Research Article
Surgical Management of Hemorrhagic Stroke with Intra-Ventricular Extension and Acute Obstructive Hydrocephalus: 12 Months Retrospective Review
Ugwuanyi CU1*, AnigboAA1, Ogungbo B1, Solanke O2, Nwaribe EE3, Ugwu E3, Udoh LE1, Onwuka SI1 and Jamgbadi SS2
1Neurosurgery Unit, National Hospital Abuja & Wellington Neurosurgery Centre Abuja, Nigeria
2Neuroanasthesia Unit, National Hospital Abuja & Wellington Neurosurgery Centre Abuja, Nigeria
3Neuroanasthesia Unit, Federal Medical Centre Abuja & Wellington Neurosurgery Centre Abuja, Nigeria
*Corresponding author: Ugwuanyi CU, Neurosurgery Unit, National Hospital Abuja & Wellington Neurosurgery Centre Abuja, Nigeria
Published: 07 Sep, 2018
Cite this article as: Ugwuanyi CU, Anigbo AA, Ogungbo B,
Solanke O, Nwaribe EE, Ugwu E, et al.
Surgical Management of Hemorrhagic
Stroke with Intra-Ventricular Extension
and Acute Obstructive Hydrocephalus:
12 Months Retrospective Review. Clin
Surg. 2018; 3: 2095.
Abstract
Background: Some clinicians believe that the mortality/morbidity associated with Intracerebral
Hemorrhage (ICH) associated with Intracerebral Hemorrhage (IVH) and Acute Obstructive
Hydrocephalus (AOHCP) does not justify any aggressive management, others think otherwise.
Aims/Objectives: To evaluate the impact of surgical intervention on the outcome of Hemorrhagic
Stroke (HS) with IVH and AOHCP.
Methods: 12 months retrospective review of case notes who met the selection criteria. Primary
outcome measure was mortality. Secondary outcome was modified Rankin score (mRs) at discharge,
at one month and at six months.
Results: 15 patients (M: F=2.7:1) met the study criteria. Average age was 51years (age range =14-72
years). Headache and impaired consciousness were present in 100% cases. Hypertension was the
most outstanding risk factor in 93% (n=14) cases with average Mean Arteria Blood Pressure (MABP)
of 130 mmHg. Admitting brain CT scan showed ICH in 10/15 (66%) and SAH in 5/15(33%) cases.
Both were associated with IVH and AOHCP. Hypertension was responsible for 9/10 (90%) of ICH
while AVM contributed 10%. Most (77%) of the hypertensive bleeds (7/9) were deep (thalamic/basal
ganglia). Aneurysm type by location favored (anterior communicating artery aneurysm) ACOM
3/5 (60%). EVD was inserted same day of admission with an average opening pressure of 52.9 cm
H2O (38.6 mmHg) (Mean 52.9 cm H20, Std Dev=5.92 cm H20). Average duration on EVD was
6.6 days and 8/15 (63%) required conversion to VP shunts. 3/5 (60 %) of SAH patients had open
craniotomy for clipping of aneurysm while 1/5 (20%) had endovascular coiling 5/10 (50%) of the
ICH had conservative NCCU treatment but 2/10 (20%) merited craniotomy for clot evacuation.
Mortality was 8/15 (53%).
Conclusion: Mortality though still high, was comparatively reduced. None of the survivors returned
to full functional status at six months. However early active management should not be discouraged
where facilities exist.
Abbreviations
HS: Hemorrhagic Stroke; IVH: Intraventricular Hemorrhage; AOHCP: Acute Obstructive Hydrocephalus; CT/CTA: Computerized Tomography/Angiography; ICH: Intracerebral Hemorrhage; SAH: Subarachnoid Hemorrhage; EVD: External Ventricular Drain
Introduction
Hemorrhagic stroke is the most fatal form of stroke and account for approximately 20% of all strokes globally (ICH 15% and SAH 5%) [1,2]. ICH may result from poorly controlled hypertension, vascular malformations, amyloid angiopathy, etc. SAH results from ruptured aneurysm. In either case the cerebrum is abnormally exposed to direct contact with blood and its products. Often acute bleed dissects through some natural barriers in the brain into the ventricular system and cause interference with the normal physiology of Cerebrospinal Fluid (CSF). Acute obstructive hydrocephalus (HCP) frequently follows. The mass effect of the initial hematoma and the associated acute obstructive HCP inevitably cause a rapid rise in Intracranial Pressure (ICP). This results in abnormal cerebral cellular metabolism, impairment of neurotransmission and drop in Glasgow Coma Scale (GCS). Without intervention, death results from brain herniation. Reported mortality from ICH alone is 44% [1], but may be up to 80%when associated with IVH and acute HCP [2]. Aside medical management strategies in the setting of a neurocritical care unit, there are clearly defined surgical strategies in managing this condition ranging from simple external ventricular drain insertion to complex procedures for evacuation of hematomas, clipping or coiling of aneurysms and excision/embolization of vascular malformations. Whereas some clinicians believe that the mortality and morbidity associated with this condition does not justify any such aggressive management, others think otherwise. This study therefore aims to review our local experience in mobilizing active surgical management for hemorrhagic stroke with intraventricular extension and obstructive hydrocephalus.
Figure 1
Figure 2
Figure 3
Materials and Methods
In line with the ethical standards of the institutional clinical
directorate, relevant clearance was obtained to access e- records of the
target study population. This study was conducted at the Wellington
Neurosurgery Centre Abuja. Selection criteria were restricted
to hemorrhagic stroke patients who suffered intraventricular
hemorrhage, acute obstructive hydrocephalus and required surgical
intervention were selected for this study. This study was conducted
over a period of 12 months between Jan 2016 to Dec 2016. Patients
who suffered ICH secondary to clotting disorder such as liver diseases,
anticoagulants use (warfarin) were excluded from this study. Patients
who suffered ICH but with no intraventricular extension were equally
excluded. All patients had immediate non contrasted CT brain to
confirm the diagnosis. Initial resuscitation and management were
conducted in the Neurocritical care unit using the Addenbrookes
protocol [3]. The five pillars of this protocol include measures to
maintain normotension, normoxemia, normocarbia, normoglycemia
and normothermia.
Following satisfactory anesthesia review, these patients
underwent emergency insertion of external ventricular drain. Unless
otherwise dictated by the pattern of ventricular blood casting, the
right frontal horn was usually preferable for ventricular access. Initial
opening pressure was measured in cm H2O with a manometer to
confirm raised ICP. The ventricular catheter was then connected to
an external drainage system usually the Becker’s drainage system
and set at 15mmHg height to prevent over drainage. Post-operative
observation and management was continued in the neurocritical care
unit in line with already instituted Addenbrooke’s protocol. EVD
output was recorded hourly and any malfunction was detected early
and corrected. Patients who were unable to sustain spontaneous
ventilation were escalated to mechanical ventilatory support.
Similarly patients who were unable to maintain a good MABP to
drive adequate CPP were escalated to inotropic support.
All patients subsequently had CT angiography. This defined the
source of aneurysmal bleed or vascular malformation. Subsequent
treatment involved weaning patient from EVD. Those who were
unable to be weaned off EVD successfully required conversion of the
EVD to a definitive VP shunt. Definitive surgical interventions were
craniotomy for clot evacuation, craniotomy for clipping of cerebral
aneurysm, endovascular coiling of cerebral aneurysm, endovascular
embolization of AVM etc.
Patients requiring such high-tech neurovascular interventions
especially the interventional endovascular procedures were referred
to a cooperation hospital, the National Hospital for Neurology
and Neurosurgery London, United Kingdom. However follow up
continued following successful treatment and return. Other surgical
interventions such as open craniotomy for the clipping of aneurysms
and or clot evacuations have become a routine at the Wellington
Abuja.
Relevant data extracted from the e-records of the patients who
met the inclusion criteria for this study was assembled on excel spread
sheet. Study parameters of interest were demography, admitting
GCS, admitting BP, time to intervention from onset of symptoms,
opening pressure at insertion of EVD, duration of EVD, conversion
to VP shunt. Definitive surgical procedures include craniotomy for
clot evacuation, craniotomy for clipping of aneurysm, endovascular
coiling of aneurysm and endovascular embolization of AVM.
Primary Outcome measure was mortality rate. Secondary outcome
measures for survivors only were modified Rankin score (mRs) at
discharge, three months and six months. Simple analytical statistics
was employed for data processing and presentation.
Figure 4
Figure 5
Figure 6
Results
A total of 15 cases met the inclusion criteria. There were 11 males
and 4 females with M:F =2.7:1. Age ranged from 14 years to 60 years
with an average age at presentation of 51 years (SD=23.2 years). All 15
cases (100%) presented with headache and impaired consciousness
while 10/15(66.6%) presented with associated vomiting, 8/15(53.3%)
with neck pains and 4/15(26.6%) with additional seizure. The highest
recorded GCS at presentation was 14 while the lowest was 3 with an
average presenting GCS of 9. Earliest recorded presentation was six
hours while the delayed was 14 days. Average time to presentation was
three days. The most prevalent risk factor was systemic hypertension
which was recorded in 14/15(93.3%) cases. The average admitting
MABP was 130 mmHg (SD 53.4.). Other recorded risk factors were
diabetes mellitus in 5/15(33.3 %), cigarette smoking (4/15(26.6%) and
hyperlipidemia in 4/15(26.6%) cases. Admitting non-contrasted CT
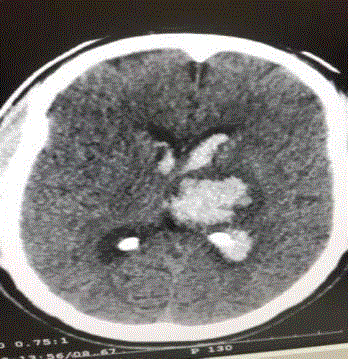
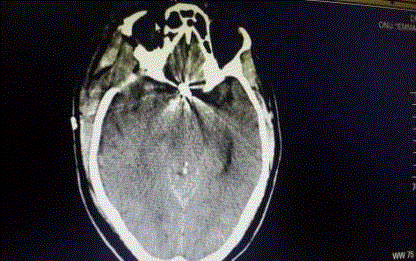
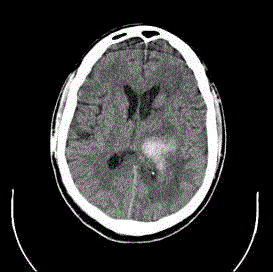
brain revealed ICH with IVH/AOHC in 10/15(66.6%) cases (Figure
1) and SAH with IVH/AOHC in 5/15(33.3 %) cases (Figure 2).
Hypertension was found to be associated with 9/10 (90%) of all ICH
while AVM was responsible for 1/10(10%). Further analysis revealed
that deep brain nuclei bleed (thalamic/basal ganglia) were observed
in 7/9 (77.7%) of the hypertensive bleeds while lobar bleeds were
observed in 2/9(22.2%) of the hypertensive bleeds. CTA of all SAH
cases revealed ACOM aneurysm in 3/5(60%) and one each of PCOM
and MCA aneurysms (20%).
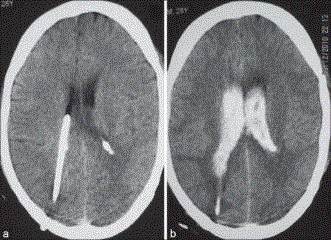
Initial operative intervention for all cases was emergency insertion
of EVD (Figure 3 and 4) on same day of admission. The EVD was
usually connected to an external drainage chamber for continuous
monitoring (Figure 5). The opening pressure was recorded upon
initial ventricular access. Lowest recorded OP was 43 cm H20 (31.6
mmHg) and highest recorded was 62 cm H20 (45.5 mmHg) with an
average OP of 52.9 cm H20 (38.6 mmHg) (SD=5.92 cm H20). This
confirmed a raised ICP in all cases well in excess of the normal (20.4
cm H20=15 mmHg). The recorded maximum duration on EVD was
14 days and shortest was one day with an average duration on EVD
of 6.6 days. It was not possible to wean 8/15 (53.3%) of the patients
off EVD and they were subsequently converted to VP shunts. Only
four out of the five aneurysmal SAH patients made it to definitive
treatment. 3/5(60%) had open craniotomy for clipping of aneurysm
while one (20%) had endovascular coiling of aneurysm. Check
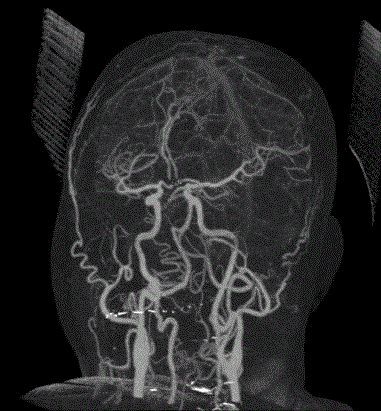
scans including skull xrays (Figure 6) plain CT (Figure 7) and CT
Angigraphy (Figure 8 and 9) were all conducted post operatively to
confirm correct placement of the aneurysm clip. Definitive treatment
of ICH involved conservative monitoring and necessary systemic
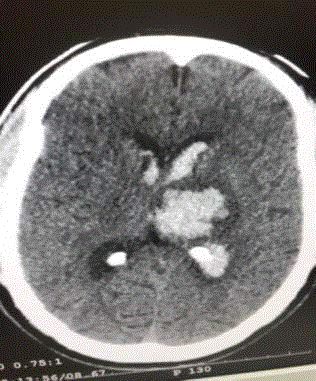
support in the NCCU in 5/10(50%) cases. Check brain scan conducted
at intervals revealed a gradual resolution of the previously noted ICH
(Figure 10 and 11). Only 2/10 (20%) cases of ICH had craniotomy
for clot evacuation while one (10%) had endovascular embolization
of AVM. Two died before any intervention. The rest 5/10(50%) were
treated non-operatively in the ICU because the location was surgically
non accessible and unattractive. This study recorded 6/10(60%)
deaths in the ICH group and 2/5(40%) deaths in the SAH group with
a combined mortality of 8/15(53.3%). Average mRs for survivors at
discharge was 3/5. It improved to 2/5 at one month follow up and to
1/5 at six months follow up.
Figure 7
Figure 8
Figure 9
Figure 10
Discussion
A total of 15 patients met the inclusion criteria during the period
under study. The number seems low but all hemorrhagic CVA
without intraventricular extension, acute obstructive hydrocephalus
and impairment of consciousness were excluded. Furthermore,
neurosurgical intervention and Neurocritical care is a very expensive
venture and only patients who met logistics of care were enrolled
for full treatment as outlined above. This was to ensure compliance
with Neurocritical care guidelines which will provide a true picture
of the behavior of these patients and generate some local evidence
for the management of similar cases. The rest were referred to public
hospitals.
Sex Ratio of 2.7:1 raises a question as to why males were more
affected. Gender differences in the incidence of ICH have been
reported in Asian and South American populations where men seem
to suffer a higher incidence [4]. Access and compliance to preventive
measures, prevalence of known risk factors such as hypertension,
alcohol consumption, cigarette smoking, sympathomimetic drug
abuse and genetic variance may explain some of the differences in
gender distribution [4]. But the gender difference was not as obvious
in another report on European populations5. It is however a general
impression those males are usually more socio-economically engaged
and therefore exposed to more stressful condition that may affect
their blood pressure.
Age distribution -The youngest recorded age was 14 years while
the oldest recorded age was 60 years with an average age of 51 years.
It is interesting to note that the youngest patient suffered ICH with
intraventricular extension secondary to a vascular malformation
which was subsequently confirmed on DSA to be draining to the
internal cerebral veins and feeding from the posterior cerebral artery.
Therefore hemorrhagic stroke in the younger age group demands
that efforts should be made to exclude vascular malformations by
deploying the necessary neuroimaging modalities. To corroborate
this fact, Toffol et al. [6] in a review of 40 young patients aged 15
years to 45 years who suffered lobar ICH, observed that 37.5% were
found to be due to AVM. Digital subtraction angiography is the gold
standard and will show AVMs and aneurysms when associated with
ICH [7], but it is not readily available in our environment. CTA is
reasonably sensitive, reliable and is very much available, safe and
accessible locally. Hypertensive bleeds and aneurysmal subarachnoid
hemorrhage was commoner in the older age groups. Some study
reported that the peak age for aneurysmal SAH was 55 years to 60
years with only 20% occurring between 15 years to 45 years [8]. From
south East Nigeria, Ohaegbulamet et al. [9] reports that the frequency
of Lobar Hemorrhage (LH) and Deep Cerebral Hemorrhage (DCH)
varied with age. LH peaked before the age of 40 while DCH peaked
at a later age (40 years to 49 years). The age distribution of different
types of ICH may suggest a higher role of other factors apart from
hypertension [10]. These include serum cholesterol levels, cigarette
smoking, substance abuse and diabetes mellitus. The individual and
collective contributions of these factors were exposed in this study
but to what extent each is contributory remain a subject of further
evaluation.
Presentation –Abrupt onset of focal neurological symptoms is
presumed to be vascular in origin until proven otherwise; however, it
is impossible to know whether symptoms are caused by ischemia or
hemorrhage on the basis of clinical characteristics alone [11]. Headache
and impairment of consciousness were recorded in all the patients
in this study. This was associated with vomiting and neck pains in
about half the cases and seizure in about a third of cases. For ICH, the
symptoms are said to be smooth in onset but rather progressive over
minutes to hours. For SAH, the symptom onset is rather dramatic,
sudden and may progress very rapidly to deep coma and death. The
SAH headache is often described as” the worst headache of a life time”.
Early neuroimaging is therefore not negotiable in defining clearly the
exact type of stroke. Deterioration of consciousness following ICH
is caused by raised ICP from hematoma expansion, cerebral edema,
emerging acute obstructive hydrocephalus, diffuse cerebral ischemia
and seizure [12]. More than 20% of patients will experience a decrease
in the Glasgow Coma Scale (GCS) of 2 or more points between the
pre-hospital Emergency Medical Services (EMS) assessment and the
initial evaluation in the Emergency Department (ED) [13]. Therefore
the risk for early neurological deterioration and the high rate of
poor long-term outcomes underscore the need for aggressive early
management [11]. In this study, we have observed that the earliest
recorded presentation was six hours while the longest was 14 days. An
average time to presentation of three days therefore is late for all forms
of stroke. According to ECASS III [14] guidelines, a CT brain ought
to have differentiated the ischemic and hemorrhagic stroke before
4.5 hrs to set the stage for appropriate treatment. That means patient
should present within the first three hours. It is rather unfortunate
that the earliest presenting patient in this series at 6 hr did not even
meet up with any acceptable timelines. Perhaps advocacy and health
awareness campaigns could improve time to early presentations
and outcome. A plain CT Brain on admission revealed ICH with
IVH/AOHC in 10/15(66.6%) cases (Figure 1) and SAH with IVH/
AOHC in 5/15(33.3%) cases (Figure 2). Hypertension was found to
be associated with 90% of all ICH while AVM was responsible for
10%. Further analysis revealed that deep cerebral bleeds (thalamic/
basal ganglia) were observed in 7/9 (77.7%) of the hypertensive bleeds
while lobar bleeds were observed in 2/9(22.2%) of the hypertensive
bleeds. Subsequent CT Angiography revealed Arterio-Venous
Malformation (AVM) in one of the ICH (10%), ACOM aneurysm in
3/5(60%) of the SAH and one each of PCOM and MCA aneurysms
(20%). Right from previous AHA/ASA guidelines it has long been
established that rapid neuroimaging with CT or MRI is recommended
to distinguish ischemic stroke from ICH (Class I; Level of Evidence:
A) [13]. In the current and updated version of AHA/ASA guidelines,
CT angiography and contrast-enhanced CT may be considered
to help identify patients at risk for hematoma expansion (Class
IIb; Level of Evidence: B), and CT angiography, CT venography,
contrast-enhanced CT, contrast-enhanced MRI, magnetic resonance
angiography, and magnetic resonance venography can be useful
to evaluate for underlying structural lesions, including vascular
malformations and tumors when there is clinical or radiological
suspicion (Class IIa; Level of Evidence: B) [13]. In line with these
recommendations, these neuroimaging investigations were deployed
timely and accordingly to all the patients studied as shown in the
results above. There was no recorded ambiguity in the definitive
diagnosis in this study; therefore the definitive management was
clearly defined on time and implemented.
The most prevalent risk factor was systemic hypertension which
was recorded in 14/15(93.3%) cases. The average admitting MABP
was 130 mmHg. Other recorded risk factors were diabetes mellitus
in 5/15 (33.3%), cigarette smoking (4/15(26.6%) and hyperlipidemia
in 4/15(26.6%) cases. Blood Pressure (BP) is frequently, and often
markedly, elevated in patients with acute ICH; these elevations in BP
are greater than that seen in patients with ischemic stroke [15,16].
The INTERACT [17] and ATACH [18] studies now represent the
best available evidence to help guide decisions about BP lowering in
ICH. Although these studies have shown that intensive BP lowering
is clinically feasible and potentially safe, the BP target, duration of
therapy, and whether such treatment improves clinical outcomes
remain unclear. However recent evidence from the AHA/ASA
guidelines recommends that in patients presenting with a systolic BP
of 150 to 220 mmHg, acute lowering of systolic BP to 140 mmHg is
probably safe (Class IIa; Level of Evidence: B) [13]. Blood pressure
management in line with the above guidelines was an integral part of
the neurocritical care management of these patients.
The opening pressure of the EVD (Figure 5) was recorded upon
initial ventricular access. Lowest recorded OP was 43 cm H20 (31.6
mmHg) and highest recorded was 62 cm H20 (45.5 mmHg) with
an average OP of 52.9 cm H20 (38.6 mmHg). This confirmed a
raised ICP in all cases well in excess of the normal (20.4 cm H20=15
mmHg). The factors probably responsible for acute hydrocephalus
include the presence of blood in the ventricular system including
the aqueduct of Sylvius, fourth ventricle, subarachnoid space which
interferes with the free flow of CSF. It was previously documented
that improvement in GCS was observed in more than 80% of patients
treated with EVD in this condition [19]. The instant improvement in
GCS recorded in most patients in this study rather demands that we
resist the temptation to rapid over drainage. Therefore, the drainage
setting was usually set at 15 mmHg and above in order to prevent
over drainage and associated complications including re-bleeding. In
line with this, it has been recommended that EVD setting should be in
the range of 15 mmHg to 25 mmHg to avoid rapid pressure reduction
and reduce the risk of rebleeding [20].
The recorded maximum duration on EVD was 14 days and
shortest was one day with an average duration on EVD of 6.6 days. In
a study by Rammos et al. [21], the mean period of external ventricular
drainage before VP shunt placement was 14.1 days (range 3 days
to 45 days) and 21% of patient in their series suffered ventriculitis.
Although there is no stern evidence on the duration of EVD, we tend
to follow the guidelines of the Great Ormond street hospital London
whereby EVD placement should be reviewed on Day 10. After this
time the entire system was removed or changed to another site in
theatre. Therefore any patient requiring further EVD is usually re sited
at another site and if a prolonged derangement of CSF circulation is
anticipated, then a VP shunt was constructed. In this study, it was not
possible to wean 8/15 (53.3%) of the patients off EVD and they were
subsequently converted to VP shunts. As useful as EVD may sound
in the foregoing discussion, some conflicting reports suggest that its
use has been associated with very high mortality [22] and morbidity
[23] rates. When an intraventricular catheter is used to monitor ICP,
CSF drainage is an effective method for lowering ICP. This can be
accomplished by intermittent drainage for short periods in response
to elevations in ICP or as continuous mode as preferred in this study.
The principal risk associated with ventriculostomy is infection. The
incidence of ventriculostomy associated bacterial meningitis varies
between 6% and 22% [22,23] even though catheter colonization is up
to 29%.
The definitive treatment of aneurysmal subarachnoid hemorrhage
traditionally aims at securing the aneurysmal neck as early as possible
in order to prevent re-bleeding. It has been reported that 15% to
20% of unsecured aneurysmal SAH re-bleed in two weeks and up to
50% re-bleed in six months [24]. The two internationally acceptable
methods of securing aneurysm are open craniotomy for clipping
and endovascular coiling with Gugliemi detachable coils [25]. In
this study, four out of the five cases of SAH made it to definitive
treatment. Whereas one opted for endovascular treatment and hence
was referred to our cooperation hospital in the UK, three successfully
underwent open craniotomy for clipping of the aneurysm here in our
hospital. There was no adherence to ISAT [25] guidelines here because
local constraints clearly favored the open craniotomy for clipping.
Majority of the ICH were deeply situated in the deep nuclei (basal
ganglia, thalamus) and hence treated non-operatively in the ICU
because this location was surgically unattractive and the available
evidence from STICH trial [26] does not favor any such intervention.
For these lesions it has been reported also that surgery fares no better
than medical management and as a matter of fact both have little
to offer in the overall prognosis [27]. However the two cases whose
ICH were located within 1cm from the supratentorial cortical surface
although with associated intraventricular hemorrhage, did benefit
from craniotomy and clot evacuation drawing some little evidence
from international STICH II trial [28]. The lone case of AVM
associated ICH was quite deep in location with a high SPETZLER
–MARTIN [29] score and this made open surgical intervention very
unattractive. Rather she benefited from endovascular embolization
which was successfully conducted in our cooperation hospital in the
UK.
Though slightly lower in the SAH group (40%) compared to the
ICH group (60%), the combined mortality rate of 53.3% recorded
in this study reflects the poor prognosis usually associated with
hemorrhagic stroke with intraventricular extension and obstructive
hydrocephalus. In comparison to previous studies by Holly et al. [2]
which reported mortality as high as 80%, the mortality in this study
though still high at 53% is much reduced. Hypertension and low
admitting GCS are independent contributors in this high mortality.
The main cause of death is cerebral herniation and is reported to occur
mainly in the first week [30]. The mass effect of hematoma, cerebral
edema, and associated CSF circulation disorder contribute to a raised
ICP which predispose to brain herniation and subsequent death.
Brain tissue repair though slow, due to a well-known concept
of plasticity was the most probable reason for the progressive
improvement in mRs over the period of observation among the
survivors in this study. However even at six months none returned to
normal neuropsychological status.
Figure 11
Conclusion
Hemorrhagic stroke is a major cause of acute brain injury and therefore is associated with high morbidity and mortality. But it is not necessarily a death sentence. With facilities in place early deployment of all necessary management strategies is recommended in order to improve the outcome of this condition in our environment.
References
- Broderick JP, Brott T, Tomsick T, Miller R, Huster G. Intra cerebral hemorrhage more than twice as common as subarachnoid hemorrhage. J Neurosurg. 1993;78(2): 188-91.
- Holly E Hinston, Daniel F Hanly, Wendy C. Zai: Curr Neurol Neurosc Rep. 2010;10(2):73.
- Helmy A, Vizcaychipi M, Gupta AK. Traumatic brain injury: intensive care management. Br J Anaesth. 2007;99 (1):32-42.
- Tanaka H, Ueda Y, Date C, Baba T, Yamashita H, Hayashi M, et al. Incidence of stroke in Shibata, Japan: 1976-1978. Stroke. 1981; 12(4):460-466.
- D'Alessandro G, Di Giovanni M, Roveyaz L. Incidence and prognosis of stroke in the Valle d'Aosta, Italy. First-year results of a community-based study. Stroke. 1992; 23(12):1712-5.
- Toffol GJ, Biller J, Adams HP. Non traumatic intracerebral hemorrhage in young adults. Arch Neurol. 1987;44(5):483-5.
- Ropper AH, Davis KR. Lobar cerebral hemorrhages: Acute clinical syndromes in 26 cases. Ann Neurol. 1980;8:141-47.
- Biller J, Toffol GJ, Kassell NF, Adams HP Jr, Beck DW. Spontaneous subarachnoid hemorrhage in young adults. Neurosurgery. 1987;21(5):664-7.
- Ezeala-Adikaibe BA, Ohaegbulam SC. Pattern and location of intracerebral hemorrhage in Enugu, South-East Nigeria: A review of 139 cases. Niger J ClinPract. 2016;19(3):332-5.
- Moon JS, Janjua N, Ahmed S, Kirmani JF, Harris-Lane P, Jacob M, et al. Prehospital neurologic deterioration in patients with intracerebral hemorrhage. Crit Care Med. 2008;36(1):172-5.
- Claude Hemphill J, Steven M Greenberg, Craig S Anderson, Kyra Becker, Bernard R Bendok, Mary Cushman, et al. Guidelines for the management of hemorrhagic stroke. Stroke. 2015;46:2032-60.
- Broderick JP, Brott TG, Tomsick T, Barsan W, Spilker J. Ultra early evaluation of intracerebral hemorrhage. J Neurosurg. 1990;72(2):195-9.
- Morgenstern LB, Hemphill JC 3rd, Anderson C, Becker K, Broderick JP, Connolly ES Jr, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2010;41(9):2108-29.
- Lansberg MG, Bluhmki E, Thijs VN. Efficacy and safety of tissue plasminogen activator 3-4.5 hours after acute ischemic stroke: A metanalysis. Stroke. 2009;40(7):2438-41.
- Qureshi AI, Ezzeddine MA, Nasar A, Suri MF, Kirmani JF, Hussein HM, et al. Prevalence of elevated blood pressure in 563,704 adult patients with stroke presenting to the ED in the United States. Am J Emerg Med. 2007;25:32-8.
- Zhang Y, Reilly KH, Tong W, Xu T, Chen J, Bazzano LA, et al. Blood pressure and clinical outcome among patients with acute stroke in Inner Mongolia, China. J Hypertens. 2008;26:1446-52.
- Anderson CS, Huang Y, Wang JG, Arima H, Neal B, Peng B, et al. Investigators. Intensive blood pressure reduction in acute cerebral haemorrhage trial (INTERACT): a randomised pilot trial. Lancet Neurol. 2008;7(5):391-9.
- Qureshi AI. Antihypertensive Treatment of Acute Cerebral Hemorrhage (ATACH): rationale and design. Neurocritical Care. 2007;6(1):56-66.
- Hasan D, Vermeulen M, Wijdicks EF, Hijdra A, van Gijn J. Management problems in acute hydrocephalus after subarachnoid hemorrhage. Stroke. 1989;20(6):747-53.
- Voldby B, Enevoldson EM. ICP changes following aneurysmal rupture. Recurrent haemorrhage. J Neurosurg. 1992;56:784-9.
- Rammos S, Klopfenstein J, Augspurger L, Wang H, Wagenbach A, Poston J, et al. Conversion of external ventricular drains to ventriculoperitoneal shunts after aneurysmal subarachnoid hemorrhage: effects of site and protein/red blood cell counts on shunt infection and malfunction. J Neurosurg. 2008;109(6):1001-4.
- Adams RE, Diringer MN. Response to external ventricular drainage in spontaneous intracerebral hemorrhage with hydrocephalus. Neurology. 1998;50(2):519-23.
- Schinkel J, Visser LG, Van Dijk JM, Voormolen JH, Kuijper EJ. Bacterial meningitis caused by the use of ventricular or lumbar cerebrospinal fluid catheters. J Neurosurg. 2005;102(2):229-34.
- Winn HR, Richardson AE, Jane JA. The long term prognosis of untreated cerebral aneurysms, the incidence of late hemorrhage in cerebral aneurysm: A 10 year evaluation of 364 patients. Ann Neurol. 1997;1(4):358-70.
- Molyneux A, Kerr R, Stratton I. International Subarachnoid Hemorrhage Aneurysm Trial (ISAT) Collaborative Group: International subarachnoid hemorrhage aneurysm trial (ISAT)of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysm: A randomized trial. Lancet. 2002;360(9342):1267-74.
- Mendelow AD, Gregson BA, Fernandes HM, Murray GD, Teasdale GM, Hope DT, et al. Early surgery versus initial conservative treatment in patients with spontaneous intracerebral hematomas in the international surgical trial in intracerebral hemorrhage (stich): A randomized trial. Lancet. 2005;365(9457):387-97.
- Batjer HH, Reisch JS, Allen BC, Plaizier LJ, Su CJ. Failure of surgery to improve outcome in putaminal hemorrhage: A prospective randomized trial. Arch Neurol. 1990;47(10):1103-6.
- Mendelow AD, Gregson BA, Rowan EN, Murray GD, Gholkar A, Mitchell PM. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet. 2013;382:397-408.
- Spetzler RF, Martin NA. A proposed grading system from for arteriovenous malformation. J Neurosurg. 1986;65(4):476-83.
- Poungvarin N, Bhoopat W, Viriyavejakul A, Rodprasert P, Buranasiri P, Sukondhabhant S. Effects of dexamethasone in primary supratentorial intracereral hemorrhage. New Engl J Med. 1987;316(20):1229-33.