Research Article
Appendectomy versus Conservative Antibiotics Treatment for Non-Complicated Acute Appendicitis: A Meta-Analysis of Randomized Controlled Trials
Hyun Kang1, Yoo Shin Choi2, Suk Won Seo2, Geun Joo Choi1 and Seung Eun Lee2*
1Department of Anesthesiology, Chung-Ang University College of Medicine, South Korea
2Department of Surgery, Chung-Ang University College of Medicine, South Korea
*Corresponding author: Seung Eun Lee, Department of Anesthesiology, Chung-Ang University College of Medicine, 102 Heukseok-ro, Dongjak-gu, Seoul 06973, South Korea
Published: 16 Oct, 2018
Cite this article as: Kang H, Choi YS, Seo SW, Choi
GJ, Lee SE. Appendectomy versus
Conservative Antibiotics Treatment for
Non-Complicated Acute Appendicitis:
A Meta-Analysis of Randomized
Controlled Trials. Clin Surg. 2018; 3:
2164.
Abstract
Purpose: In this meta-analysis, we compared the safety and efficacy of antibiotic treatment with
appendectomy for the primary treatment of uncomplicated acute appendicitis.
Methods: A comprehensive search was conducted using MEDLINE, EMBASE, and the Cochrane
Central Register of Controlled Trials to identify randomized controlled trials that compared
antibiotic treatment with appendectomy in uncomplicated acute appendicitis. The primary outcome
measure was the initial treatment failure rate. Secondary outcomes included the overall treatment
failure rate, length of primary hospital stay, time for sick leave, and cost.
Results: In total, 1,480 patients from 6 randomized controlled trials (751 undergoing antibiotic
treatment, 729 undergoing appendectomy) were included in the final analysis. Overall treatment
efficacy in the antibiotic group was 0.77 (95% CI, 0.68 to 0.84; Pchi
2<0.001; I2=81.51%) versus 0.91
(95% CI, 0.81 to 0.96; Pchi
2<0.001; I2=80.23%) in the appendectomy group. There were no differences
with regard to overall treatment efficacy (Risk ratio (RR), 0.88; 95% CI, 0.74 to 1.04; Pchi
2<0.001;
I2=90.98%; NNT=6), complication rate (Risk ratio(RR), 1.56; 95% CI, 0.48 to 5.05; Pchi
2=0.008;
I2=71.21%; NNT=186), hospital stay (mean difference(MD), -0.05; 95% CI, -0.65 to 0.55; Pchi
2<0.001;
I2=98.24%), time for sick leave (MD, -4.36; 95% CI, -12.00 to 3.28; Pchi
2<0.001; I2=99.45%) and cost
(MD, -12230.46; 95% CI, -17086.77 to -7374.15; Pchi
2=0.321; I2=0.00%).
Conclusion: Antibiotic treatment is effective and safe as primary treatment for patients with
uncomplicated acute appendicitis. Therefore, antibiotic treatment could be considered as an
alternative option in the treatment of patients with acute uncomplicated appendicitis.
Keywords: Acute appendicitis; Appendectomy; Antibiotic; Meta-analysis
Introduction
Acute appendicitis is one of the most common surgical emergencies and early appendectomy has been regarded as the gold standard of therapy since Mc Burney recommended immediate appendectomy in every case of presumed acute appendicitis in order to avoid potential fatal outcome in the pre-antibiotic era [1]. Even though appendectomy has been the mainstay for appendicitis, Coldrey reported treating 471 patients with antibiotics in 1956, relatively soon after antibiotics were available [2]. However, although an initial nonsurgical approach for complicated appendicitis has been relatively widely accepted, mainly on the basis of tradition rather than evidence, the role of antibiotic treatment in acute uncomplicated appendicitis may have been over looked. Noncomplicated acute appendicitis is defined as absence of any of criteria of complicated appendicitis with peritonitis: extra luminal gas, periappendiceal fluid, or disseminated intraperitoneal fluid. The rationale of appendectomy in acute appendicitis has its origins in the assumption that nonperforated appendicitis progresses to perforation, with intra luminal appendiceal obstruction as the main cause [3,4]. However, since the beginning of the 20th century, many researchers entertained an infectious cause for appendicitis [5-8]. If infection were the prevailing etiology of acute appendicitis, it would be logical to treat it with antibiotics rather than appendectomy. Several randomized controlled trials have been performed and these have also been subjected to meta-analysis [9-14]. Each of these meta-analyses had several limitations such as including withdrawn trial since publication, nonrandomized trial, or omitting some published randomized controlled trials, and without definite evidence of superiority, Appendectomy remains the standard approach for treating appendectomy [15-19]. The meta-analysis presented here provides a valid and up-todate summary of the relevant literature, including a recently published randomized controlled trial and excluding retracted publication, and publication that is unclear if the patients were randomized. The aim of this meta-analysis for randomized controlled trials was to compare the safety and efficacy of antibiotic treatment with appendectomy for the primary treatment of uncomplicated acute appendicitis.
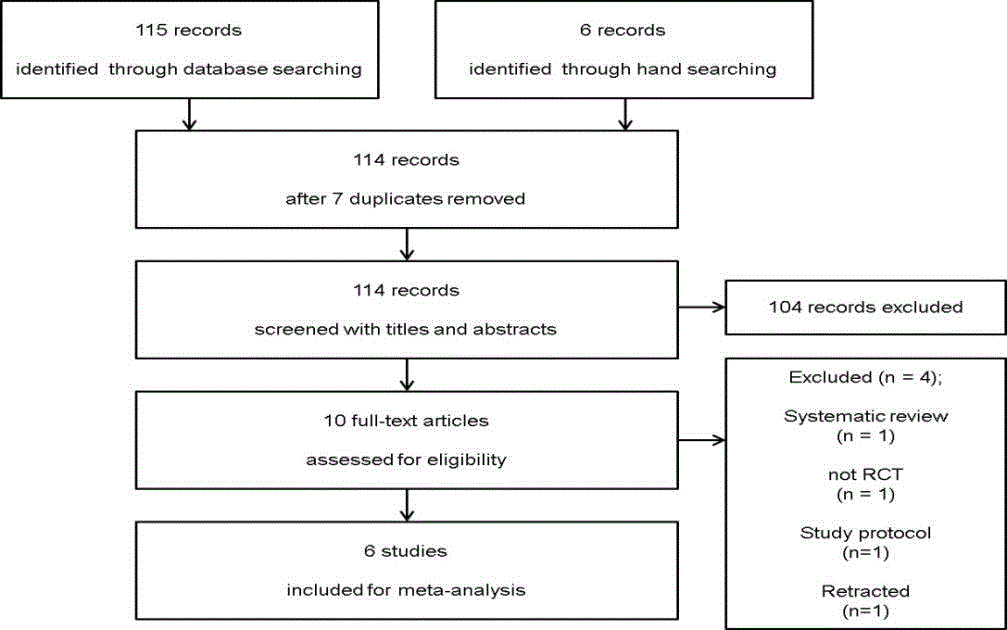
Figure 1
Figure 1
Flow diagram showing the number of abstracts and articles identified and evaluated during the review process.
Materials and Methods
The present systematic review and meta-analysis was registered
in PROSPERO (CRD42016038179) and was conducted following the
guideline with the Preferred Reporting Items for Systematic Reviews
and Meta-Analysis (PRISMA) [20].
Literature search
Two authors (KH and LSE) independently carried out database
searches in April 2016 using OVID-MEDLINE, EMBASE, the
Cochrane Central Register of Controlled Trials (CENTRAL), and
Google Scholar. There were no language limitations in our study. The
reference lists of the identified literatures and eligible articles were also
searched manually. The following Mesh search headings were used:
‘‘complicated appendicitis,’’ ‘‘perforated appendicitis,’’ ‘‘appendiceal
abscess,’’ ‘‘appendiceal mass,’’ ‘‘conservative treatment,’’ ‘‘acute
appendectomy,’’ ‘‘interval appendectomy,’’ ‘‘comparative study,’’
and ‘‘treatment outcome.’’ The above terms and their combinations
were also searched as text words. The search strategy, which included
a combination of free text, Medical Subject Headings and EMTREE
terms, is described in the Appendix.
Study selection
The study’s inclusion and exclusion criteria were determined
before systematic search. Randomized controlled trials (RCTs) were
included that compared surgery (either laparotomy or laparoscopy)
with antibiotic therapies alone for the treatment of patients with noncomplicated
acute appendicitis. Review articles, case reports, caseseries,
letters to the editor, commentaries, proceedings, laboratory
science studies, and any other non-relevant studies were excluded.
Two authors (CGJ and SSW) independently scanned the titles and
abstracts of the reports identified via the search strategies described
above. If a report was determined eligible from the title or abstract,
the full paper was retrieved. Potentially relevant studies chosen by at
least one author were retrieved, and full-text versions were evaluated.
Two authors (CGJ and SSW) discussed their opinion to arrive at a
consensus as to whether a study should be included or excluded.
Disagreement over inclusion or exclusion was settled in discussion
with a third investigator (LSE).
Outcome measure
We recorded clinical outcomes according to the intention to
treat analysis where available. The primary outcome measure of this
meta-analysis was initial treatment efficacy. Treatment efficacy was
defined as a patient who was treated successfully with antibiotics
without treatment failure. For antibiotics treatment, success was
defined as definite improvement without need for surgery and
subsequent hospital discharge without an operation. Therefore, initial
treatment failure in the antibiotic group was defined as performance
of an appendectomy within 48 hrs after initial hospitalization with
antibiotics and on the contrary, initial treatment efficacy was defined
as completeness of antibiotics treatment with no appendectomy
within 48 hrs after initial hospitalization. The secondary outcome
measures were overall treatment efficacy, complicated appendicitis,
and complication, length of primary hospital stay, sick leave, and
costs. Overall treatment failure and efficacy were determined during
follow-up period of up to one year. Overall treatment efficacy was
defined as no appendectomy during follow-up period of up to one
year after antibiotics treatment. For surgical treatment, treatment
success was defined as patients who were successfully treated with
appendectomy. Complicated appendicitis was defined as perforated
or gangrenous appendicitis. Complications included any antibioticrelated
or surgery-related morbidity such as abscess formation, ileus,
wound infection, and so on.
Data extraction
All interrelated data from the included studies were independently
extracted and entered into standardized forms by two authors (CYS,
KH), and then cross-checked. Any discrepancy was resolved through
discussion. If an agreement could not be reached, the dispute was
resolved with the aid of a third investigator (LSE). The standardized
form included the following items: (1) title, (2) name of first author,
(3) name of journal, (4) year of publication, (5) study design, (6)
registration of clinical trial, (7) competing interest, (8) country, (9)
risk of bias, (10) inclusion criteria, (11) exclusion criteria, (12) sex,
(13) age, (14) number of patients, (15) diagnosis, (16) regimen of
antibiotic used, (17) type of surgery performed, (18) primary and
secondary outcome, (19) complication of intervention, (20) hospital
stay, (21) time for sick leave, and (22) total cost. The data were
initially extracted from tables or text. In cases involving missing or
incomplete data, an attempt was made to contact the study authors to
obtain the relevant information.
Risk of bias assessment
The quality of studies was independently assessed by two authors
(CGJ and SSW) using the tool of “risk of bias” according to the
software Review Manager (Version 5.3, The Cochrane Collaboration,
Oxford, UK). Quality was evaluated using the following potential
sources of bias: sequence generation, allocation concealment,
blinding of participants or outcome assessor, incomplete data, and
selective reporting. The methodology for each study was graded as
“high”, “low”, or “unclear”, which reflected a high risk of bias, low
risk of bias, or uncertain bias, respectively.
Statistical analysis
We conducted this meta-analysis by Review Manager (Version
5.3, The Cochrane Collaboration, Oxford, UK) and Comprehensive
Meta-Analysis software (Version 2.0; Biostat, Englewood, NJ, USA).
Two authors (KH and LSE) independently input all data into the
software. The pooled Risk Ratio (RR) or mean difference (MD)
and their 95% Confidence Intervals (CIs) were calculated for each
outcome. We used the chi-squared test for homogeneity and the I2
test for heterogeneity. A level of 10% significance (P<0.1) for the
chi-squared statistic or an I2 greater than 50% was considered to
indicate considerable heterogeneity. If the P-value for chi-square
test was >0.10 and the I2 value was <50%, a fixed effects model was
selected. In the cases for which the I2 value was >50%, a random
effects model was used [21,22]. Since, the total number of studies that
showed substantial heterogeneity was less than 10; t-statistics (the
Hartung-Knapp-Sidik-Jonkman method) were used instead of Z-test
in all random effects analysis to lower the error rate [23]. We also
conducted sensitivity analysis in terms of heterogeneous outcomes.
If the reported data were median (P25-P75), median (range), or mean
(standard error of mean), then mean and standard deviation were
calculated from these values [24]. We calculated the number needed
to treat (NNT) using a 95% CI based on the absolute risk reduction
as an estimate of the overall clinical impact of the intervention [25].
Publication bias was assessed by Begg’s funnel plot and Egger’s linear
regression test and a P value <0.05 was used to identify the presence
of a publication bias, or funnel plots for each data set were visually
assessed for asymmetry. If a publication bias was present, a trim-andfill
analysis was performed to evaluate the effect of that bias [26].
Ethics approval and consent to participate
This meta-analysis was approved by the Institutional Review
Board in Chung-Ang University Hospital in Seoul, South Korea. No
ethical approval or patient consent was required because all analyses
were based on previous published studies.
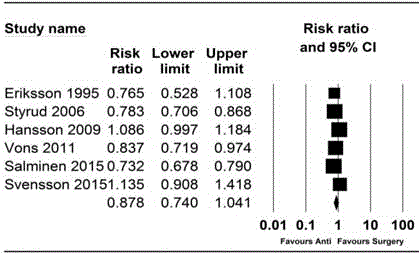
Figure 2
Figure 2
Forest plot showing the overall treatment efficacy of antibiotic
treatment and appendectomy.
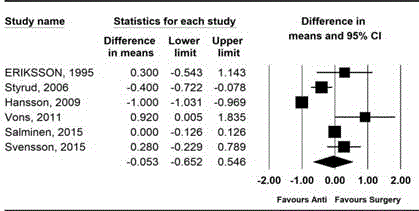
Figure 3
Figure 3
Forest plot showing the comparison of hospital stay between
antibiotic treatment group and appendectomy group.
Results
The search of OVID-MEDLINE, EMBASE, and the Cochrane
Central Register of Controlled Trials (CENTRAL), produced 115
studies, and 6 in manual research. After adjusting for duplicates, 114
studies remained. Of these, 104 studies were discharged, because
reviewing the title and abstracts indicated that these studies were not
of interest. The full texts of the remaining 10 studies were reviewed
in detail and 4 studies were excluded for the following reasons:
systematic review (n=1), lack of evidence of randomization (n=1),
study protocol (n=1) and retracted article (n=1) [17,27-29]. Thus, 6
studies with a total of 1,480 patients, of whom 751 patients received
antibiotic therapy while 729 patients underwent appendectomy, were
included in the final analysis as having met the inclusion criteria and
were included in this systematic review and meta-analysis (Figure 1).
Study description of the trials
A description and summary of each trial’s methodology is
shown in Table 1. All studies had only two arms, antibiotics or
appendectomy. Because of the nature of the treatment arms, none
of the studies was blinded. The studies were conducted mainly in
Sweden with the exception of one in France and one in Finland
[9-12,14]. Five studies are performed for adult patients except for
one study which was conducted in children [9-13]. Selection of the
patients in two studies was mainly on clinical grounds, while in
recently performed three studies computed tomography scans were
obtained from all [10-12,14]. There were differences in the choice of
antibiotic, antibiotic dosage, and duration of antibiotic therapy. The
details including number of patients, diagnosis and management are
summarized in Table 1.
Risk of bias
The random sequence generation method was clearly described
in three studies and one study allocated the patients using birth date
which meant that those with an uneven date of birth were allocated
to antibiotics and those with an even date of birth to appendicectomy
group [11-14]. The method for allocation concealment was described
in three studies [10,12,13]. Four RCTs were registered in clinical trial,
and conflict of interest was described in five studies [10-14]. The
overall risks of bias are shown in Table 2.
Initial and overall treatment efficacy
The initial treatment efficacy in the antibiotic group was 0.78 (95%
CI, 0.68 to 0.85; Pchi
2<0.001; I2=82.16%). The overall treatment efficacy
in the antibiotic group was 0.77 (95% CI, 0.68 to 0.84; Pchi
2<0.001;
I2=81.51%) vs. 0.91 (95% CI, 0.81 to 0.96; Pchi
2<0.001; I2=80.23%) in
the appendectomy group. However, the combined result showed
no evidence of difference for overall treatment efficacy between the
two groups (Risk ratio (RR), 0.88; 95% CI, 0.74 to 1.04; Pchi
2<0.001;
I2=90.98%; NNT=6) (Figure 2).
Complicated appendicitis
Among 172 patients who underwent appendectomy due to the
failure of antibiotic treatment, 37 patients (21.5%) were pathologically
diagnosed as perforated or gangrenous appendicitis (Event Rate
(ER) 0.23; 95% CI, 0.14 to 0.34; Pchi
2=0.071; I2=50.75%). Of the 729
patients who underwent appendectomy, 137 (18.8%) patients had
pathological diagnosis of perforated or gangrenous appendicitis (ER
0.23; 95% CI, 0.14 to 0.34; Pchi
2=0.071; I2=50.75%). The combined
result showed no evidence of difference for complicated appendicitis
between the two groups (Risk ratio (RR), 1.73; 95% CI, 0.76 to 3.94;
Pchi
2=0.001; I2=76.01%; NNT=-29).
Complications
The complication rate was compared between groups in five
studies [9-13]. The combined results showed no evidence of difference
(Risk Ratio (RR), 1.56; 95% CI, 0.48 to 5.05; Pchi
2=0.008; I2=71.21%;
NNT=186). The pooled complication rate in the antibiotics group
was 0.13 (95% CI, 0.006 to 0.25; Pchi
2<0.001; I2=73.20%).
Hospital stay
The hospital stay was compared in all included studies [9-14]. The
combined results showed no evidence of difference (MD, -0.05; 95%
CI, -0.65 to 0.55; Pchi
2<0.001; I2=98.24%). On performing sensitivity
analysis with removing Hansson’s study, heterogeneity was decreased
and hospital stay became shorter in the surgery group than the
antibiotics group, but was still insignificant (MD, 0.06; 95% CI, -0.26
to 0.38; Pchi
2=0.022; I2=64.89%) (Figure 3) [11].
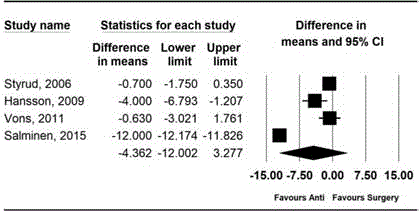
Sick leave
Sick leave was compared in four studies [10-13]. There were no
differences between the groups in days for sick leave (MD,-4.36; 95%
CI, -12.00 to 3.28; Pchi
2<0.001; I2=99.45%). On performing sensitivity
analysis with removing which defined duration of disability, and
which showed greatest difference between groups, significance did
not change significance did not change (MD, -5.59; 95% CI, -14.37
to 3.19; Pchi
2<0.001; I2=99.78%, MD, -1.47; 95% CI, -3.28 to 0.33;
Pchi
2=0.384; I2=0.00%). (Figure 4) [12,13].
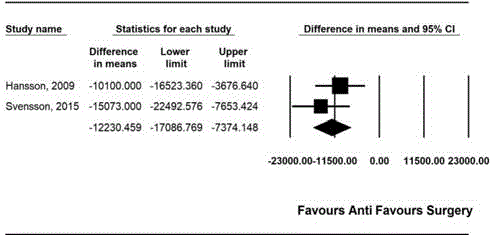
Cost
Cost was compared in two studies [11,14]. The combined results
showed that the cost was lower in the antibiotics group than in the
appendectomy group (MD, -12230.46; 95% CI, -17086.77 to -7374.15;
Pchi
2=0.321; I2=0.00) (Figure 5).
Publication bias
There was no evidence of publication bias detected by Egger’s
linear regression test and funnel plot. There was no P-value of Egger’s
regression test showing <0.05, which is indicative of publication bias.
Figure 4
Figure 4
Forest plot showing the comparison of sick-leave between antibiotic
treatment group and appendectomy group.
Figure 5
Figure 5
Forest plot showing the comparison of costs between antibiotic
treatment group and appendectomy group.
Table 1
Table 2
Discussion
This meta-analysis of six randomized controlled trials including 1,480 patients, of whom 751 patients received antibiotic therapy and 729 patients underwent appendectomy, has demonstrated that there was no significant difference in overall treatment efficacy for uncomplicated appendicitis between the antibiotic group and the appendectomy group. The overall treatment efficacy of the antibiotic group was 0.77 (95% CI, 0.68 to 0.84; Pchi 2<0.001; I2=81.51%) and that of the appendectomy group was 0.91 (95% CI, 0.81 to 0.96; Pchi 2<0.001; I2=80.23%) (Risk (RR), 0.88; 95% CI, 0.74 to 1.04; Pchi 2<0.001; I2=90.98%; NNT=6), and there were no significant differences in either complications, length of hospital stay, or incidence of complicated appendicitis. This meant that there was no increased risk of developing complicated appendicitis even if the patients underwent appendectomy after recurrence despite antibiotics treatment, suggesting that in the primary treatment of uncomplicated appendicitis, antibiotic treatment is as safe an option as appendectomy. To date, several meta-analyses have been published. However, most of these meta-analyses had several limitations, such as including withdrawn trial, non-randomized trial, or omitting some published randomized controlled trials [15-19]. Therefore, conflicting results were offered, and without definite evidence of superiority, appendectomy was thought to be the standard approach for treating uncomplicated appendicitis [17]. When compared with previous meta-analyses, the present meta-analysis included only relevant randomized trials, and a recently published trial [9-14]. There are several theoretical advantages to antibiotics treatment over appendectomy. First, non-operative management for uncomplicated appendicitis could not result in any surgery-related morbidity. Second, there is an absence of incisional pain in the patients treated non-operatively, and this could lead to less pain medication, and shorter sick-leave. Furthermore, several authors reported that recurrence after antibiotics treatment can be treated efficiently with the administration of a second course of antibiotics [30,31]. However, it could be controversial to conclude that antibiotic therapy was one of the standard treatments for uncomplicated appendicitis, because of several protocol design limitations in the included randomized controlled trials, such as the loose criteria chosen for the definition of acute appendicitis, type and duration of antibiotic treatment, unclear determination of the primary endpoint, and highly restrictive selection. First, acute appendicitis was diagnosed based on clinical examination without confirmation by radiology, or ultrasonography of the right iliac fossa [9-11]. Because of the absence of morphological investigation in the surgical group of two large trials, 10% to 15% of patients did not have appendicitis, and about 40% of patients had complicated appendicitis, which should be excluded [10,11]. Another limitation was the absence of consensus for the selection of antibiotics and duration of the treatment. The route, type and timing of antibiotics administration were variable between the studies. One study administered oral antibiotics as the first line treatment, with the intravenous route only used in patients with nausea and vomiting [12]. The intervention durations of intravenous antibiotics were various, from 24 hr to 72 hrs. The type of antibiotics was also variable, with three studies using a combination of ß-lactam (third-generation cephalosporins) and nitroimidasole antibiotics, two using a carbapenem antibiotic, and one using a combination of ß-lactam antibiotics [9-14]. The rate of Escherichia coli resistant to thirdgeneration cephalosporins is from 10% to 15% in Europe and the United States, and carbapenem is not considered as a first-line antibiotic for uncomplicated antibiotics [32,33]. To be effective treatment, although the antibiotics must provide broad-spectrum coverage for all the pathogens that might cause appendicitis, broadspectrum antibiotics have the potential problem of a risk of developing antibiotic resistance, hence, future trials should be conducted to determine adequate antibiotics with a more restricted antibacterial spectrum. The next limitation is that the primary end point was not stated or unclear, and if it was stated, the definition for the same end point differed from trial to trial [9-11]. In particular, in the appendectomy group, the definition of treatment efficacy was not obvious. Two studies defined normal appendix on pathology as the treatment failure in the appendectomy group because they regarded normal appendix as significant complication or not appropriate surgical indication [11,14]. Since patients were randomized, the same rate of negative appendicitis could be expected in both antibiotics and appendectomy treatment group. And by considering negative appendicitis only in the appendectomy group as a treatment failure, the results may favor the antibiotics treatment. Since the limits of the Relative Risk confidence interval are closed to 1, considering negative appendicitis only in one group or in both groups may change the conclusion. The relatively large proportions of patients lost to followup by 1 year in two trials were also problematic. Another limitation is that most of the appendectomies were performed using an open operative approach [10,11]. Nowadays, for uncomplicated appendicitis, laparoscopic appendectomy has become the preferred method with less pain, shortened hospital stay, faster return to normal activity, and fewer wound infections and the European Association for Endoscopic Surgery (EAES), the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) and World Society of Emergency Surgery (WSES) have recommended laparoscopic surgery as the gold standard [3-7]. Considering that the most common cause of morbidity in the surgical group of this metaanalysis related to wound infection, the complication rate of the appendectomy group might become lower, if the appendectomy was performed using laparoscopy [34-36]. Furthermore, recently there have been several reports that laparoscopic appendectomy can be performed successfully on an outpatient basis. In this regard, future trials should compare antibiotics group and laparoscopic appendectomy group with an admission basis or with an outpatient basis [37-39]. The high rate of protocol violation is also a limitation. In one study, 50% of patients allocated to antibiotic treatment underwent appendectomy immediately after randomization [11]. The recurrence rates may not be representative of the true recurrence rates after antibiotic therapy because of the relatively short follow-up of within one year. Unfortunately, there are still no follow-up data extending beyond 1 year after conservative management. Further long-term surveillance for the antibiotic group is required to establish the true recurrence rate. The obvious advantage of surgery is the complete and long-term avoidance of further appendicitis. The present meta-analysis showed that 28.8% of the patients, i.e., about one of three patients, experienced readmission and surgery after antibiotics treatment. Despite the evidence reinforcing the safety and efficacy of primary antibiotic therapy for the patients with uncomplicated appendicitis this treatment has not yet gained widespread acceptance. Also, it should be considered that the general lifetime risk of 6.7% to 8.6% for acute appendicitis persists in antibiotics group patients [15,40,41]. The low rate of false-negative appendectomies also shows that unnecessary appendectomies are rare in these times of improved diagnostic tools. Therefore, to establish antibiotics treatment as a first line alternative treatment for uncomplicated appendicitis, further study of risk factors for recurrence after antibiotics treatment, and to decrease treatment failure should be performed. Regardless of limitations of already published randomized controlled studies, our study applied rigorous methodology, excluding withdrawn article and non-randomized trial, and including omitted study in other meta-analyses, to compare safety and efficacy of the antibiotic treatment with appendectomy as a primary treatment of uncomplicated acute appendicitis.
Conclusion
Despite the above limitations, this meta-analysis reveals that antibiotic therapy could be considered as an alternative option in the treatment of patients with acute uncomplicated appendicitis. If the patient is willing to accept initial failure or a subsequent recurrence, and forgoing appendectomy, antibiotic treatment can be recommended. However, to be established as the gold standard of treatment, further well-organized randomized controlled trials should be performed.
References
- McBurney C. II. The Indications for Early Laparotomy in Appendicitis. Ann surg. 1891;13(14):233-54.
- Coldrey E. Treatment of Acute Appendicitis. Br Med J. 1956;2(5007):1458-61.
- Wangensteen OH, Buirge RE, Dennis C, Ritchie WP. Studies in the Etiology of Acute Appendicitis: The Significance of the Structure and Function of the Vermiform Appendix in the Genesis of Appendicitis a Preliminary Report. Ann Surg. 1937;106(5):910-42.
- Wangensteen OH, Dennis C. Experimental Proof of the Obstructive Origin of Appendicitis in Man. Ann Surg. 1939;110(4):629-47.
- Andersson R, Hugander A, Thulin A, Nystrom PO, Olaison G. Clusters of acute appendicitis: further evidence for an infectious aetiology. Int J Epidemiol. 1995;24(4):829-33.
- Arnbjornsson E, Bengmark S. Role of obstruction in the pathogenesis of acute appendicitis. Am J Surg. 1984;147(3):390-2.
- Sisson RG, Ahlvin RC, Harlow MC. Superficial mucosal ulceration and the pathogenesis of acute appendicitis. Am J Surg. 1971;122(3):378-80.
- Rivera-Chavez FA, Peters-Hybki DL, Barber RC, Lindberg GM, Jialal I, Munford RS, et al. Innate immunity genes influence the severity of acute appendicitis. Ann Surg. 2004;240(2):269-77.
- Eriksson S, Granstrom L. Randomized controlled trial of appendicectomy versus antibiotic therapy for acute appendicitis. Br J Surg.1995;82(2):166-9.
- Styrud J, Eriksson S, Nilsson I, Ahlberg G, Haapaniemi S, Neovius G, et al. Appendectomy versus antibiotic treatment in acute appendicitis. a prospective multicenter randomized controlled trial. World J Surg. 2006;30(6):1033-37.
- Hansson J, Korner U, Khorram-Manesh A, Solberg A, Lundholm K. Randomized clinical trial of antibiotic therapy versus appendicectomy as primary treatment of acute appendicitis in unselected patients. Br J Surg. 2009;96(5):473-81.
- Vons C, Barry C, Maitre S, Pautrat K, Leconte M, Costaglioli B, et al. Amoxicillin plus clavulanic acid versus appendicectomy for treatment of acute uncomplicated appendicitis: an open-label, non-inferiority, randomised controlled trial. Lancet. 2011;377(9777):1573-9.
- Salminen P, Paajanen H, Rautio T, Nordstrom P, Aarnio M, Rantanen T, et al. Antibiotic Therapy vs Appendectomy for Treatment of Uncomplicated Acute Appendicitis: The APPAC Randomized Clinical Trial. JAMA. 2015;13(23):2340-8.
- Svensson JF, Patkova B, Almstrom M, Naji H, Hall NJ, Eaton S, et al. Nonoperative treatment with antibiotics versus surgery for acute nonperforated appendicitis in children: a pilot randomized controlled trial. Ann Surg. 2015;261(1):67-71.
- Varadhan KK, Neal KR, Lobo DN. Safety and efficacy of antibiotics compared with appendicectomy for treatment of uncomplicated acute appendicitis: Meta-analysis of randomised controlled trials. BMJ. 2012;344:2156.
- Varadhan KK, Humes DJ, Neal KR, Lobo DN. Antibiotic therapy versus appendectomy for acute appendicitis: a meta-analysis. World J Surg. 2010;34(2):199-209.
- Kirby A, Hobson RP, Burke D, Cleveland V, Ford G, West RM. Appendicectomy for suspected uncomplicated appendicitis is associated with fewer complications than conservative antibiotic management: a meta-analysis of post-intervention complications. J Infect. 2015;70(2):105-10.
- Liu K, Fogg L. Use of antibiotics alone for treatment of uncomplicated acute appendicitis: A systematic review and meta-analysis. Surgery. 2011;150(4):673-83.
- Ansaloni L, Catena F, Coccolini F, Ercolani G, Gazzotti F, Pasqualini E, et al. Surgery versus conservative antibiotic treatment in acute appendicitis: a systematic review and meta-analysis of randomized controlled trials. Dig Surg. 2011;28(3):210-21.
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. JCE. 2009;62(10):1-34.
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557-60.
- Higgins J, Green S. Cochrane handbook for systematic reviews of interventions. Version 5.1.0 the Cochrane Collaboration. 2011.
- IntHout J, Ioannidis JP, Borm GF. The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med Res Methodol. 2014;14:25.
- Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13.
- Naing C, Aung K, Mak JW. Reporting 'number needed to treat' in meta-analyses: a cross-sectional study. J Evidence-Based Med. 2012;5(4):232-7.
- Duval S, Tweedie R. Trim and fill: A simple funnel-plot-based method of testing and adjusting for publication bias in meta-analysis. Biometrics. 2000;56(2):455-63.
- Turhan A, Kapan S, Kuturkcu E, Yigitbas H, Hatipoglu S, Aygun E. Comparison of operative and non operative management of acute appendicitis. Ulus Travma Acil Cerrahi Derg. 2009;15(5):459-62.
- Paajanen H, Gronroos JM, Rautio T, Nordstrom P, Aarnio M, Rantanen T, et al. A prospective randomized controlled multicenter trial comparing antibiotic therapy with appendectomy in the treatment of uncomplicated acute appendicitis (APPAC trial). BMC Surg. 2013;13:3.
- Malik AA, Bari SU. Conservative management of acute appendicitis. J Gastrointestinal Surg. 2009;13(5):966-70.
- Kirkil C, Yigit MV, Aygen E. Long-term results of nonoperative treatment for uncomplicated acute appendicitis. Turk J Gastroenterol. 2014;25(4):393-7.
- Di Saverio S, Sibilio A, Giorgini E, Biscardi A, Villani S, Coccolini F, et al. The NOTA Study (Non Operative Treatment for Acute Appendicitis): prospective study on the efficacy and safety of antibiotics (amoxicillin and clavulanic acid) for treating patients with right lower quadrant abdominal pain and long-term follow-up of conservatively treated suspected appendicitis. Ann Surg. 2014;260(1):109-17.
- European. Antimicrobial resistance surveillance in Europe 2014. ECDC. 2015:9-21.
- Antibiotic resistance threats in the United States, 2013. CDC. 2013.
- Korolija D, Sauerland S, Wood-Dauphinee S, Abbou CC, Eypasch E, Caballero MG, et al. Evaluation of quality of life after laparoscopic surgery: evidence-based guidelines of the European Association for Endoscopic Surgery. Surg Endosc. 2004;18(6):879-97.
- Korndorffer JR Jr, Fellinger E Reed W. SAGES guideline for laparoscopic appendectomy. Surg Endosc. 2010;24(4):757-61.
- Di Saverio S, Birindelli A, Kelly MD, Catena F, Weber DG, Sartelli M, et al. WSES Jerusalem guidelines for diagnosis and treatment of acute appendicitis. World J Emerg Surg. 2016;11:34.
- Frazee RC, Abernathy SW, Isbell CL, Isbell T, Regner JL, Smith RD. Outpatient Laparoscopic Appendectomy: Is It Time to End the Discussion? J Am Coll Surg. 2016;222(4):473-7.
- Frazee R, Burlew CC, Regner J, McIntyre R, Peltz E, Cribari C, et al. Outpatient laparoscopic appendectomy can be successfully performed for uncomplicated appendicitis: A Southwestern Surgical Congress multicenter trial. Am J Surg. 2017;214(6):1007-9.
- Gee K, Ngo S, Burkhalter L, Beres AL. Safety and feasibility of same-day discharge for uncomplicated appendicitis: A prospective cohort study. J Pediatr Surg. 2018;53(5):988-90.
- Rollins KE, Varadhan KK, Neal KR, Lobo DN. Antibiotics versus Appendicectomy for the treatment of uncomplicated Acute Appendicitis: An Updated Meta-Analysis of Randomised Controlled Trials. World J Surg. 2016;40(10):2305-18.
- Addiss DG, Shaffer N, Fowler BS, Tauxe RV. The epidemiology of appendicitis and appendectomy in the United States. American J Epidemiol. 1990;132(5):910-25.