
limited study population of previous studies, there are no studies available, which have focused
on the incidence, clinicopathologic features, treatment outcome
and disease independent prognostic factors. Thus, in order to give
audiences a concise spectrum of Head Neck SCM (HNSCM), a
retrospective clinical case series was performed by using data from
registered in the Surveillance, Epidemiology, and End Results (SEER).
Figure 1
Materials and Methods
Data source
The SEER program is comprised of 18 population-based cancer
registries around the United States. Records from SEER registries are
publicly available and are managed by the National Cancer Institute
and it currently represents approximately 30% of the United States
overall population [13]. The data extraction and statistical analysis
was performed as described previously [14,15]. Briefly, International
Classification of Diseases for Oncology codes 8772/3 were used for
data extraction from SEER database with official software SEER*Stat,
version 8.3.4.
Statistical analysis
Statistical analysis was carried out by using software of the
Statistical Package for Social Sciences, Version 23.0, for Windows
(SPSS, Chicago, IL). The chi square test or Fisher exact test was
used for categorical variables comparison. The survival curves were
developed by using the Kaplan-Meier method, and the log-rank
test was performed to evaluate the survival difference. Adjusted
Hazard Ratios (HRs) along with 95% Confidence Intervals (CIs)
were calculated by using the Cox proportional hazards regression
model. When the p value was <0.05, the difference was regarded as
statistically significant. All statistical tests were two tailed.
Results
General demographic and clinicopathologic characteristics of
study cohort: A total of 2500 patients, consisted of 1704 males and
796 females with a median age of 66 years were found in the SEER
database from 1973 to 2017. A peak incidence occurred during sixth
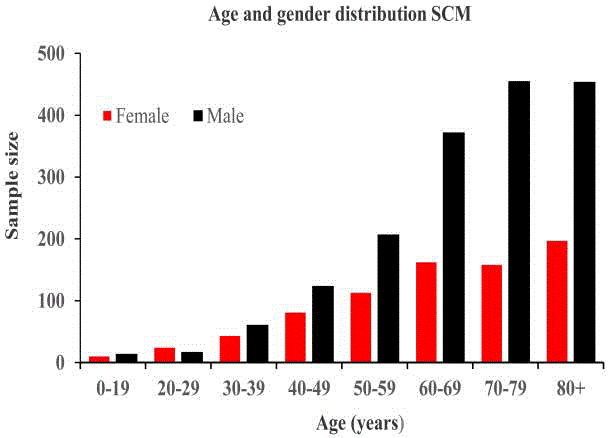
to eighth decade of life. The age and gender distribution is shown in
(Figure 1). White people account for 97.72% (2443/2500) of study
population. The overall median follow-up period was 75.2 months
(range, 0-482 months). Nearly 70% of HNSCM originated from HN
skin. The basic demographic and clinicopathologic characteristics of
the whole patients are summarized in (Table 1).
Overall survival (OS) and cox proportional hazards regression
analysis: Kaplan-Meier analysis was applied for time-to-event
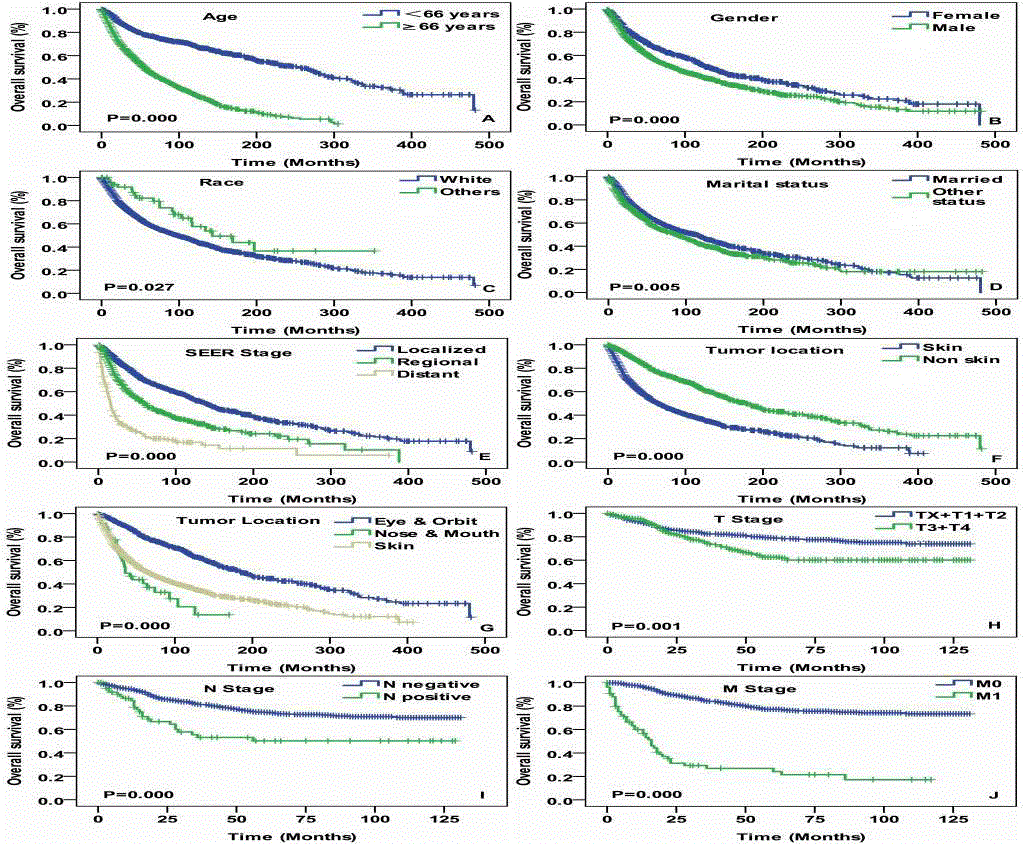
analysis. Statistically significant OS differences were found were
depending on race (p=0.027), age (p=0.000), SEER stage (p=0.000),
tumor location (p=0.000), marital status (p=0.005), gender (p=0.000),
T stage (p=0.000), N stage (p=0.000), M stage (p=0.000). These results
are presented in (Figure 2). The Cox proportional hazards regression
model was applied for univariate and multivariate survival analysis.
Age, race, gender, marital status, SEER stage, tumor location,
pathological grade, T stage, M stage, N stage were associated with OS
in the univariate Cox regression analysis. Details are shown in Table
2. In the multivariate Cox regression analysis, age >66 years [Hazard
ratio (HR) (95% confidence interval, CI) =2.785 (2.224-3.448),
p=0.000, age ≤ 66 years as reference], parameter with unmarried
status [HR (95% CI) =1.254 (1.031-1.525), p=0.023, married as
reference], T3+T4 stage [HR (95% CI) =1.330 (1.080-1.639), p=0.007,
TX+T1+T2 as reference] and M1 stage [HR (95% CI) =2.658 (1.385-
5.101), p=0.003, M0 as reference] were independently associated with
worse OS. These details are shown in (Table 3).
The demographic and clinicopathologic features with Disease-
Specific Survival (DSS) status: For Disease Specific Survival (DSS)
analysis, there were 1393 patients (489 females and 904 males)
obtainable in the total study cohort. The median age was 66.2 years
and 36.8% of patients with age ≤ 66.2 years. The median follow-up
time for DSS was 78.6 months (range, 0-482 months). The early AJCC
stage (stage I+II) cases were 555 and advanced stage (stage III+IV)
patients were 180. Nearly 80% of patients (1110/1393) were treated by
surgery. Summary statistics of DSS status are presented in the (Table
1).
DSS and cox proportional hazards regression analysis: In the
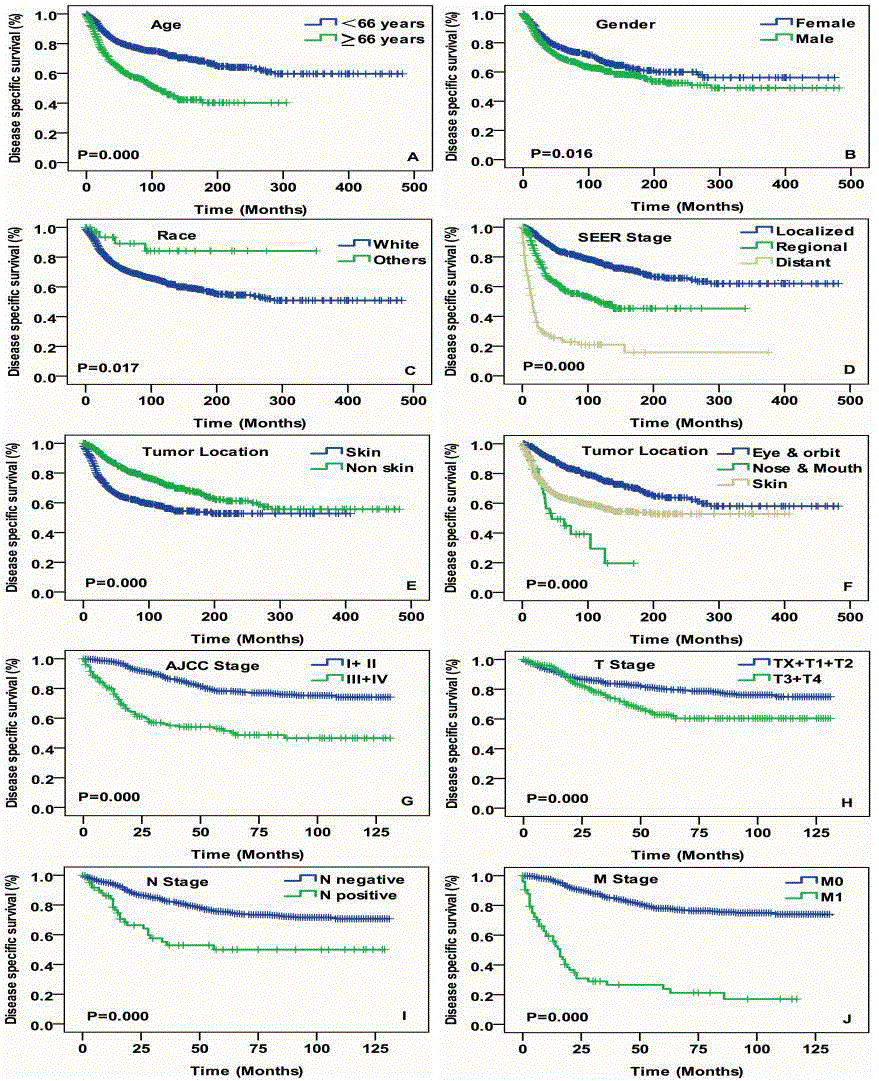
survival analysis for DSS, significant survival differences were also
identified depending on race (p=0.004), age (p=0.000), SEER stage
(p=0.000), tumor location (p=0.000), pathological differentiation
(p=0.003), T stage (p=0.000), N stage (p=0.000), M stage (p=0.000)
(Figure 3). In the univariate Cox regression analysis, gender, age, SEER
stage, race, tumor location, T stage, N stage, M stage was associated
with DSS (Table 2). In the multivariate Cox regression analysis, age
>66 years [HR (95% CI) =2.140 (1.535-2.983), p=0.000, age ≤ 66 years
as reference], SEER historic stage of regional tumor [HR (95% CI)
=1.699 (1.076-2.684), p=0.023, localized tumor as reference], T3+T4
stage [HR (95% CI) =1.577 (1.106-2.247), p=0.012, TX+T1+T2
as reference] and M1 stage [HR (95% CI) =6.894 (2.122-22.391),
p=0.003, M0 as reference] were adversely associated with DSS. Nonskin
tumor [HR (95% CI) =0.603 (0.366-0.994), p=0.047, skin tumor
as reference] was favorable associated with better DSS. Details of the
univariate and multivariate Cox regression analysis are presented in
the (Table 2, 3).
Figure 2
Figure 2
Overall survival curves of cases with head and neck spindle
cell melanoma compared according to (A) age, (B) gender, (C) race, (D)
marital status, (E) SEER stage, (F) and (G) tumor location, (H) T stage, (I)
N stage and (J) M stage. Log-rank test was utilized to compare curves, and
significance is presented on each panel.
Table 1
Table 2
Discussion
The incidence of HN melanoma is 22% to 34% of all melanoma
[16-18]. As a morphologic variant of melanoma, the incidence
of HNSCM is rare and no detailed data are available [4]. To our
knowledge, this is the first study about the incidence and survival
analysis of HNSCM. Current results demonstrated nearly 70% of
HNSCM occurred in the head neck skin with peak incidence in the
sixth to eighth decade of life and predilection of males.
Regarding to general parameters in the survival analysis,
remarkable survival differences were identified in age, gender
and race regarding OS and DSS (Figures 2A-2C, and 3A-3C). This
in accordance with previous studies on melanoma [19,20]. The
explanations behind prognosis may be men have less hair on the scalp
and more UV exposure outdoors than women, and white people are
more sensitive compared with colored people. Besides, significant
differences were shown in marital status in OS (Figure 2D), which
may indicate that the spouse’s care and support are good for physical
and mental recovery.
Because of unspecific hidden clinical presentation of HNSCM,
early detection and diagnosis are always delayed. Thus, overall
prognosis of HNSCM is poor [7,8]. Supporting these, advanced stage
of HNSCM had the worse outcome than early stage tumor in our
analysis (Figure 3D). In SEER stage survival analysis; there were also
remarkable differences on OS and DSS (Figure 2E, 2G, 3D) as AJCC
stage. Tumor locations had a great effect on the OS and DSS. In our
study, we found that HNSCM derived from skin had a significantly
worse outcome than non-skin tumor. This may be due to the greater
metastases tendency [8]. Tumors located in the nose & mouth and
skin obviously had poor OS and DSS comparing in the eye and orbit.
The results are in accordance with previous studies [21,22]. This is
likely because superficial and bare tumor sites like eyes & orbit had an
earlier diagnosis and treatment than deep-seated unexposed tumor
sites.
HNSCM are frequently found at the advanced stage and it is a
challenge to the head and neck oncologist to correctly evaluate this
aggressive tumor and choose the best suitable treatment protocol. In
the current investigation, surgery is only treatment modality we can
obtain the SEER database. However, surgery types are unavailable,
even types of neck dissections were not recorded too. Owing to the
complex anatomic structure of the head and neck region, radical
surgery with clear margins for HNSCM is difficult, especially tumor
at the advanced stage. Because, extensive excision in the head neck
region requires wide-range of surgical reconstruction. No controlled
surgical trials or multiple treatments have been carried out for
HNSCM [23]. Such a circumstance, how to best plan a surgery based
combined treatment protocol for different stage of HNSCM with
currently available therapies such as neoadjuvant chemotherapy,
immunotherapy or radiotherapy is difficult and challenging question
to answer. Therefore, multi-cancer randomized clinical trials are
urgently needed.
As a retrospective analysis, a few limitations of this study should
be noticed. Firstly, not all of the cases have complete information,
thus, the strength of the study is impaired. Secondly, the limited
number of patients did not enable us to perform more specific
subgroup comparison analysis for different tumor location or other
important clinicopathological parameters. Thirdly, besides surgical
treatment, other treatment modalities were lacking. Those missing
treatment modalities should not be neglected, because they may have
a huge impact for patient’s survival. Lastly, the follow-up time is
uneven and not long enough.
Table 3
Figure 3
Figure 3
Disease specific survival curves of cases with head and neck
spindle cell melanoma compared according to (A) age, (B) gender, (C) race,
(D) SEER stage, (E) and (F) tumor location, (G) AJCC stage, (H) T stage, (I)
N stage and (J) M stage. Log-rank test was utilized to compare curves, and
significance is presented on each panel.
Conclusion
Within the limitations of this study, we report a first largest case series about HNSCM’s demographic trends, clinico-pathologic features, outcome and disease specific independent prognostic factors. Our results demonstrate that HNSCM mostly occurred in white people with predominance in the male and reached a peak incidence during the sixth to eighth decade of life. The patient’s age, tumor location, T stage, M stage, marital status and SEER stages were independent prognostic factors for DSS and OS.
Funding
This study was supported by The Scientific Research Foundation of Shanghai Stomatological Hospital, Fudan University (SSDCZ-2016-01) and Shanghai Natural Science Foundation (17ZR1424900).
References
- Longacre TA, Egbert BM, Rouse RV. Desmoplastic and spindle-cell malignant melanoma: an immunohistochemical study. Am J Surg Pathol. 1996;20:1489-500.
- Weissinger S, Keil P, Silvers D, Klaus BM, Möller P, Horst BA, et al. A diagnostic algorithm to distinguish desmoplastic from spindle cell melanoma. Mod Pathol. 2014;27(4):524-34.
- Livestro DP, Muzikansky A, Kaine EM, Flotte TJ, Sober AJ, Mihm MC Jr. Biology of desmoplastic melanoma: a case-control comparison with other melanomas. J Clin Oncol. 2005;23(27):6739-46.
- Sundersingh S, Majhi U, Narayanaswamy K, Balasubramanian S. Primary spindle cell melanoma of the urinary bladder. Indian J Pathol Microbiol. 2011;54(2):422-4.
- Sheff J, Pane T. Spindle cell melanoma arising from decades-old burn scar. Plast Reconstr Surg. 2009;124(5).
- Dainichi T, Kobayashi C, Fujita S, Shiramizu K, Ishiko T, Kiryu H, et al. Interdigital amelanotic spindle-cell melanoma mimicking an inflammatory process due to dermatophytosis. J Dermatol. 2007;34(10):716-9.
- Piao Y, Guo M, Gong Y. Diagnostic challenges of metastatic spindle cell melanoma on fine‐needle aspiration specimens. Cancer. 2008;114(2):94-101.
- Conley J, Lattes R, Orr W. Desmoplastic malignant melanoma (a rare variant of spindle cell melanoma). Cancer. 1971;28(4):914-36.
- Prieto V, Woodruff J. Expression of HMB45 antigen in spindle cell melanoma. J Cutan Pathol. 1997;24(9):580-1.
- Prieto V, Woodruff J. Expression of basement membrane antigens in spindle cell melanoma. J Cutan Pathol. 1998;25(6):297-300.
- Kobayashi G, Cobb C. A case of amelanotic spindle-cell melanoma presenting as metastases to breast and axillary lymph node: diagnosis by FNA cytology. Diagn Cytopathol. 2000;22(4):246-9.
- Mitamura Y, Ito T, Nakano-Nakamura M, Uchi H, Furue M. S100A6 and c-Kit-Positive Spindle Cell Melanoma of the Dorsal Foot. Case Rep Dermatol. 2014;6(2):140-4.
- Cronin KA, Ries LA, Edwards BK. The Surveillance, Epidemiology, and End Results (SEER) Program of the National Cancer Institute. Cancer. 2014;120 Suppl 23:3755.
- Feng L, Cai D, Muhetaer A, Yin-Long Y, Fei R, Mumingjiang Y, et al. Spindle cell carcinoma: the general demographics, basic clinico-pathologic characteristics, treatment, outcome and prognostic factors. Oncotarget. 2017;8(26):43228-36.
- Wushou A, Jiang YZ, Liu YR, Shao ZM. The demographic features, clinicopathologic characteristics, treatment outcome and disease-specific prognostic factors of solitary fibrous tumor: a population-based analysis. Oncotarget. 2015;6(39):41875-83.
- Koto M, Demizu Y, Saitoh JI, Suefuji H, Tsuji H, Okimoto T, et al. Multicenter Study of Carbon-Ion Radiation Therapy for Mucosal Melanoma of the Head and Neck: Subanalysis of the Japan Carbon-Ion Radiation Oncology Study Group (J-CROS) Study (1402 HN). Int J Radiat Oncol Biol Phys. 2017;97(5):1054-60.
- Kılıç S, Unsal AA, Chung SY, Samarrai R, Kılıç SS, Baredes S, et al. Geographic region: Does it matter in cutaneous melanoma of the head and neck? Laryngoscope. 2017;127(12):2763-9.
- Baykal C, Atci T, Polat Ekinci A, Buyukbabani N. An update on cutaneous melanoma in Turkey: evaluation of 19‐year data in a single tertiary centre and review of the literature. J Eur Acad Dermatol Venereol. 2017;31:236-40.
- Helsing P, Robsahm TE, Vos L, Rizvi SMH, Akslen LA, Veierød MB. Cutaneous head and neck melanoma (CHNM): A population-based study of the prognostic impact of tumor location. J Am Acad Dermatol. 2016;75(5):975-2.
- Sample A, He YY. Mechanisms and prevention of UV‐induced melanoma. Photodermatol Photoimmunol Photomed. 2018;34(1):13-24.
- Khan F, Strohl A, Allen PD, Doerr TD. Desmoplastic Melanoma of the Head and Neck: Incidence and Survival, 1992-2013. Otolaryngol Head Neck Surg. 2017;157(4):648-56.
- Paolino G, Cardone M, Didona D, Moliterni E, Losco L, Corsetti P, et al. Prognostic factors in head and neck melanoma according to facial aesthetic units. G Ital Dermatol Venereol. 2017.
- Gladfelter P, Darwish NH, Mousa SA. Current status and future direction in the management of malignant melanoma. Melanoma Res. 2017;27:403-10.