Research Article
Reversible Hyponatremia and Acute Renal Failure in Cardiac Tamponade
Ales Rozman*, Vladimir Dimitric, Katja Adamic, Mateja Marc Malovrh and Julij Selb
Department of Endoscopy/Pulmonology, University Clinic of Respiratory and Allergic Diseases Golnik, Slovenia
*Corresponding author: Ales Rozman, Department of Endoscopy/Pulmonology, University Clinic of Respiratory and Allergic Diseases Golnik, Golnik 36, SI-4204 Golnik, Slovenia
Published: 14 Sep, 2018
Cite this article as: Rozman A, Dimitric V, Adamic K,
Marc Malovrh M, Selb J. Reversible
Hyponatremia and Acute Renal Failure
in Cardiac Tamponade. Clin Surg. 2018;
3: 2101.
Abstract
Background: Cardiac tamponade can be associated with hyponatremia and/or acute renal failure.
Case reports and a few case series describe conditions separately. The objective of this study was to
determine the frequencies of the reversible hyponatremia and acute renal failure in patients with
cardiac tamponade after pericardial drainage and to determine whether there is any association
between them.
Methods: In this retrospective study medical records of patients treated by pericardial drainage
between years 2010 and 2016 were reviewed. Cardiac tamponade was confirmed by cardiac chamber
compression proved by echocardiography. Sample means/medians of paired data were compared
with paired t-test (sodium concentration before and after drainage) and Wilcoxon signed-rank test
(creatinine concentration before and after drainage).
Results: Median amount of evacuated pericardial fluid was 1100 ml (rang 250 ml to 2000 ml) in 23
included patients. The mean pre-drainage sodium level was 133.1 mEq/l ± 8.5 mEq/l and the mean
post-drainage sodium level was 139.0 mEq/l ± 4.2 mEq/l (p<0.002). Among 23 patients 10 (43.5%)
had hyponatremia before drainage and 3 (13.0%) after drainage. The median pre-drainage serum
creatinine concentration was 87.5 μmol/l (range =47 - 283 μmol/l) and median post-drainage serum
creatinine concentration was 70 μmol/l (range =42 - 186 μmol/l) (p=0.014). Sodium and creatinine
concentrations were correlated in a way, that sodium concentration increases, and creatinine
concentration decreases after cardiac decompression (p<0.001).
Conclusion: Hyponatremia and acute renal failure are associated with cardiac tamponade and
recover spontaneously after pericardial drainage. Sodium concentration increase and creatinine
concentration decrease after cardiac decompression are in a correlation.
Keywords: Acute renal failure; Cardiac tamponade; Hyponatremia; Pericardial drainage; Pericardial effusion
Introduction
Cardiac tamponade is compression of the heart due to the accumulation of the fluid in the
pericardial sac and represents clinical emergency. The relationship between the amount of pericardial
fluid and pressure is not linear, thus symptoms vary and are mostly nonspecific [1].
Cardiac tamponade can be accompanied by hyponatremia as reported in several case reports and
two retrospective analyses [2-7]. Hyponatremia is frequently associated with malignant pericardial
effusion and with cardiac chambers compression [2,3]. After pericardial drainage hyponatremia
often improves spontaneously, without additional treatment [2-7]. The suggested underlying
mechanism was the increased secretion of Antidiuretic Hormone (ADH) due to decreased cardiac
output and consequential reabsorption of solute-free water in kidneys [8].
Moreover, the cardiac tamponade associated decrease in cardiac output may also be the cause
of acute renal failure. The condition was so far mentioned in a few case reports, again, with rapid
spontaneous resolution after pericardial drainage [9-14].
Reversible hyponatremia and acute renal failure may be the expressions of the same
pathophysiological mechanisms associated with reversal of decreased cardiac output in decompressed
heart, but the association has not been described yet in the medical literature. The purpose of our
retrospective study was to determine the frequencies of the reversible hyponatremia and acute renal
failure in patients with cardiac tamponade after pericardial drainage and to determine whether there
is any association between them.
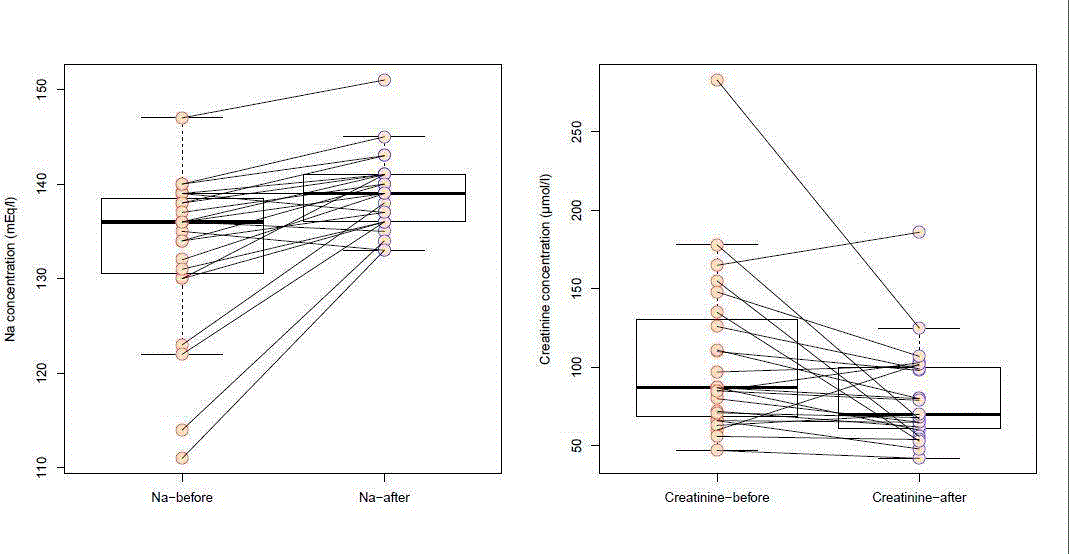
Figure 1
Figure 1
The serum concentrations of sodium (a) and creatinine (b) before and after pericardial drainage.
Methods
Patients
Patients were searched through Clinic’s electronic database to
retrieve cases by international classification of diseases (ICD-10-AM,
version 6) codes I30, I31 and I32 between years 2010 and 2016. We
searched also Clinic’s cytology database for samples of pleural effusion
during the same period. Electronic and paper charts were reviewed
for the history, vital signs and other physical findings, laboratory data,
cardiac echo findings, chest X-rays and treatment of each individual
patient. Cardiac tamponade was diagnosed by clinical presentation,
chest X-ray and cardiac echo, where besides pericardial effusion
cardiac chamber compression ought to be described (end diastolic
right atrial and/or early diastolic right ventricular invagination)
either as clearly seen or as “suspected” [15].
Patients undergoing pericardial drainage were eligible for
enrolment if serum sodium and creatinine concentrations were
measured within 2 days prior drainage and within 5 days after
drainage. Hyponatremia was defined as sodium concentration below
135 mEq/l.
Patients without complete cardiac echo data, electrolyte
measurements and those who received hyponatremia treatment
including diuretics were excluded. All pericardial drainages were
performed through percutaneous approach under ultrasound
guidance. Patients with benign diagnosis were followed up for at least
2 years. The study was performed in a single tertiary medical center,
approved by Clinic’s medical board and conducted in accordance
with the Declaration of Helsinki.
Statistical analysis
All the statistical analyses were conducted using R statistical
software (R Core Team (2017). R: A language and environment
for statistical computing. R Foundation for Statistical Computing,
Vienna, Austria, https://www.R-project.org). Data was checked for
normality using Shapiro-Wilk test. Sample means/medians of paired
data were compared with paired t-test (sodium concentration) and
Wilcoxon signed-rank test (creatinine concentration). A p-value
<0.05 was considered statistically significant.
Results
Thirty-one patients were identified in the study period. Eight
patients were excluded due to incomplete data or use of diuretics.
Twenty-three patients were included into analysis and their clinical
data are summarized in Table 1.
Median amount of evacuated pericardial fluid was 1100 ml (rang
250 ml to 2000 ml). In two patients, only a partial evacuation with
cardiac chamber decompression has been succeeded due to technical
difficulties and clogged catheter.
The mean pre-drainage sodium level was 133.1 mEq/l ± 8.5
mEq/l and the mean post-drainage sodium level was 139.0 mEq/l ±
4.2 mEq/l. The post-drainage sodium level was significantly higher
with the main increase of 6.0 mEq/l ± 6.4 mEq/l (p=0.002). Among
23 patients 10(43.5%) had hyponatremia before drainage and
3(13.0%) after drainage. Among patients with malignant pericardial
effusion 5/13(38.5%) had low sodium level before and 2/13(15.4%)
after drainage. Among patients with benign pericardial effusion
5/10(50.0%) had low sodium level before and 1/10(10.0%) after
drainage. In all groups of patients, serum sodium concentration
raised significantly after pericardial drainage (Table 2).
The median pre-drainage serum creatinine concentration 87.5
μmol/l (range =47 - 283 μmol/l) was significantly higher (p=0.014)
than the median post-drainage serum creatinine concentration 70
mol/l (range =42 - 186 mol/l).
To test the relation between the changes in sodium and
creatinine concentrations before and after pericardial drainage,
we hypothesized, that changes of two variables would fall into one
of the four categories (Crerise Narise, Crerise Nafall, Crefall Nafall and
Crefall Narise) with the probability of 0.25 if they are not correlated.
The distribution in our sample was the following: Crerise Narise = 5,
Crerise Nafall= 0, Crefall Nafall = 3 and Crefall Narise =15). The p-value for
getting a result of 15 positive hits in a sample of 23 draws, where we
sample from a binominal distribution with the 0.25 probability of a
positive draw, was <0.001. Sodium and creatinine concentrations are
therefore correlated in a way, that sodium concentration increases,
and creatinine concentration decreases after cardiac decompression
(Figure 1).
Table 1
Table 2
Discussion
Cardiac tamponade is a life-threatening condition, due to
numerous malignant and benign diseases. In the present study we
described the incidences of hyponatremia and acute renal failure,
which spontaneously recovered after cardiac decompression and the
relation between them. Although both conditions were described in
case reports and case series separately, relationship between them has
not been addressed yet [2-7,9-14].
We detected hyponatremia in 43,5% of patients with cardiac
tamponade, what was less in comparison with previously published
data, where incidences were 65% and 54.2% [2,3]. We didn’t observe
the positive relationship between malignant disease and hyponatremia
as published before, because the incidence of hyponatremia in the
group of patients with malignant disease was only 38,5% in contrast
to the group of patients with benign diseases where incidence reached
50%. However, the spontaneous recovery in patients with benign
disease was better than in patients with malignancy, which suggests
some additional mechanisms of hyponatremia in malignant disease
[16]. The increase in serum sodium concentration raised significantly
in all groups of patients after cardiac decompression. Sodium raised
even in the group of patients with baseline sodium concentration
within the reference interval.
In parallel with hyponatremia we observed a reversible
renal failure with increased creatinine concentration. Creatinine
concentration decreased significantly after pericardial drainage.
Although our sample of patients was small, a clear correlation
between hyponatremia and worsening of the renal function seemed
to exist.
Pathophysiological mechanisms running in the background
are complex, but fragments of evidence from the existing literature
enabled us to connect both manifestations of compressed hearth in a
of compensatory mechanisms of the body.
The increased pericardial pressure compresses cardiac chambers
after the volume of pericardial effusion overwhelms the pericardial
capacity to stretch. Cardiac chambers become progressively smaller,
cardiac inflow is reduced and effective circulatory volume is
consequentially decreasing [1].
The condition is detected by hypothalamus, which releases
Antidiuretic Hormone (ADH) in order to restore circulatory volume
by free water retention in the kidneys [8]. The elevated serum ADH
was detected in dogs with experimental cardiac tamponade and
declined rapidly after pericardiocentesis [17].
At the same time, mean arterial pressure and consequently
Glomerular Filtration Rate (GFR) are decreased in pericardial
tamponade [18]. Renal denervation prevented these changes in dogs,
what suggested sympathetic nerve mediated mechanism [18].
In a small case series of patients with constrictive pericarditis
increased concentrations of plasma norepinephrine, renin and
aldosterone were detected [19]. Pressures in the right atrium
and in the pulmonary vasculature were increased in parallel with
increased pulmonary and systemic vascular resistance [19]. After
pericardiectomy all measurements in all 8 patients returned toward
normal levels [19].
Retention of water in the body which manifests as edema
maintains the arterial pressure, and diuretic therapy at this point can
dangerously compromise the patient. Pericardial drainage and heart
decompression rapidly restore cardiac output and accumulated body
water is detected mainly by increased transmural atrial pressure and
consequential atrial distension, which stimulates Atrial Natriuretic
Peptide (ANP) secretion [20-22]. Although atrial pressure is high
during cardiac tamponade, transmural pressure is not elevated
because of opposing pericardial fluid pressure and ANP is not
released [18]. Massive diuretic response usually follows pericardial
drainage with the restoration of kidney function and serum sodium
concentration [5,10-12].
It is unclear, why some patients develop hyponatremia in cardiac
tamponade and the other don’t. It seems, that the effects of pericardial
fluid volume and the time course of its accumulation on cardiac
output vary in individual patients and thus also the magnitude of the
adaptive response. The assumption was confirmed by our observation,
where serum sodium concentration raised in all patients significantly
after pericardial drainage even in those, who didn’t meet the criteria
for hyponatremia.
The constellation of hyponatremia and increased serum creatinine
has important clinical consequences: diuretic treatment should not
be started, until cardiac tamponade is excluded, to avoid harmful
effects on patient’s compensatory mechanisms.
Our study had several limitations. The first is the retrospective
design with incomplete set of data in some patients that had to be
excluded from the analysis. This might include some selection bias.
Next disadvantage is relatively low number of patients, which didn’t
allow us to perform subgroup analyzes. Interpretation of cardiac
echo is, again, a subjective process to some extent, where certain
investigators might differently assess cardiac chamber compression.
Besides routine laboratory, no hormone assays were performed
before and after pericardiocentesis. And finally, pericardial pressures
or pressures in cardiac chambers and pulmonary vasculature were
not measured before and after pericardial drainage. The study was
focused on direct clinical effects of pericardial drainage and didn’t
address the underlying mechanisms which can be the object of future,
prospective trial.
Cardiac tamponade compromises effective circulatory volume
and can cause hyponatremia and acute renal failure. Both conditions
spontaneously recover after pericardial drainage, without additional
therapy. Diuretic therapy might harm patients if cardiac tamponade
was not excluded as the cause of hyponatremia and acute renal failure,
despite clinical signs of cardiac failure.
References
- Spodick DH. Acute cardiac tamponade. N Engl J Med. 2003;349(7):684-90.
- Jong BH, Wei CC, Shyu KG. Improved hyponatremia after pericardial drainage in patients suffering from cardiac tamponade. BMC Cardiovasc Disord. 2016;16:135.
- Chang FK, Lee YC, Chiu CH. Hyponatremia in patients with symptomatic pericardial effusion. J Chin Med Assoc. 2012;75(10):509-12.
- Mouallem M, Wolf I, Mindlin G, Farfel Z. Pericardial tamponade-associated hyponatremia. Am J Med Sci. 2003;325(1):51-2.
- Shafique R, Sarwar S, Wall BM, Cooke CR. Reversible hyponatremia related to pericardial tamponade. Am J Kidney Dis. 2007;50(2):336-41.
- Weekes MP, Reddi BA, Wharton S, Gazis A. Hyponatraemia associated with cardiac tamponade and chronic fluid excess. BMJ Case Rep. 2010;2010:bcr0720092113.
- Dalia T, Masoomi R, Sahu KK, Gupta K. Cardiac tamponade causing severe reversible hyponatraemia. BMJ Case Rep. 2018;2018:bcr-2017-222949.
- Groves PH, Shah AM, Hutchison SJ. Hyponatraemia secondary to an inappropriately high release of antidiuretic hormone in cardiac tamponade. Br Heart J. 1990;64(3):206-7.
- Phadke G, Whaley-Connell A, Dalal P, Markley J, Rich A. Acute Cardiac Tamponade: An Unusual Cause of Acute Renal Failure. Cardiorenal Med. 2012;2(2):83-6.
- Saklayen M, Anne VV, Lapuz M. Pericardial effusion leading to acute renal failure: two case reports and discussion of pathophysiology. Am J Kidney Dis. 2002;40(4):837-41.
- Seo JW, Kang Y, Bae EJ, Hwang K, Cho HS, Chang SH, et al. Anuric acute renal failure associated with pericardial effusion without signs of cardiac tamponade. Ren Fail. 2012;34:1040-2.
- Gluck N, Fried M, Porat R. Acute renal failure as the presenting symptom of pericardial effusion. Intern Med. 2011;50(7):719-21.
- Giljaca V, Mavrić Z, Zaputović L, Vuksanović-Mikulicić S, Mesaros-Devcić I, Maleta I, et al. Acute renal failure due to pericardial tamponade in a 60-year-old male patient. Scand J Urol Nephrol. 2009;43(6):509-11.
- Singh G, Sabath B. Over-diuresis or cardiac tamponade? An unusual case of acute kidney injury and early closure. J Community Hosp Intern Med Perspect. 2016;6(2):31357.
- Adler Y, Charron P, Imazio M, Badano L, Barón-Esquivias G, Bogaert J, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC)Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2015;36(42):2921-64.
- Onitilo AA, Kio E, Doi SA. Tumor-related hyponatremia. Clin Med Res. 2007;5(4):228-37.
- Stokhof A, Overduin L, Mol J, Rijnberk A. Effect of pericardiocentesis on circulating concentrations of atrialnatriuretic hormone andar ginine vasopressin in dogs with spontaneous pericardial effusion. Eur J Endocrinol.1994;130(4):357-60.
- Osborn JL, Lawton MT. Neurogenic antinatriuresis during development of acute cardiac tamponade. Am J Physiol. 1986;250(2 Pt 2):H195-201.
- AnandIS, Ferrari R, Kalra GS, Wahi PL, Poole-Wilson PA, Harris PC. Pathogenesis of edema in constrictive pericarditis. Circulation.1991;83(6):1880-7.
- Casale PN, Fifer MA, Graham RM, Palacios IF. Relation of atrial pressure andplasma level of atrial natriureticfactor in cardiac tamponade. Am J Cardiol. 1994;73(8):610-3.
- Svanegaard J, Thayssen P, Arendrup HK. Atrial natriureticpeptide and hemodynamic response to pericardiectomyfor chronic constrictive pericarditis. Am J Cardiol. 1990;66:117-20.
- de Groote P, Millaire A, Vantyghem MC, Dalmas S, Racadot A, Wurtz A, et al: Responseof atrial natriuretic factor to surgical pericardialdrainage in patients with chronic pericardial effusion. IntJ Cardiol.1994;46(1):15-22.