Case Report
Distal Embolism Secondary to a Ventricular Pseudoaneurysm Associated an Abdominal Aortic Aneurysm
Mª Lourdes Del Río Solá* and Carlos Vaquero Puerta
Department of Vascular Surgery, University Hospital of Valladolid, Spain
*Corresponding author: Mª Lourdes Del Río Solá, Department of Vascular Surgery, University Hospital of Valladolid, Av Ramón Y Cajal Nº 3, 47003, Valladolid, Spain
Published: 18 Sep, 2018
Cite this article as: Lourdes Del Río Solá M, Puerta
CV. Distal Embolism Secondary
to a Ventricular Pseudoaneurysm
Associated an Abdominal Aortic
Aneurysm. Clin Surg. 2018; 3: 2111.
Abstract
Acquired pseudoaneurysm of the left ventricle is a rare disorder that usually occurs after myocardial infarction (MI) or after cardiac surgery. Rupture of the free wall of the left ventricle due to myocardial infarction (MI) occurs in almost 4% of patients with infarcts and in 23% of those dying of myocardial infarction. Left ventricular pseudoaneurysms are often asymptomatic and are discovered incidentally upon investigation of some other condition. It is extremely infrequent that their diagnosis is carried out as a result of a distal embolization in the lower extremities, as presented in the following clinical case.
Introduction
Regardless of treatment strategy, left ventricle pseudoaneurysms are associated with a high mortality rate. We report on the extremely rare occurrence of a patient presenting one episode of distal embolism in the lower extremities associated with giant post-infarction left ventricular pseudoaneurysm and abdominal aortic aneurysm [1-3]. The key to success in this case is related with the diagnosis of an undiagnosed ventricular pseudoaneurysm following acute myocardial infarction and masked by a possible embolic origin of the abdominal aortic aneurysm [4,5].
Case Presentation
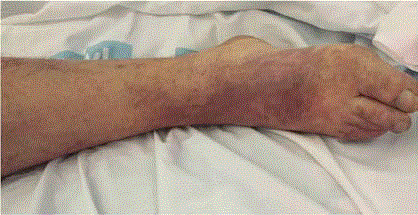
A 72-year-old patient was admitted in the Vascular Surgery Department on July 20th, 2016, after
consulting at the emergency department for presenting atheroembolic lesions on the toes of both
feet associated with resting pain in both lower extremities (Figure 1).
The patient had no known drug allergies, had stopped smoking three months ago, and had had
an acute non-Q myocardial infarction in March 2016, requiring endovascular treatment with a stent
implantation on the right circumflex and obtuse marginal coronary arteries, although there was no
possibility to revascularize the anterior descending artery by chronic obstruction with a sequela of
paroxysmal atrial fibrillation post infarction. On physical examination, cardiac auscultation was
rhythmic, with no murmurs, and pulmonary auscultation presented a preserved vesicular murmur.
In the lower limbs no edema was observed, and the femoral, popliteal and distal pulses were
preserved in both lower extremities with cyanotic lesions on both feet.
On the electrocardiogram, the rhythm was sinus, at 55 bpm, axis -30º, PR 160 msec, QRS
100 msec, negative T waves in II, III, aVF, aVR, V4 to V6, Q waves in III, aVR, aVF. The chest
X-ray showed a cardiomegaly and pulmonary hyperinflation. On the duplex-scan, the infrarenal
abdominal aorta had a maximum diameter of 4 cm; the right common iliac artery had an aneurysm
of 3.1 cm maximum diameter, the left common iliac artery 2.5 cm. And both external iliac arteries
were ectasic but not aneurysmal. At the level of the lower extremities, the right common femoral
artery had a maximum diameter of 2.4 cm, a right popliteal artery of 1.3 cm, the left common
femoral artery of 1.1 cm in diameter and a left popliteal artery of 1.6 cm maximum diameter.
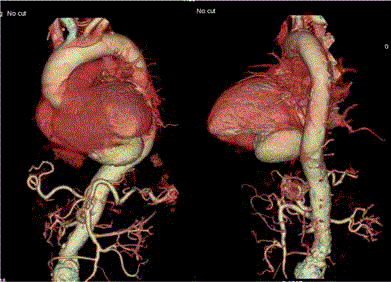
Subsequently, a computerized axial tomography was performed in which an aneurysm was seen
in the lower face of the left ventricle with dimensions of approximately 6.9 cm x 7.3 cm x 4.8 cm in
its transverse, antero-posterior and cranial-caudal diameters, respectively (Figure 2). The abdominal
aorta presented an aneurysmal dilatation of approximately 4.2 cm x 4.5 cm in its transverse and
antero-posterior diameters and iliac aneurysms with maximal diameter of the right common iliac
aneurysm of 2.9 cm and 2.5 cm in the left common iliac artery.
A transthoracic echocardiogram was completed in which there
was dilation of left cavities with normal dimensions of the right
heart cavities. A large basal inferior pseudoaneurysm was observed
with a 45 mm neck and a depth of 46 mm with diastolic expansion
that originated from the mitral annulus to the insertion of the
posteromedial papillary muscle. The systolic function was moderately
depressed (39%). Diastolic function presented severe alteration
of relaxation. The aortic valve was trivalva with moderate stenosis.
Thrombotic material was observed inside of pseudoaneurysm.
The supra-aortic arteries duplex-scan showed a stenosis of 70% at
internal carotid artery and a peak-systolic velocity of 170 cm/s.
Finally, cardiac scintigraphy showed the anterior descending
artery chronically occluded at the mean level revascularizing a distal
level by homo coronal collateral, circumflex artery without intra stent
restenosis, right coronary artery chronically occluded at the mean
level revascularizing a distal level by homo and hetero coronary
collateral.
With the diagnosis of giant inferobasal pseudoaneurysm of the
left ventricle, moderate aortic valve stenosis and ischemic heart
disease with a single revascularisable vessel, the patient underwent
Surgery on the ninth day of entry.
In the surgical intervention we observed severe pericardial
adhesions at the apex, lower and posterior ventricular left side, mild
to moderate left ventricular dysfunction, severe dilatation of the
cardiac cavities, and horizontality of the heart and the presence of
left ventricular pseudoaneurysm from the proximity of the mitral
annulus to the basal and posterior middle segments. Surgical repair
consisted of ventricular reconstruction using endoaneurismorrhaphy
with bovine pericardial patch attached to Teflon patch anchored
by simple and Teflon-supported points on the healthy muscular
zone of the left ventricular wall, in intimate relation with papillary
muscles and tendinous cords. In addition, a sandwich fold of the
pseudoaneurysm wall was performed on the patch, supported
on two Teflon strips. Subsequently, the aortic valve was replaced
by Perimount Magna prosthesis nº23 supraanular and surgical
myocardial revascularization by internal mammary artery bypass
to the anterior descending artery. After the intervention, the patient
was admitted to the cardiac reanimation unit. A new echocardiogram
revealed a hematoma that compressed the right atrium causing
cardiac tamponade that required surgical reintervention. In the
postoperative period, he required pharmacological support with
noradrenaline, dobutamine, and polytransfusion. The patient was
extubated on the Fourth postoperative day.
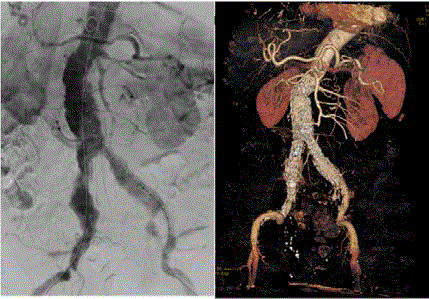
Twenty days later, the patient underwent a third surgical
intervention for endovascular repair of infrarenal aortic and
right common iliac artery aneurysms with a bilateral aortoiliac
endoprosthesis and embolization of the aneurysmal sac with coils
(Figure 3). The patient also required bilateral transmetatarsal
amputation.
At the time of discharge, the echocardiogram the mitral valve
with central insufficiency. The prosthetic aortic valve was normal
functioning. The ventricular ejection function was 55%. The right
ventricle had preserved its dynamic (TAPSE 20mm). The patient was
discharged with oral anticoagulation therapy, antiaggregation with
clopidogrel 75 mg in addition to his usual treatment.
Figure 1
Figure 2
Figure 3
Comment
Left ventricular pseudoaneurysms are characterized by a small,
narrow necked that links the ventricle with an aneurysmal sac,
which contains thrombus and blood and is surrounded by fibrous
pericardial tissue without myocardial tissue [5].
Left ventricular pseudoaneurysms are often asymptomatic and
are diagnosed in the context of another concomitant pathology,
usually cardiologic, such as congestive heart failure or ischemic heart
disease. Diagnosis can be made preoperatively by several imaging
techniques, including computed tomography, echocardiography, and
magnetic resonance imaging; however, contrast ventriculography and
coronary angiography seem to be necessary in evaluating the location
and anatomy of the aneurysm and the state of the coronary arteries.
The differential diagnosis between pseudoaneurysm and a true
aneurysm is difficult, although the presence of a narrow neck in color
flow Doppler echocardiography or ventriculography suggests the
existence of a pseudoaneurysm. The natural history of left ventricular pseudoaneurysm is not well known given its low frequency [6].
In conclusion, regardless of treatment strategy, left ventricle
pseudoaneurysms are associated with a high mortality rate. The key
to success in this case is related with the diagnosis of an undiagnosed
ventricular pseudoaneurysm following acute myocardial infarction
and masked by a possible embolic origin of the abdominal aortic
aneurysm.
References
- Pretre R, Linka A, Jenni R, Turina MI. Surgical treatment of acquired left ventricular pseudoaneurysms. Ann Thorac Surg. 2000;70(2):553-7.
- Atik FA, Navia JL, Vega PR, Gonzalez-Stawinski GV, Alster JM, Gillinov AM, et al. Surgical treatment of postinfarction left ventricular pseudoaneurysm. Ann Thorac Surg. 2007;83(2):526-31.
- Singh S, Puri A, Narain V, Sahni J. Post-traumatic left ventricular pseudoaneurysm. Interact Cardiovasc Thorac Surg. 2012;14(3):359-61.
- Nair VV, Malankar D, Kothari SS, Das S, Gulati GS, Airan B. Unusual left ventricular pseudoaneurysm in a child after disseminated bacterial infection. World J Pediatr Congenit Heart Surg. 2014;5(1):121-3.
- Davies MJ. Ischaemic ventricular aneurysms: true or false? Br Heart J. 1988;60(2):95-7.
- Komeda M, David TE. Surgical treatment of postinfarction false aneurysm of the left ventricle. J Thorac Cardiovasc Surg. 1993;106:1189-91.