Research Article
Coronary Switch: Total Reversal of Coronary Circulation in Acute Canine Model
Ahmed Elwatidy F1,2*, Jeffrey Danetz1, Ibrahim Hegab M1, Cynthia Allen1, Mohamed Alfagih R2 and Anwar Abd-Elfattah S1
1Department of Surgery, Medical College of Virginia Virginia Commonwealth University, USA
2Department of Surgery, Prince Sultan Cardiac Center Riyadh, Saudi Arabia
*Corresponding author: Ahmed Elwatidy, Department of Surgery, Prince Sultan Cardiac Center Riyadh, Saudi Arabia
Published: 21 Sep, 2018
Cite this article as: Ahmed Elwatidy F, Danetz J, Ibrahim
Hegab M, Allen C, Mohamed Alfagih R,
Anwar Abd-Elfattah S. Coronary Switch:
Total Reversal of Coronary Circulation
in Acute Canine Model. Clin Surg. 2018;
3: 2124.
Abstract
Background: Till date there is no optimal coronary revascularisation technique for diffuse
coronary artery disease. This study was conducted to proof the concept of total coronary venous
retro perfusion, for end-stage coronary artery disease patients. The coronary venous circulation is
arterialised while the coronary arteries drain the heart to the right atrium “Coronary Switch”.
Methods: Sixteen adult Mongrel dogs were used. After preparation for bypass, the heart was
instrumented with minor axis son micrometric crystals, carotid and left ventricular Millar catheters.
Colored microspheres were used for Regional Myocardial Blood Flow “RMBF” measurements. After
aortic cross clamping and cardioplegia, the coronary sinus ostium was closed, and then a 3 mm
Gore-Tex tube was anastomosed to the coronary sinus and its top-end to the aorta. A similar tube
was sequentially anastomosed between the three coronary arteries and ended in the right atrium.
Coronary switch was established by occluding the coronary arteries proximal to the anastomoses.
Results: The mean left ventricular function was 85,820.1 dyn. Cm2 "Preoperative" versus 87,984.2
"Postoperative" (p=NS). The RMBF was 1.38 ± 1.1 ml/min/gm “Preop” versus 1.44 ± 0.98 in
combined "ante/retro flow" and 1.58 ± 0.29, 1.28 ± 0.19, and 1.45 ± 0.35 at 60, 120, 180 minutes after
"Coronary Switch" (p=NS).
Conclusion: Primary results support “Coronary Switch” as an alternative therapeutic modality for
end-stage coronary artery disease. Survival studies and long-term follow-up are needed.
Introduction
Although the initial attempts to treat myocardial ischemia were directed towards the coronary venous system, which is free from atherosclerotic disease, most of the coronary interventions for augmenting blood flow to the ischemic myocardium are directed towards the coronary arterial system where unstable atherosclerotic lesions are found [1-4]. The coronary veins and venous microcirculation considerably outnumber the coronary arterial system; therefore, most of the myocardium can be reached by a perfusate if applied via the coronary sinus [5]. Research findings suggested that the access to and protection of areas supplied by stenotic or occluded coronary arteries is much better with retrograde delivery than with antegrade infusion [6,7]. In 1948, Beck et al. [8] reported the method of a systemic artery coronary sinus anastomosis (Beck II procedure), which included partial occlusion of the coronary sinus toward the right atrium done at a second operation. The mortality was high (29%), and the procedure was eventually abandoned. Arealis and colleagues [9] were the first to demonstrate experimentally the potential immediate beneficial effects of myocardial perfusion by anastomosing the Internal Mammary Artery (IMA) to a ligated coronary vein and few clinical short-term successes were obtained, but soon the procedure lost favour as a result of alternative arterial revascularisation techniques such as sequential grafting, coronary artery patching techniques, and extensive coronary endarterectomy [10,11,12]. The Selective Retrograde Coronary Venous Bypass (SRCVB) technique was investigated and performed clinically for patients with extensive and diffuse CAD by a number of investigators [9-17] and the results of the last extensive research by Hockberg et al. [18] were encouraging, although no significant long term conclusion could be made. This group of patients with extensive diffuse CAD represents 12% 15% of possible candidates for coronary revascularisation [13] and it may be reasonable to offer this alternative technique of coronary venous retro perfusion to the desperate patient (not amenable to endarterectomy or transplantation) or to patient in whom multiple conventional coronary artery bypass procedures have failed. We are introducing a new surgical technique of coronary retro perfusion, whereby the whole coronary venous circulation is arterialised and the coronary arteries are redirected to drain the venous blood to the right atrium “coronary switch”. This research work may answer the following: (1) If the “Coronary Switch” can protect the heart against acute multiple ischemia? (2) If there is an adequate RMBF in the switched heart? (3) If there is a change in myocardial function after the switch?
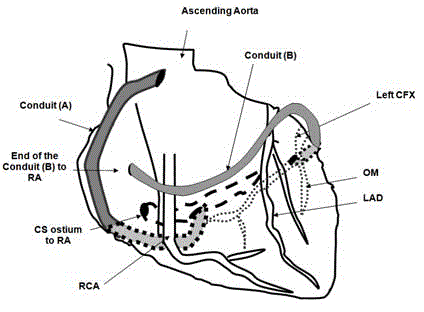
Figure 1
Figure 1
Diagrammatic representation of the Coronary Switch Procedure.
Conduit-A: Conduit taking arterial blood from the aorta to the coronary sinus;
Conduit-B: Conduit taking venous blood from the occluded coronary arteries
to the right atrium; RA: Right Atrium; Lt. CFX: Left Circumflex Coronary
Artery; OM: Obtuse Marginal branch of CFX; LAD: Left Anterior Descending
Coronary Artery; CS: Coronary Sinus
Material and Methods
Preparation
Animals were treated in accordance with the Guide to the Care
and Use of Laboratory Animals published by the US National Institute
of Health (NIH publication 85-23, revised 1985). The Institutional
Animal Care and Use Committee (IACUC) of the Medical College
of Virginia, Virginia Commonwealth University, USA; approved the
experimental protocol.
Sixteen adult Mongrel dogs (mean weight 22.5 Kg) were used
for eight experiments (n=8). Animals were anesthetized, ventilated
with Harvard respirator. Right femoral arterial and venous lines
were inserted, as well as ECG leads. Right carotid micro monometertipped
catheter (Millar, Houston, Texas) was inserted. The chest
was opened through a median sternotomy, and a pericardial cradle
was created. Another LV Millar catheter was introduced into the
LV cavity. LV dimension data were obtained with pulse transit son
micrometry (Triton Technology, San Diego, CA). One pair of 2.5 mm
diameter LTZ-piezoelectric hemispheric crystals was sewn onto the
anterior and posterior myocardial wall along the minor axis of the left
ventricle. The epicardial diameter of the left ventricle ranged from 40
mm to 66 mm. Analog data were digitized at 200 Hz and stored on the
hard disk of a personal computer.
The surgical technique
Following instrumentation, full heparinization (12,000–15,000
IU Heparin, ACT >500 Sec.), arterial cannulation through the left
common carotid artery and venous cannulation through the RA were
achieved. Once CPB was established, the aorta cross-clamped and
cardioplegia solution was delivered in the aortic root. The heart was
retracted cephalade to expose the CS. The CS ostium was closed with
5/0-proline suture just at the junction with the right atrium making
sure that none of the CS tributaries were occluded. An end to side
anastomosis was fashioned between the CS and a 3 mm Gore-Tex
tube graft using a continuous 7/0 proline suture. A similar Gore-Tex
tube graft was anastomosed end to side with the proximal left CFX
coronary artery, proximal LAD and RCA (Figure 1). The top-end of
the first conduit to the CS was anastomosed to the aorta, while the
second conduit from the coronary arteries was anastomosed to the
RA and intermittently occluded to prevent steal phenomenon.
After weaning off CPB, alternating antegrade/retrograde flow
was allowed for 30 min, and then complete occlusion of the three
coronary arteries proximal to the anastomoses was done gradually
with opening the conduit to the RA, allowing complete switch of
coronary circulation to be established (when total occlusion of
coronary arteries is done using adjustable snares). All hearts were
monitored for three and half hours off pump after full established
switch procedure; then arrested with an intracardiac injection of 80
meq KCl and harvested.
Blood flow determination
Using an established technique [19], RMBF was determined
using IMT’s NuFlowTM Colored Microspheres “CM” (Interactive
Medical Technologies, Ltd., Los Angeles, CA). Two and a half
million Microspheres were diluted in 20 ml of normal saline solution,
sonicated for 10 min and thoroughly dispersed by vortex mixing for
two minutes prior to injection through a catheter into the LA over
20 sec. Arterial reference blood samples were obtained by Harvard
withdrawal pump from the femoral artery catheter (starting 10 sec.
before injection at a constant rate of 10 ml/min for 90 seconds).
RMBF measurement was repeated five times during each experiment
starting before the procedure as a control, off CPB with alternating
Ante/Retro flow and then 60-120-180 mins after the full switch
procedure (retrograde flow only). Corrected counts obtained for each
heart sample were multiplied by the known reference withdrawal
rate and divided by the corrected reference blood sample counts to
determine sample flow expressed as ml/min/gm of tissue.
Histologic staining
Vital staining of the heart was used to demonstrate areas of
myocardial infarction “necrosis”. The territories of the three major
coronary arteries were identified and labelled. Fifteen sections were
taken from the base to the apex at 5-mm intervals and incubated for
30 min at 37°C, in freshly prepared Triphenyltetrazolium Chloride
(TTC, 1%) (Sigma Chemical Co, St. Louis, MO), and then fixed
with formaldehyde (10% in phosphate buffer) [20]. Normal healthy
myocardial tissues containing dehydrogenase enzyme are stained
brick red by TTC, but areas of infarction “necrosis” depleted of these
enzymes are not stained and appear as white or pale yellow [21].
Following vital staining, slices were examined for infarcted areas, and
then specimens (mean weight 3-4 grams) from the three layers of the
heart muscle (endomyocardium, myocardium, and epimyocardium)
were collected from each of the three major coronary artery territories
“LAD, CFX, and RCA” (Figure 2). The specimens were placed, each
in a separate tube, labelled, and sent with the reference blood for
Spectrophotometric analysis and RMBF measurement.
Measurements
Hemodynamic measurements were performed at base line
“Control” before the CPB, after off CPB with alternating ante/retro
flow, then 60 min, 120 min, and 180 min after the fully established
switch procedure. Heart performance was measured from the slope
of the relationship between Stroke-Work and End-Diastolic Length
(SW/EDL). The slope of this linear relationship is a sensitive index of
contractility, independent of load within the physiological range. This
method is more accurate than hemodynamic and load-dependent
indices of contractility such as LV arterial pressure and positive
and negative dp/dt values [22]. The arterial graft flow to the CS was
monitored as well as the Coronary Sinus Pressure (CSP). Any ECG
changes suggestive of ischemia as well as arrhythmias were recorded.
Infarcted areas were detected by vital staining. RMBF measurements
were recorded as mentioned above at five different stages of the
experiment. The RMBF for each Myocardial layer (endomyocardium,
myocardium, and epimyocardium) was calculated as the average of
all samples of the same layer in all territories. While RMBF in any
coronary artery territory, was calculated as the average of all samples
from all layers of the same territory (LAD, CFX, and RCA).
Statistical analysis
Statistical analysis was performed using the SPSS software
(SPSSR 7.5 for windows). All results are reported as Mean ± Standard
Deviation. Analysis of Variance “ANOVA” (F test) was used for
comparison of more than two groups. Significance was assigned at p
less than 0.05. Post-Hoc multiple comparison test would be used if p
value was significant.
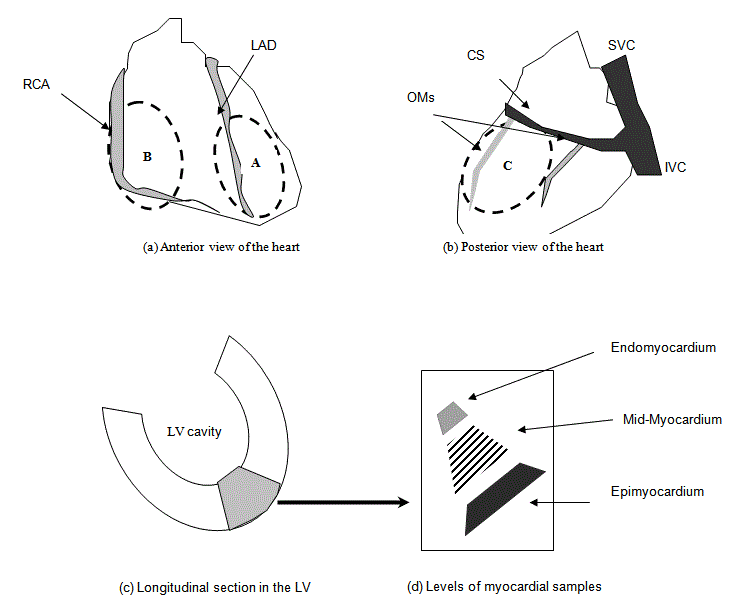
Figure 2
Figure 2
Diagrammatic illustration of myocardial sample sites.
Circles A, B, C: Represents the Biopsy Site of the Corresponding Coronary Artery; LAD: Left Anterior Descending Coronary Artery territory; CFX: Left Circumflex
Coronary Artery; RCA: Right Coronary Artery; CS: Coronary Sinus; IVC: Inferior Vena Cava; SVC: Superior Vena Cava
Results
Coronary sinus pressure and flow
The average coronary sinus pressure was 25 ± 4.6 mmHg
corresponding to an average arterial graft flow of 100 ± 14.6 ml/min
provided by a 3 mm Gore-Tex tube graft to the coronary sinus.
Left ventricular function
The slope of the relationship between stroke work and end
diastolic length (SW/EDL slope) (Table 1) was used to assess the LVF.
The average slope before the procedure “control” was 85,828.3 ± 4235
Dyn.Cm2 compared with 87,165.1 ± 3215, 91,564.4 ± 6235, 87,724.3
± 8652, and 85,484.5 ± 4352 Dyn.Cm2 at the stage of ante/retro flow,
and 60 mint, 120 mint, and 180 minutes after the switch procedure.
There was no significant statistical difference between the systolic
function before and after the switch procedure.
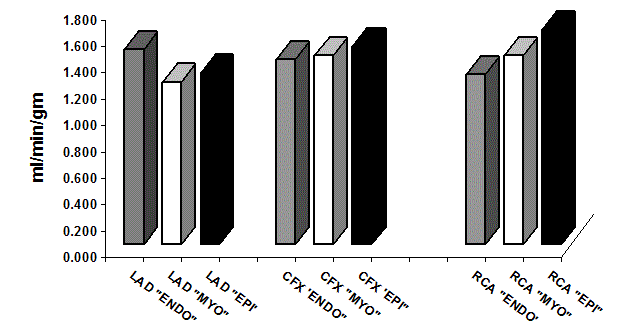
Regional myocardial blood flow “RMBF”
Regional myocardial blood flow “RMBF” was measured in the
endomyocardial, myocardial, and epimyocardial layers of the heart
muscle in the LAD, CFX, and RCA territories expressed as ml/min./
gm of tissue (Table 2 and 3, Figure 3 and 4). The average RMBF
before surgery “control” in the endomyocardial layer was 1.329 ±
0.38 compared with 1.38 ± 0.23 in ante/retrograde flow, and 1.467
± 0.34, 1.303 ± 0.12, 1.433 ± 0.25 at 60, 120, and 180 mins after the
switch. There was no significant statistical difference between the
RMBF in the endomyocardial layer before and after the switch. In
the myocardial layer, the average RMBF before surgery “control” was
1.337 ± 0.36 compared with 1.276 ± 0.34, 1.57 ± 0.34, 1.39 ± 0.23,
and 1.51 ± 0.56 at similar stages of the procedure with no significant
statistical difference. In the epimyocardial layer, the average RMBF
before surgery “control” was 1.44 ± 0.18 compared with 1.66 ±
0.41, 1.71 ± 0.11, 1.15 ± 0.22, and 1.42 ± 0.31 at similar stages of
the procedure with no significant statistical difference as well. In
general, the average RMBF in the endomyocardium was relatively
lower compared with the RMBF in the myocardium and epicardium
respectively, though statistically insignificant. Also comparing RMBF
of the three coronary artery territories (LAD, CFX, and RCA) at the
different stages of the experiment did not show a significant statistical
difference.
Myocardial infarction (Necrosis)
Using TTC vital staining, there were no detectable areas of
myocardial infarction “necrosis” following the switch procedure.
Post-bypass events
All animals were weaned off CPB easily. No inotropic support
was used. Ischemic ST segment elevation was noticed in the first 30
min to 45 min after the switch in 4 experiments (50%). The average
ST segment elevation was 1.9 0.92 mm. These ST changes disappeared
spontaneously one hour after the switch procedure.
Premature Ventricular Contractions (PVC’s) and short runs of
ventricular tachycardia were reported as well in four experiments
(50%) and were easily terminated with 25-50 mgm, intravenous
Lidocain bolus. The ventricular arrhythmias were usually detected
immediately after the switch procedure and disappeared within one
hour.
Figure 3
Figure 3
Average Regional Myocardial Blood Flow “RMBF”. In the Cardiac
Muscle Layers and Different Coronary Artery Territories (p=NS).
LAD: Left Anterior Descending Coronary Artery territory; CFX: Left Circumflex
Coronary Artery; RCA: Right Coronary Artery; “ENDO”: Endomyocardium;
“MYO”: Myocardium; “EPI”: Epimyocardium
Figure 4
Figure 4
Average Regional Myocardial Blood Flow “RMBF”. Before and
After Switch (p=NS).
PREOP: Preoperative; ANTE/RETRO: Combined Antegrade and Retrograde
Flow; Switch 60”, 120”, 180”: 60, 120, 180 minutes following switch procedure
Table 1
Table 2
Table 2
Transmural Regional Myocardial Blood Flow. Average RMBF measurements (ml/mint/gm wet weight) in the three layers of myocardial muscle in the three
coronary artery territories.
Table 3
Discussion
The rationale of this study was to provide a proof of concept of
retrograde coronary perfusion as a therapeutic modality for diffuse
CAD not amenable for CABG or PCI. Coronary Switch technique
introduces the idea of global revascularisation, by total coronary
venous retro perfusion (arterialization) in the presence of acute
ischemia involving the three major coronary arteries, a situation
similar to multiple coronary artery disease. This technique avoids the
use of external pumps or timing devices [20].
Unlike the pig heart, the canine model has relatively significant
coronary collaterals that mimic chronic ischemic hearts, which
developed collaterals. Gore-Tex tube grafts of 3 mm calibre were
used to provide a standard flow not exceeding 100 ml/min to the
CS. This flow was recommended by some investigators [11-13]
to provide an effective perfusion and avoid high CS pressure. The
second conduit draining the coronary venous effluent from the
occluded coronary arteries to the right atrium aimed at establishing
a normal physiological circulation, therefore preventing myocardial
congestion, edema, hemorrhages and late stiffness reported by some
investigators [23].
Although the mortality was high with Beck’s procedure [3,4,9],
Bakst and Bailey [23] demonstrated that retrograde perfusion of
the myocardium did occur, myocardial oxygen extraction during
retrograde perfusion was measurable, and increased intercoronary
collateral flow was present. They also demonstrated obliterative
changes in the coronary veins within few weeks of the operation, which
could be attributed to the absence of the normal venous drainage of
the heart, or longstanding combined antegrade/retrograde flow, a
technique that was avoided in the switch procedure.
From the research of Hockberg and colleagues [17,18], effective
perfusion of all layers of the myocardium was well demonstrated. They
concluded that encouraging long term patency data in animals (3 to 5
months) suggested that possible judicious use of this bypass method
might be of value in selected patients with diffuse atherosclerosis or
a previously failed coronary artery bypass. The primary data of the
switch procedure proves clearly, as well the effectiveness of retrograde
perfusion to all layers of the myocardium in different territories
supplied with occluded coronary arteries.
Some factors that reduce the efficiency of retrograde coronary
sinus perfusion are eliminated by the switch technique, such
as exclusion of some tributaries of CS by a retrograde balloon,
concomitant antegrade flow, and the absence of free coronary venous
blood drainage. Therefore, RMBF measurements in the endocardium,
myocardium, and epicardium of both antegrade “control” and
retrograde perfusion “Coronary Switch” is comparable in this study.
The presence of transient ischemic ST segment changes as well
as ventricular arrhythmias in switched hearts could be explained by
a degree of perfusion mismatch or initially low retrograde perfusion.
The absence of myocardial infarction, in addition to the preserved LV
function after the switch procedure proves the efficiency of RMBF
provided by this technique. Recently, Martin et al. [20] proved that
LV-powered coronary sinus retroinfusion “LVCSR” reduces infarct
size in acutely ischemic pigs, which was enhanced by pressure
controlled sinus occlusion “PCSO” with failure to improve RMBF
and myocardial function. Postischemic myocardial stunning was
likely responsible.
Mechanisms of salvaging ischemic myocardium by CS
retroinfusion remain to be defined. However, enhanced washout of
toxic reactive oxygen metabolites, diminished granulocyte trapping,
cellular and interstitial edema, plugging of the microcirculation,
and actual delivery of blood substrate to the ischemic myocardium
beyond an acute coronary artery occlusion have all been suggested
[5,20].
Limitations
The long-term efficacy of this retro perfusion technique in a chronic ischemic model was not investigated in this study. In summary, the primary results of coronary switch technique, demonstrated that there is an immediate protection resulting from the “Coronary Switch” in the presence of multiple acute coronary artery occlusions, an effective perfusion of all layers of the myocardium in different coronary artery territories, and preserved LV function after the procedure. Developing a chronic ischemic model, similar to chronic diffuse coronary artery disease and applying the switch procedure in a survival study is warranted.
References
- Aldea GS, Zhang X, Rivers S, Shemin RJ. Salvage of ischemic myocardium with simplified and even delayed coronary sinus retroperfusion. Ann Thorac Surg. 1996;62(1):9-15.
- Beck CS. Revascularization of the heart. Ann Surg. 1948;128:854-64.
- Beck CS, Leighninger DS. Operation for coronary artery disease. JAMA. 1951;147(8):1726-32.
- Von Ludinghausen M. Nomenclature and distribution patterns of cardiac veins in man. In: Mohl W, editor. Clinics of coronary sinus interventions. New York: Springer Verlag. 1986:13-32.
- Engelman RM, Levitsky S. A Textbook of Cardioplegia for Difficult Clinical Problems. New York; Mount Kisco. 1992:18-20.
- Pratt FH. The nutrition of the heart through the vessels of thebesius and the coronary veins. Am J Physiol. 1889;1:86-89.
- Batson OV, Bellet S. The reversal of flow in cardiac veins. Am Heart J. 1930;6:206-209.
- Beck CS, Stanton E, Batilechuk W. Revascularization of heart by graft of systemic artery into coronary sinus. J Am Med Assoc. 1948;137(5):436-42.
- Arealis EG, Volder JGR, Kolff WJ. Arterialization of the coronary vein coming from an ischemic area. Chest. 1973;63(3):462-63.
- Park SB, McGovern Gj, Liebler GA, Dixon CM, Begg FR, Fisher DL, et al. Direct selective myocardial revascularization by internal mammary artery coronary vein anastomosis. J Thorac Cardiovasc Surg. 1975;69(1):63-72.
- Benedict JS, Buhl TL, Henney RP. Cardiac veins myocardial revascularization. Ann Thorac Surg. 1975;20(5):550-57.
- Moll JW, Dziatkowiak AJ, Edelman M, e Iljin W, Ratajczyk-Pakalska E, Stengert K. Arterialization of the coronary veins in diffuse coronary atherosclerosis. J Cardiovasc Surg. 1975;16(5):520-25.
- Bates RI, Toscano M, Balderman SC, Anagnostipoulous CE. The cardiac veins and retrograde coronary venous perfusion. Ann Thorac Surg. 1977;23(1):83-90.
- Kay EB, Suzuki A. Coronary venous retroperfusion for myocardial revascularization. Ann Thorac Surg. 1975;19:327-30.
- Zajtchuk R, Heydorn WH, Miller JG, Strevey TE, Treasure RL. Revascularization of the heart through the coronary veins. Ann Thorac Surg. 1976;21(4):318-21.
- Marco JD, Hahn JW, Barner HB, Jellinek M, Blair OM, Standeven JW, et al. Coronary venous arterialization: acute hemodynamic, metabolic, and chronic anatomical observations. Ann Thorac Surg. 1977;23(5):449-54.
- Hockberg MS. Hemodynamic evaluation of selective arterialization of the coronary venous system. J Thorac Cardiovasc Surg. 1977;74(5):774-83.
- Hockberg MS, Roberts WC, Morrow AG, Austen WG. Selective arterialization of the coronary venous system. J Thorac Cardiovasc Surg. 1979;77(1):1-12.
- Kowallik P, Schulz R, Guth B, Schade A, Paffhausen W, Gross R, et al. Measurement of regional myocardial blood flow with multiple colored microspheres. Circulation. 1999;83(3):974-82.
- Martin JS, Byrne JG, Ghez OY, Sayeed-Shah U, Grachev SD, Laurence RG, et al. LV-Powered coronary sinus retroperfusion reduces infarct size in acutely ischemic pigs. Ann Thorac Surg. 2000;69:84-89.
- Franco KL, Uretzky G, Paolini D, Milton G, Cohn LH. Effects of reperfusion after acute coronary occlusion on the beating working heart compared to the arrested heart treated locally and globally with cardioplegia. J Thorac Cardiovasc Surg. 1984;87(4):561-66.
- Abd-Elfattah AS, Wechsler AS. Purines and myocardial protection. Norwell: Kluwer. 1995;65-97.
- Bakst AA, Bailey CP. Arterialization of the coronary sinus in occlusive coronary artery disease: IV. Coronary flow in dogs with aortocoronary sinus anastomosis of twelve months duration. J Thorac Surg. 1956;31:559-68.