Research Article
Does Iloprost Ameliorate Oxidative Stress Induced by Obstructive Jaundice? An Experimental Study in Rats
Onur Bayraktar1, AhatAndican2, Gülnur Andican3, Sibel Erdamar4, ÇetinKaraca5, Erman Aytac6, Barış Bayraktar7 and Ilknur Erenler Bayraktar1*
1Department of General Surgery, T.C. Istanbul Bilim University, Turkey
2Department of General Surgery, Istanbul Okan University, Turkey
3Department of Medical Biochemistry, Istanbul University, Turkey
4Department of Pathology, Acibadem University, Turkey
5Department of Medical Biochemistry, Istanbul Üniversity, Turkey
6Department of General Surgery, Acibadem University, Turkey
7Department of General Surgery, Konak Hospital, Turkey
*Corresponding author: Ilknur Erenler Bayraktar, Department of General Surgery, T.C. Istanbul Bilim University, Turkey
Published: 12 Oct, 2018
Cite this article as: Bayraktar O, Andican A, Andican G,
Erdamar S, Karaca Ç, Aytac E, et al.
Does Iloprost Ameliorate Oxidative
Stress Induced by Obstructive
Jaundice? An Experimental Study in
Rats. Clin Surg. 2018; 3: 2160.
Abstract
Background/Aims: Iloprost is a synthetic analogue of epoprostenol which is synthesized by
endothelial cells from arachidonic acid. We aimed to evaluate presumed hepatoprotective and
antioxidant effects of iloprost in experimentally created obstructive jaundice in rats.
Materials and Methods: Thirty-two adult male Wistar albino rats were randomly divided into
four groups: sham group (S) (n=8); after sham operation, iloprost administered (SI) group (n=8);
Obstructive Jaundice (OJ) group (n=8); iloprost administered and obstructive jaundice group (OJI)
(n=8). The rats were scarified ten days after the induction of obstructive jaundice. Blood levels of
isoprostane, protein carbonyl, lipid hydro peroxide and 8-hydroxy-2-deoxyguanosine (8-OHdG)
were measured.
Results: Levels of the oxidative stress products including protein carbonyl [mean, (40.46 vs. 24.7
nmol/ml, p=0.005)], lipid hydro peroxide [mean, (5.11 vs. 1.85 M mol/ml, p=0.001)] and 8-OHdG
[mean, (1.53 vs. 1.13 ng/ml, p=0.03)] in the OJI group were significantly lower than the OJ group.
Conclusion: Iloprost supports antioxidant activity in obstructive jaundice without causing further
liver damage in rats.
Keywords: Antioxidants; Bile duct ligation; Iloprost; Obstructive jaundice; Oxidative stress
Introduction
Obstructive jaundice is a clinical entity occurring after blockage of bile flow from the liver
to the small intestine. Direct hyperbilirubinemia developed after obstruction of bile flow causes
several disruptions including immune suppression, liver toxicity, coagulation disorders, intestinal
barrier damage, increased oxidative stress, bacteremia and endotoxemia [1]. Oxidative stress is an
imbalance between the production and degradation of reactive oxygen species in vivo. It damages the
hepatocytes and intestinal cells in obstructive jaundice [2,3]. Patients with obstructive jaundice are
prone to develop serious complications during treatment process such as sepsis, renal complications
and even death [4]. Oxidative stress has a major role in the pathogenesis of hepatic injury and sepsis
[3,5]. New treatment alternatives reducing oxidative stress may be effective to prevent complications
associated with obstructive jaundice.
Iloprost is a synthetic analogue of epoprostenol (prostaglandin I2, prostacyclin) which is
synthesized by endothelial cells from arachidonic acid. Beside the vasodilatory, antiplatelet and
cytoprotective effects of the iloprost, it has fibrinolytic activity and it increases red cell elasticity,
increases smooth muscle proliferation and supports microcirculation [6,7]. Considering its beneficial
effects, iloprost seems as a good candidate to be evaluated experimentally against oxidative stress
induced by obstructive jaundice. In this study, the effects of systematic application of iloprost on
oxidative stress parameters and liver histopathology were assessed in rats with obstructive jaundice.
This is the first study showing antioxidant effects of iloprost against oxidative stress induced by
obstructive jaundice.
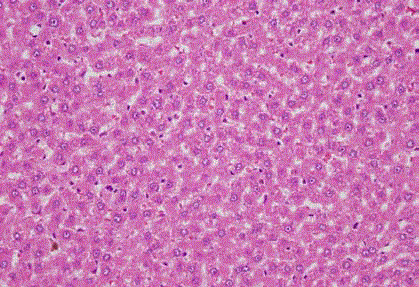
Figure 1
Figure 1
Control group. It’s seen almost normal looking hepatocytes except
mild hydropic changes. (HE x 400).
Materials and Methods
The Ethics Committee of Experimental Animal Research of
our university approved the study protocol. Thirty two Wistar-
Albino male rats, weighing 250 g to 300 g, were used. The rats were
housed four in per cage, under constant temperature (20°C - 22°C)
and humidity (50F) with an artificial 12 hr light-dark cycle and
randomized into four groups. Obstructive jaundice was induced
by ligation of the common bile duct. The rats were fed ad libitum
with standardized industrial rat prey during the study period except
12 hrs prior to operation. Twelve hrs before the operation feeding
was stopped. The rats were divided randomly into four equal weightmatched
groups containing eight rats each. The study groups were
sham (S) group; after sham operation, intraperitoneal iloprost
applied (SI) group; Obstructive Jaundiced (OJ) group (the control
group); after obstructive jaundice induction, intraperitoneal iloprost
applied (OJI) group. A daily dose of 2 mcg/kg iloprost was given
to the rats in the SI and OJI groups. Beneficial effects of the same
dose iloprost in Wistar rats were shown previously [8]. The S and OJ
groups had been administered daily same volume 0.9 % NaCl solution
intraperitoneally, while the others receiving iloprost injections. Ten
days after the induction of obstructive jaundice, blood samples were
taken with intra cardiac puncture under general anesthesia. A cervical
dislocation was performed before liver tissue sampling.
Bile duct ligation
The rats were anesthetized by intraperitoneal administration of
100 mg/kg ketamine hydrochloride (Ketalar; Parke- Davis, Istanbul,
Turkey) and 5 mg/kg xylasine (Rompun; Bayer, Istanbul, Turkey).
A 2 cm midline incision was made. In the Sham groups, the gastro
duodenal ligament was isolated and the common bile duct was
mobilized without any ligation or division. In the obstructive jaundice
groups, the common bile ducts were ligated from 2 different points
in middle with 4/0 silk sutures after common bile duct mobilization
and divided. The abdominal incisions were closed in two layers with
continuous 4/0 polyglactin and 4/0 silk sutures respectively. After the
procedure, the rats were put into the cages.
Biochemical analyses
The blood samples were stored in silicone tubes. The blood samples
were centrifuged for five minutes at 6500 rpm. The serums were then
separated. The analysis of plasma 8-hydroxy-2-deoxyguanosine (8-
OHdG), an indicator of oxidative damage on DNA, was made with
competitive ELISA method using ELISA kits (NWLSSTM 8-OHdG
ELISA, Northwest and Vancouver Canada). The concentration of
samples was calculated using the standard calibration curve. The
standard concentration ranges were 0.125 ng/ml, 0.250 ng/ml, 0.5 ng/
ml, 1 ng/ml, 4 ng/ml and 10 ng/ml respectively.
The plasma isoprostane possesses potent biological activity
and are released from tissues in a number of disease states, such as
during is chemic injury and neurodegenerative brain disease. The
measurement of isoprostane is considered to be the «gold standard»
to assess oxidative stress in various human diseases. The analysis of
isoprostane was made with competitive ELISA method using ELISA
kits (NWLSSTM 8-OHdG ELISA, Northwest, Vancouver, Canada).
The concentration of samples was calculated using the standard
calibration curve, and the concentration ranges were 0.005 ng/ml, 0.1
ng/ml, 1 ng/ml, 5 ng/ml, 10 ng/ml, 50 ng/ml, 100 ng/ml respectively.
The plasma protein carbonyl, which reveals after the oxidation
of amino-acid chains, analysis was made using a commercial kit
(NWLSSTM Protein Carbonyl Assay, Northwest, Vancouver,
Canada) based on spectrophotometric principles. Absorbance of the
samples was measured at a wavelength of 320 nm. Concentrations of
the samples were measured by using molar absorptivity coefficient
(22.000 M-1, cm-1) of protein carbonyl.
The plasma lipid hydro peroxide (LOOH), an indicator of the
peroxidation of lipids and damage of the cell membrane, analysis
was made using a commercial kit (NWLSSTM Protein Carbonyl
Assay, Northwest, Vancouver Canada) based on spectrophotometric
principles. Absorbance of the samples and calibrator was measured
at a wavelength of 560 nm. Concentrations of the samples were
measured by using molar absorptivity coefficient (33.700 M-1, cm-1)
of protein carbonyl.
Histopathologic examination
The liver samples were placed in 10% buffered formaline solution
for 3 hrs. After alcohol, acetone, xylene and paraffin procedures, the
blocks were prepared. Then four microns cuts were obtained from
the blocks and they were stained with hematoxylin and eosin (H&E)
stain. Knodell index was used for histologic grading and staging [9].
The prepared slides were evaluated under light microscope with
magnification of 40x and 100x. Their photos were taken by the camera
connected to the microscope. The slides were assessed between -
(0) and +++ (3) according to adiposity, hydropic degeneration,
Kupffer cell proliferation/ sinusoidal cell proliferation, bile stasis,
inflammatory activity in portal space (mononuclear cell infiltration),
bile stasis, cholangitis (neutrophil and mononuclear cell infiltration
to bile ducts), micro abscess formation.
Statistical analysis
The data were presented as mean ± standard deviation.
Differences among the groups were evaluated by Kruskal-Wallis test
by using SPSS 12 (SPSS Inc. Chicago USA). P values less than 0.05
were considered significant. When statistically significant values were
identified, Mann-Whitney U test was used for paired comparisons
and Bonferroni correction was used to detect statistical significance.
P values less than 0.0083 were considered significant.
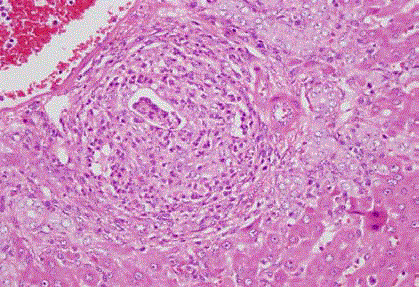
Figure 2
Figure 2
Iloprostgrubu: mixt inflammatory cells infiltrating portal area is
associated with ductular proliferation.
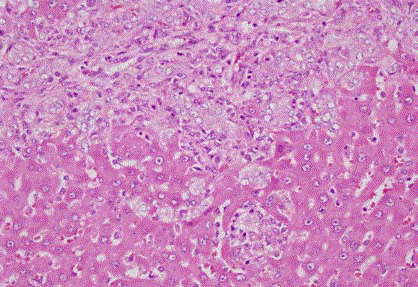
Figure 3
Results
No rats died during study period.
Biochemical parameters
There was no statistically significant difference in biochemical
parameters between S and SI groups (Table 1). 8-OHdG (p=0.006),
isoprostane (p=0.003), lipid hydro peroxide (p=0.001) parameters
were found to be increased in OJ group compared to S group. Protein
carbonyl (p=0.005) and lipid hydro peroxide (p=0.001) parameters
were found to be decreased in OJI group compared to OJ group.
Isoprostane (p=0.003) were found to be increased in OJI group
compared to SI group.
Histopathologic parameters
There were no statistically significant differences in histologic
parameters between S (Figure 1) and SI (Figure 2) groups. Bile stasis
(p=0.001), portal inflammation (p=0.001), bile ductus proliferation
(p=0.001) and cholangitis (p=0.001) parameters were found to be
increased in OJ (Figure 3) group compared to S group.
Hydropic degeneration (p=0.006), Kupffer cell proliferation
(p=0.004), bile stasis (p=0.000), portal inflammation (p=0.001), bile
ductus proliferation (p=0.000) and cholangitis (p=0.001) parameters
were found to be increased in OJI group compared to SI group. No
statistically significant differences were observed between OJ and OJI
groups in histologic evaluation. Micro abscess formation in liver were
observed in one rat (p=0.37). The histopathologic results between of
the S versus SI groups and OJ versus OJI groups were given (Table 2).
Table 1
Discussion
Internal or external biliary drainage is the cure for obstructive
jaundice. Our experimental obstructive jaundice model does not
reflect real clinical scenario in general. Palliative drainage or definitive
restoration bile flow can be achieved in majority of the patients with
obstructive jaundice. However, high bilirubin levels may maintain in
certain time period even after bile drainage or attempts to eliminate
bile flow obstruction may fail. Under these circumstances prevention
from the harmful effects of hyperbilirubinemia is needed until the
definitive treatment. Since extrahepatic bile drainage obstructed
permanently, severity of obstructive jaundice increased gradually
during study period. Nevertheless, iloprost supplementation reduced
DNA oxidation under gradually increasing hyperbilirubinemia.
Since the liver has a high regeneration capacity and capable to
regain its functions, protecting integrity of DNA may be beneficial
during management of obstructive jaundice. Hyperbilirubinemia
may be genotoxic via its oxidant effects [10]. In addition to its use
to monitor DNA damage, 8-OHdG has been suggested as a factor
initiating carcinogenic pathways [11]. An anti-metastatic effect of
iloprost was shown decades ago [12]. Prostacyclin analogues may
reduce risk of metastasis by interacting between tumor cell and host
[13]. It has been shown that iloprost can be used in cancer patients
safely for treatment of systemic sclerosis [14]. Iloprost may prevent
genotoxicity under hyper bilirubinemic condition. However, there is
no clinical evidence supporting anti-neoplastic effects of iloprost. We
observed beneficial effects of iloprost on oxidative stress parameters,
but there was no improvement in histological evaluation of the liver.
As we mentioned above, we blocked the bile flow permanently and
created a gradually increasing hyper bilirubinemic condition as an
experimental model. This may be the major factor for not observing
any favorable histologic results. Isik et al. declared that combination
of antioxidants may provide synergistic effects and increase each
other’s activity. Combination of iloprost with other antioxidants may
be analyzed to obtain better results on histological evaluation of the
liver [15].
Obstructive jaundice complicates many vital functions
maintaining homeostasis [16]. High bile acid levels, systemic
endotoxemia and associated inflammatory response, over expression
of inducible nitric oxide synthase, increased neutrophil chemotaxis,
superoxide anion production and decreased levels of vitamin
E augment oxidative damage in obstructive jaundice [17,18].
Oxidative stress is hepatotoxic [2,3]. Biliary cholestasis induces lipid
peroxidation by reducing antioxidant capacity [19]. Obstructive
jaundice caused oxidative damage in our control group. Since our
surgeons are experienced on obstructive jaundice model in rats
with the same technique and we carry out continuous research on
obstructive jaundice [20,21]. We did not measure blood bilirubin,
aspartate aminotransferase and alanine aminotransferase to prove
hyperbilirubinemia and associated liver damage biochemically in
the rats which were included to this study for minimizing expenses
of the biochemical analyses. Although there is some data regarding
to antioxidant effect of iloprost [22], use of iloprost in OJ induced
oxidative stress has been poorly studied. Considering the significant
decrease in oxidative stress parameters in OJI group compared to
OJ group, our results may reveal that iloprost decrease the oxidative
damage caused by oxidative jaundice.
Nowadays, a growing number of studies have suggested that
reduction of oxidative stress is considered to have an important
role for the prevention of liver damage due to cholestasis [23]. The
antioxidant mechanisms of iloprost are under investigation [24].
Atherosclerosis, senescence and oxidative stress reduce prostacyclin
production by inhibiting cyclooxygenase which is the major
enzyme in prostanoids synthesis [25]. Since oxidative stress causes
endothelial dysfunction, prostacyclin production decreases [26].
Lack of prostacyclin may worsen microcirculation and subsequently
augment production of oxidants as a vicious cycle. Toxic effects of
oxidants on endothelium well known for decades and obstructive
jaundice complicates hepatic microcirculation [27,28]. Iloprost
supplementation may ameliorate microcirculation, improve tissue
oxygenation and reduce production of oxidants in obstructive
jaundice. It has been clinically and experimentally shown that iloprost
improves micro vascular functions [29,30]. In our OJ group, iloprost
may have decreased the oxidation of the proteins and lipids with its
effect on hepatic microcirculation.
In conclusion, iloprost supports antioxidant activity in obstructive
jaundice without causing further liver damage in rats. New studies
to explain molecular pathways of antioxidant activities supported by
iloprost are needed.
References
- Guicciardi ME, Malhi H, Mott JL, Gores GJ. Apoptosis and Necrosis in the Liver. Compr Physiol. 2013;3(2): 977-1010.
- Liu TZ, Lee KT, Chern CL, Cheng JT, Stern A, Tsai LY. Free radical-triggered hepatic injury of experimental obstructive jaundice of rats involves overproduction of proinflammatory cytokines and enhanced activation of nuclear factor kappaB. Ann Clin Laboratory Sci. 2001;31(4):383-90.
- Vendemiale G, Grattagliano I, Lupo L, Memeo V, Altomare E. Hepatic oxidative alterations in patients with extra-hepatic cholestasis. Effect of surgical drainage. J Hepatol. 2002;37(5):601-5.
- Pain JA, Cahill CJ, Bailey ME. Perioperative complications in obstructive jaundice: therapeutic considerations. Br J Surg. 1985;72(12):942-5.
- Allameh A, Razavi-Azarkhiavi K, Mohsenifar A, Jamali-Zavarei M. Effect of acute ethanol treatment on biochemical and histopathological factors in rat liver in an experimental sepsis model. Pathol Res Pract. 2012;208(6):331-7.
- Della Bella S, Molteni M, Mocellin C, Fumagalli S, Bonara P, Scorza R. Novel mode of action of iloprost: in vitro down-regulation of endothelial cell adhesion molecules. Prostaglandins Other Lipid Mediat. 2001;65(2-3):73-83.
- Grant SM, Goa KL. Iloprost. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in peripheral vascular disease, myocardial ischaemia and extracorporeal circulation procedures. Drugs. 1992;43(6):889-924.
- Vasiliadis K, Pramateftakis MG, Blouhos K, Mantzoros I, Koliakos G, Zaraboukas T, et al. Effect of iloprost on impaired anastomotic healing caused by 5-fluorouracil plus leucovorin. Dis Colon Rectum. 2007;50(6):899-907.
- Knodell RG, Ishak KG, Black WC, Chen TS, Craig R, Kaplowitz N, et al. Formulation and application of a numerical scoring system for assessing histological activity in asymptomatic chronic active hepatitis. Hepatology. 1981;1(5):431-5.
- Basu S, De D, Dev Khanna H, Kumar A. Lipid peroxidation, DNA damage and total antioxidant status in neonatal hyperbilirubinemia. Journal of perinatology: official journal of the California Perinatal Association. 2014;34(7):519-23.
- Valavanidis A, Vlachogianni T, Fiotakis C. 8-hydroxy-2' -deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2009;27(2):120-39.
- Sava G, Perissin L, Zorzet S, Piccini P, Giraldi T. Antimetastatic action of the prostacyclin analog iloprost in the mouse. Clin Experiment Metastasis. 1989;7(6):671-8.
- Schneider MR, Tang DG, Schirner M, Honn KV. Prostacyclin and its analogues: antimetastatic effects and mechanisms of action. Cancer metastasis reviews. 1994;13(3-4):349-64.
- Pehlivan Y, Turkbeyler IH, Balakan O, Sevinc A, Yilmaz M, Bakir K, et al. Possible anti-metastatic effect of iloprost in a patient with systemic sclerosis with lung cancer: a case study. Rheumatol Int. 2012;32(5):1437-41.
- Isik A, Peker K, Gursul C, Sayar I, Firat D, Yilmaz I, et al. The effect of ozone and naringin on intestinal ischemia/reperfusion injury in an experimental model. Int J Surg. 2015;21:38-44.
- Scott-Conner CE, Grogan JB. The pathophysiology of biliary obstruction and its effect on phagocytic and immune function. J Surg Res. 1994;57(2):316-36.
- Sakaguchi S, Furusawa S, Yokota K, Sasaki K, Takayanagi M, Takayanagi Y. The enhancing effect of tumour necrosis factor-alpha on oxidative stress in endotoxemia. Pharmacol Toxicol. 1996;79(5):259-65.
- Tsai LY, Lee KT, Lu FJ. Biochemical events associated with ligation of the common bile duct in Wistar rats. J Formosan Med Assoc = Taiwan yi zhi. 1997;96(1):17-22.
- Pastor A, Collado PS, Almar M, Gonzalez-Gallego J. Antioxidant enzyme status in biliary obstructed rats: effects of N-acetylcysteine. J Hepatol. 1997;27(2):363-70.
- Alturfan AA, Aytac E, Emekli-Alturfan E, Yarat A, Saribeyoglu K, Pekmezci S, et al. Serum total sialic acid as a novel complementary candidate marker of hepatic damage in obstructive jaundice. Ann Clin Lab Sci. 2014;44(1):56-61.
- Aydin S, Aytac E, Uzun H, Altug T, Mansur B, Saygili S, et al. Effects of Ganoderma lucidum on obstructive jaundice-induced oxidative stress. Asian J Surg. 2010;33(4):173-80.
- Erre GL, Passiu G. Antioxidant effect of Iloprost: current knowledge and therapeutic implications for systemic sclerosis. Reumatismo. 2009;61(2):90-7.
- Padillo FJ, Cruz A, Navarrete C, Bujalance I, Briceno J, Gallardo JI, et al. Melatonin prevents oxidative stress and hepatocyte cell death induced by experimental cholestasis. Free Radic Res. 2004;38(7):697-704.
- Aytac E, Seymen HO, Uzun H, Dikmen G, Altug T. Effects of iloprost on visual evoked potentials and brain tissue oxidative stress after bilateral common carotid artery occlusion. Prostaglandins Leukot Essent Fatty Acids. 2006;74(6):373-8.
- Whorton AR, Montgomery ME, Kent RS. Effect of hydrogen peroxide on prostaglandin production and cellular integrity in cultured porcine aortic endothelial cells. J Clin Invest. 1985;76(1):295-302.
- Giugliano D, Ceriello A, Paolisso G. Oxidative stress and diabetic vascular complications. Diabetes care. 1996;19(3):257-67.
- Sacks T, Moldow CF, Craddock PR, Bowers TK, Jacob HS. Oxygen radicals mediate endothelial cell damage by complement-stimulated granulocytes. An in vitro model of immune vascular damage. J Clin Invest. 1978;61(5):1161-7.
- Okaya T, Nakagawa K, Kimura F, Shimizu H, Yoshidome H, Ohtsuka M, et al. Obstructive jaundice impedes hepatic microcirculation in mice. Hepatogastroenterology. 2008;55(88):2146-50.
- Ciuffetti G, Sokola E, Lombardini R, Pasqualini L, Pirro M, Mannarino E. The influence of iloprost on blood rheology and tissue perfusion in patients with intermittent claudication. Kardiol Pol. 2003;59(9):197-204.
- Rasmussen I, Arvidsson D, Zak A, Haglund U. Splanchnic and total body oxygen consumption in experimental fecal peritonitis in pigs: effects of dextran and iloprost. Circ Shock. 1992;36(4):299-306.