Research Article
Re-Endothelialized Small Diameter Vascular Grafts Derived from Human Umbilical Arteries as Coronary Artery Substitutes
Panagiotis Mallis1,2, Aggeliki Papapanagiotou2, Eva Kassi2,3, Athanasios Velentzas4, Michalis Katsimpoulas5, Catherine Stavropoulos Giokas1 and Efstathios Michalopoulos1*
1Hellenic Cord Blood Bank, Biomedical Research Foundation Academy of Athens, Greece
2Department of Biological Chemistry, Medical School, National and Kapodistrian University of Athens, Greece
3Department of Internal Medicine, Laiko Hospital, Medical School, National and Kapodistrian University of Athens, Greece
4Department of Biology, Section of Cell Biology and Biophysics, School of Science, National and Kapodistrian University of Athens, Greece
5Center of Experimental Surgery, Biomedical Research Foundation Academy of Athens, Greece
*Corresponding author: Michalopoulos Efstathios, Hellenic Cord Blood Bank, Biomedical Research Foundation Academy of Athens, 4 Soranou Ephessiou Street, Athens, 11527, Greece
Published: 16 Aug, 2018
Cite this article as: Mallis P, Papapanagiotou A, Kassi E,
Velentzas A, Katsimpoulas M, Giokas
CS. Re-Endothelialized Small Diameter
Vascular Grafts Derived from Human
Umbilical Arteries as Coronary Artery
Substitutes. Clin Surg. 2018; 3: 2069.
Abstract
Background: Primary therapeutic strategy in Cardiovascular Disease (CVD) is the coronary
artery bypass. Damaged coronary vessels need to be replaced with small diameter vascular grafts.
Tissue engineering has focused on the production of vascular grafts, although major adverse
reactions such as graft calcification, low patency rate and lumen occlusion still exist. This study
aimed to the development of re-endothelialized small diameter vascular grafts in order to avoid the
aforementioned adverse reactions.
Methods: Human umbilical arteries were isolated and decellularized. The evaluation of the
decellularization process was assessed by histological analysis. In addition, differentiated Endothelial
Cells (ECs) derived from Wharton’s Jelly Mesenchymal Stromal Cells (WJ-MSCs) were used for the
re-endothelialization process.
Results: Our results indicated the successful decellularization of the vessels. Specifically, human
Umbilical Arteries (hUAs) were characterized by absence of cellular and nuclear materials, while
their extracellular matrix retained intact. Differentiated ECs successfully expressed CD31, VEGF-A,
VEGF-R, vWF, FLK-1 and were able to form capillary like structures when cultured in Matrigel.
Finally, the re-endothelialization of the decellularized hUAs with the differentiated ECs was
successful based on the results of indirect immunofluorescence.
Conclusion: In conclusion, the hUAs is a very promising source for the production of small
diameter vascular grafts that could be used as coronary artery substitutes.
Keywords: Human umbilical arteries; Decellularization; Vascular graft; Cardiovascular disease
Introduction
The use of small diameter vascular grafts (<6 mm) is considered the primary therapeutic strategy
for CVD. It is estimated that over of 15 million of patients are suffering from CVD, thus demanding
surgical operation [1,2]. Until now, several vessels have been used as substitutes of damaged
coronary arteries. Among them autologous vessels such as saphenous vein and fabricated synthetic
vascular grafts made of Dacron and expanded polytetrafluoroethylene (ePTFE) have been applied
in cardiovascular surgeries [2,3]. However, these vascular grafts are accompanied by major adverse
reactions, such as thrombus formation, graft calcification and occlusion. In this way, new surgical
operation is needed for their replacement. Additionally, only 20% of patients who are suffering from
CVD have suitable autologous vessels that can be used in coronary artery bypass [4].
This widely demand of suitable vascular grafts could be overpassed by the use of tissue
engineered small diameter vascular grafts [5]. Since the first established report in 1954, regarding
the development of functional vessels, major achievements have been performed towards this
direction [6]. A great number of studies have focused in successful production of small diameter
vascular grafts by lowering risk factors such as degeneration, infection
and immunogenicity [7]. On the other hand, thrombogenicity of
decellularized and fabricated vascular grafts is of major importance
and still demands considerable attention.
Decellularization approach aims to eliminate the vascular
populations such as ECs and vascular smooth muscle cells (VSMCs),
without affecting the vessel’s ultrastructure. Due to ECs loss, the
decellularized vessels are characterized by exposed collagen in their
vascular wall, thus increasing the thrombogenicity of the graft.
Several groups have tried to modify the exposed vascular wall with
the use of chemical anti-thrombogenic factors such as heparin sulfate
in order to avoid the platelet aggregation and thrombus formation
[8,9]. On the other hand, chemically anti-thrombogenic methods
reduce significantly the in vivo remodeling properties of the vessels in
a similar way as the glutaraldehyde fixed vascular grafts.
Under this scope, we proposed the re-endothelialization of the
decellularized vascular grafts, thus avoiding platelet aggregation and
thrombus formation. ECs can be isolated from various sources such
as peripheral or cord blood and arterial biopsy. However, successful
re-endothelialization demands approximately 5 × 105 cells/cm2,
indicating that these numbers are quite difficult to be achieved with
the previously mentioned approaches [10]. Most times, insufficient
numbers of ECs are obtained from patients with CVD, making their
expansion even more challenging.
For this purpose, differentiation of MSCs into ECs could be
a solution to address this issue. In this study, decellularization of
hUAs was performed according to previous described protocols
[4,5], resulting to the production of acellular vessels. Then, reendothelialization
was performed by using differentiated ECs
derived from WJ-MSCs in order to produce non thrombogenic small
diameter vascular grafts.
Materials and Methods
Isolation of hUAs
Human Umbilical Cords (hUCs), used for the isolation of
hUAs, were derived from end term normal and caesarian gestations
(gestational ages 38-40 weeks). The isolation of the hUCs was
performed by experienced midwives and each specimen was
accompanied by signed informed consent from the mothers. The
informed consent was in accordance to declaration of Helsinki and
the ethical standards of Greek National Ethical Committee. Delivery
of the hUCs was performed within 24 hr after the gestation followed
by immediate isolation of the hUAs. Briefly, the hUCs were rinsed
in phosphate buffer saline 1x (PBS 1x, Sigma-Aldrich, Darmstadt,
Germany) for removal of excess blood and blood clots. Then, isolation
of hUAs was performed with the use of sterile instruments, while the
human umbilical vein and Wharton’s Jelly tissue were discarded.
Finally, the hUAs were placed in PBS 1x and kept at 4°C until further
use.
Decellularization of hUAs
HUAs (n=10) were decellularized according to a previous described
protocol with some modifications [4,5]. The decellularization protocol
involved the incubation of hUAs in CHAPS buffer (8 mM CHAPS, 1
M NaCl and 25 mM EDTA in PBS 1x, Sigma-Aldrich, Darmstadt,
Germany) for 22 hr (h) accompanied by rotational agitation. Then,
hUAs were briefly rinsed in PBS 1x for 5 min. The hUAs were further
incubated in SDS buffer (1.8 mM SDS, 1 M NaCl and 25 mM EDTA
in PBS 1x, Sigma-Aldrich, Darmstadt, Germany) for another 22 hr
with rotational agitation, followed by briefly rinses in PBS 1x. Finally,
hUAs were placed in a-Minimum Essentials Medium (a-MEM,
Sigma-Aldrich, Darmstadt, Germany) supplemented with 40% v/v of
Fetal Bovine Serum (FBS, Sigma-Aldrich, Darmstadt, Germany) and
incubated at 37°C for 48 hr.
Histological analysis of hUAs
Evaluation of the decellularized hUAs was performed by
histological analysis. Non-decellularized (n=10) and decellularized
hUAs (n=10) were fixed in 10% v/v formalin (Sigma-Aldrich,
Darmstadt, Germany), paraffin embedded and sectioned at 5 μm.
Histological analysis involved the use of hematoxylin and eosin (H
& E, Sigma-Aldrich, Darmstadt, Germany), for determination of cell
nuclei and Extracellular Matrix (ECM) components. Images were
acquired in DM L2 light microscope (Leica Microsystems Wetzlar
Germany). Indirect immunofluorescence against fibronectin was
performed in non-decellularized (n=10) and decellularized hUAs
(n=10). Briefly, the slides were deparaffinized, rehydrated and
blocked. Then, monoclonal antibody against human fibronectin
(1:2000, Sigma-Aldrich, Darmstadt, Germany) was added, followed
by secondary FITC-conjugated mouse IgG antibody (1:100, Sigma-
Aldrich, Darmstadt, Germany). Finally, the slides were mounted with
glycerol (Sigma-Aldrich, Darmstadt, Germany) and images were
acquired with LEICA SP5 II microscope equipped with LAS Suite v2
software (Leica, Microsystems, Wetzlar, Germany).
Biochemical analysis of the hUAs
Non-decellularized (n=10) and decellularized hUAs (n=10) were
evaluated for their collagen, sulphated Glycosaminoglycans (sGAGs)
and DNA content. Total collagen amount was measured based on
the quantification of the hydroxyproline content. For this purpose,
the Hydroxyproline Assay kit (MAK008, Sigma-Aldrich, Darmstadt,
Germany) was used according to manufacturer’s instructions.
Further biochemical analysis involved the quantification of sGAG
content. Non-decellularized (n=10) and decellularized (n=10) hUAs
were digested in 125 μg/ml papain buffer (Sigma-Aldrich) at 60°C
for 12 hrs, followed by addition of dimethylene blue (Sigma-Aldrich).
The sGAG content was quantified photometrically at 525 nm. Final
concentration of sGAG content was obtained through interpolation
to standard curve. Chondroitin sulfate standards of 12 μg/ml, 25 μg/
ml, 50 μg/ml, 100 μg/ml and 150 μg/ml were used for the development
of the standard curve.
The amount of DNA was measured in non-decellularized (n=10)
and decellularized (n=10) hUAs. All samples were digested in a
lysis buffer (0.1 M Tris pH 8, 0.2 M NaCl, 5 mM EDTA in PBS 1x,
Sigma-Aldrich, Darmstadt, Germany) supplemented with 30 mg/ml
Proteinase K (Sigma-Aldrich, Darmstadt, Germany). The digestion
was performed at 56°C for 12 hrs and then inactivation of the enzyme
was followed at 90°C for 5 min. Total DNA content was isolated,
eluted in 100 μl of DNAse free water (Sigma-Aldrich, Darmstadt,
Germany) and quantified photometrically at 260 nm to 280 nm.
ECs differentiation protocol
WJ-MSCs were submitted to differentiation into ECs with the use
of VEGF-A. Briefly, WJ-MSCs in a number of 2 × 105 were seeded in
6-well plates (Sigma-Aldrich, Darmstadt, Germany). The following
day, EC differentiation medium consisted of a-MEM with 5% v/v FBS
and 100 ng/μl of Vascular Endothelial Growth Factor-A (VEGF-A,
Sigma-Aldrich, Darmstadt, Germany) was added in the culture. The
EC differentiation medium was changed bi-weekly for a time period
of 30 days.
Characterization of differentiated ECs
Gene expression of ECs differentiated from WJ-MSCs was
performed by Reverse Transcription (RT)-PCR, PCR and gel
electrophoresis. Briefly, total mRNA was extracted using the
TRI-reagent (Sigma-Aldrich, Darmstadt, Germany) according to
manufacturer’s instructions. The quantity and quality of mRNA was
analyzed photometrically. Then, 800 ng of RNA was transcribed into
cDNA with the use of Omniscript RT kit (Qiagen, Hilden, Germany).
Then PCR was performed with Omniscript PCR kit (Qiagen, Hilden,
Germany), using specific primers in order to evaluate the gene
expression of differentiated ECs (Table 1). All PCR products were
analyzed by electrophoresis on 1% w/v agarose gel (Sigma-Aldrich,
Darmstadt, Germany). The gene expression of differentiated ECs was
compared with undifferentiated WJ-MSCs (negative control group).
Flow cytometric analysis of ECs
Differentiated ECs were analyzed for cell surface antigens by flow
cytometry. The ECs samples (n=5) were tested for the expression
of CD31 and CD29. More specifically, the cells were labeled with
phycoerythrin conjugated anti-CD31 (Beckman Coulter, Marseille,
France) and CD29 (Beckman Coulter, Marseille, France). The
phenotypes were analyzed in Cytomics FC 500 (Beckman Coulter,
Marseille, France) flow cytometer accompanied with CXP Analysis
software (Beckman Coulter, Marseille, France). CD marker expression
was compared with undifferentiated WJ-MSCs.
Capillary tube formation assay
The capillary tube formation assay was performed in order to
evaluate the ability of ECs to form vascular networks. Specifically,
Matrigel© (Becton Dickinson, Heidelberg, Germany) was initially
thawed on ice overnight according to manufacturer’s instructions.
Then, 30 μl of the Matrigel© was spread over each well of 24-well plate,
followed by incubation for 30 min at 37°C. Differentiated ECs in a
number of 3 × 104 cells were seeded in triplicate into each well and 500
μl of culture medium was added in each sample. The culture medium
for this assay consisted of α-ΜΕΜ supplemented with Endothelial
Growth Medium-2 (EGM-2, Sigma-Aldrich). Finally, incubation
of the samples was performed at 37°C, 5% CO2. The formation of
vascular networks by differentiated ECs was evaluated after 6 hr of
the initial culture. As negative control group, non differentiated WJMSCs
were used. Images of the vascular networks were acquired with
inverted DM L2 light microscope (Leica, Microsystems, Wetzlar
Germany). Total vascular network length was measured in 5 random
areas in each sample using the Image Pro Plus v6 (Media Cybernetics,
Washington, USA).
Re-endothelialization of the decellularized hUAs
Segments (n=5) of decellularized hUAs in length of 1 cm were
re-endothelialized under static seeding conditions. For the reendothelialization
experiments, initially the decellularized hUAs
were placed in 6-well plates. Differentiated ECs at a density of 3 ×
105 cells/cm2 were seeded into the vessel’s lumen. Finally, 1 ml of
culture medium consisted of a-MEM supplemented with EGM-2, 1%
v/v Penicillin-Streptomycin and 1% v/v L-glutamine (Sigma-Aldrich,
Darmstadt, Germany) was added in each well and the cultures were
incubated at 37°C and 5% CO2 for a time period of 4 weeks.
Evaluation of the re-endothelialization assay was performed
by histological analysis. The segments of hUAs were fixed in
10% v/v formalin buffer (Sigma-Aldrich, Darmstadt, Germany),
paraffin embedded and sectioned at 5 μm. Determination, of ECs
in vessel’s lumen was performed with H & E stain. In addition,
indirect immunofluorescence against fibronectin in combination
with DAPI (Sigma-Aldrich, Darmstadt, Germany) staining was
applied. The slides were deparaffinized, rehydrated and incubated
with monoclonal antibody against human fibronectin (1:2000,
Sigma-Aldrich, Darmstadt, Germany) followed by secondary FITC
conjugated mouse IgG antibody (1:100, Sigma-Aldrich, Darmstadt,
Germany). Finally, the DAPI stain was applied in order to detect
the cell nuclei. Images were acquired with LEICA SP5 II microscope
equipped with LAS Suite v2 software (Leica, Microsystems, Wetzlar,
Germany).
Scanning electron microscopy
Decellularized and re-endothelialized hUAs (n=5, l=5 mm) were
processed for scanning electron microscopy (SEM). Specifically, the
samples were fixed with 1% v/v glutaraldehyde solution (Sigma-
Aldrich, Darmstadt, Germany) for 30 min, followed by briefly
rinses in distilled water. The samples were dehydrated by 10 min
submission in 70% v/v, 80% v/v, 95% v/v aqueous ethanol, absolute
ethanol and finally placed in hexamethyldisilazane solution (Sigma-
Aldrich, Darmstadt, Germany) for 10 min. The samples were air
dried and sputter-coated with gold (Cressington Sputter, Coater 108
auto, Watford, United Kingdom) and examined with Phillips XL-30
scanning electron microscope (Phillips, FEI, Hillsboro, OR, USA).
Statistical analysis
GraphPad Prism v 6.01 (GraphPad Software, San Diego, CA,
USA) was used for the statistical analysis. Mann Whitney test was
applied in order to compare the DNA, collagen and sGAG content
between non-decellularized and decellularized samples. Statistical
significant difference was considered when p-value was less than 0.05.
Indicated values are mean ± standard deviation.
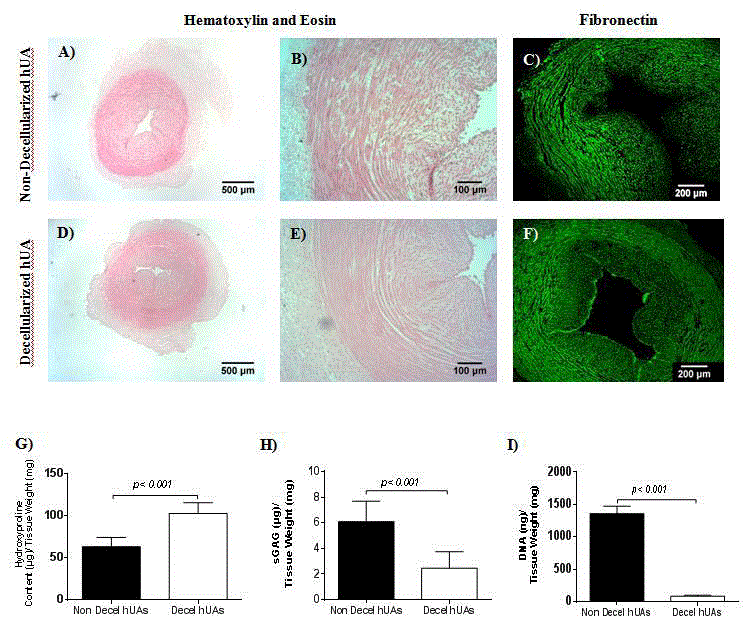
Figure 1
Figure 1
Evaluation of ECM composition in decellularized hUAs. Non-decellularized hUA with H&E (A,B) and indirect immunofluorescence against fibronectin (C).
Decellularized hUA with H&E and indirect immunofluorescence against fibronectin (F). Images A and D, original magnification 2.5x, scale bars 500 μm. Images B
and E, original magnification 10x, scale bars 100 μm. Images E and F, original magnification 5x, scale bars 200μm. Comparison in hydroxyproline (G), sGAG (H)
and DNA (I) content between non-decellularized and decellularized hUAs. Statistical significant differences between non-decellularized and decellularized hUAs
observed in all quantifications (p<0.001).
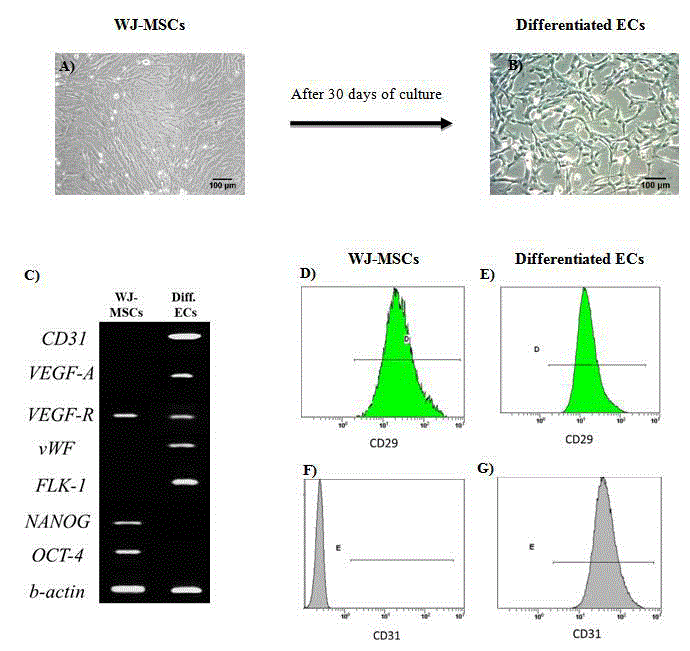
Figure 2
Figure 2
Evaluation of ECs differentiation protocol. Morphology of WJ-MSCs (A) and differentiated ECs (B) after 30 days of culture. Gene expression profile in
WJ-MSCs and differentiated ECs (C). Immunophenotypic characterization of WJ-MSCs and differentiated ECs for CD29 (D, E) and CD31 (F,G), respectively.
Table 1
Results
Histological analysis of decellularized hUAs
HUAs were successfully decellularized with the current
decellularization protocol. Specifically, decellularized hUAs retained
their initial ultrastructure, while no cellular or nuclei materials
were evident, as shown by H & E staining (Figure 1). Indirect
immunofluorescence results indicated the preservation of fibronectin
in decellularized hUAs, while no damage in the vascular wall was
observed (Figure 1).
Biochemical analysis
Hydroxyproline, sGAG and DNA quantifications were performed
in order to evaluate thoroughly the impact of decellularization process
in the vessel’s ECM. Non-decellularized hUAs characterized by 63
± 10 μg hydroxyproline/mg of tissue weight, while decellularized
hUAs characterized by 102 ± 11 hydroxyproline/mg of tissue weight.
Furthermore, sGAG content of non-decellularized and decellularized
hUAs was 6 ± 2 and 2 ± 1 μg sGAG/mg of tissue weight, respectively
(Figure 1). DNA amount in non-decellularized hUAs was 1341 ±
102 ng DNA/mg tissue weight, whereas in decellularized hUAs was
82 ± 14 ng DNA/mg tissue weight (Figure 1). Statistical significant
difference between non-decellularized and decellularized vessels was
observed in all biochemical quantifications (p<0.001), used in this
study.
Characterization of differentiated ECs
Differentiated ECs were characterized by different shape
geometry when compared to WJ-MSCs. Specifically; ECs were
smaller in size and were characterized by cobblestone shape, whereas
WJ-MSCs had fibroblastic like morphology (Figure 2). After 30
days of differentiation, ECs successfully expressed CD31, VEGF-A,
VEGF-R, vWF, FLK-1, while totally lost the expression of OCT4 and
NANOG. On the other hand, WJ-MSCs were able to express only
VEGF-R, and the key pluripotency genes OCT4 and NANOG (Figure
2). Furthermore, based on the flow cytometric analysis, unlike to WJMSCs,
differentiated ECs expressed CD31. Specifically, the expression
of CD29 for WJ-MSCs and differentiated ECs was 96 ± 4% and 95 ±
3%, respectively. In addition, the expression of CD31 in differentiated
ECs was 94 ± 5%, while WJ-MSCs didn’t express this protein marker
(Figure 2).
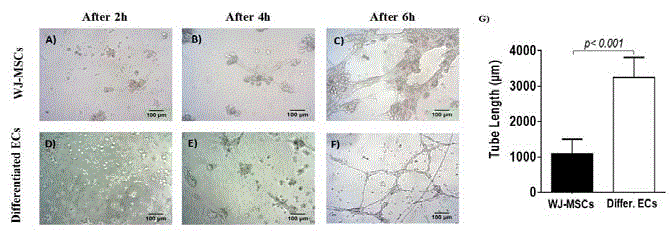
Evaluation of capillary tube formation
Differentiated ECs showed a substantial formation of capillary
tube structures after 6 hr of cultivation on Matrigel©. WJ-MSCs
were able to form a kind of vessel structure but the majority of them
failed to produce capillary tubes. On the other hand, differentiated
ECs started to form the first capillary tubes after 4 hr of culture in
the semisolid medium (Figure 3). After 6 hr, the tube length of the
capillaries was greater in differentiated ECs when compared to WJMSCs
(Figure 3).
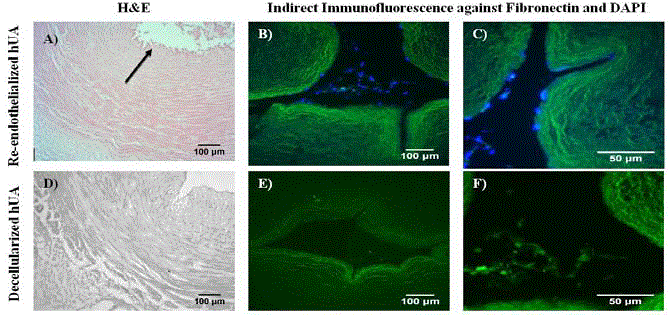
Evaluation of re-endothelialized hUAs
Histological analysis showed the successful re-endothelialization
of decellularized hUAs. Uniform distribution of ECs differentiated
from WJ-MSCs was observed in lumen of hUAs, whereas
decellularized hUAs were characterized by totally absence of cellular
and nuclear materials. Moreover, ECs were found to be in close
proximity with the fibronectin of the vascular wall as shown by
indirect immunofluorescence results.
Moreover, the presence of ECs was further confirmed by SEM
images. The ECs were attached successfully to the vessel’s lumen
(Figure 5). On the other hand, only the ultrastructure of the vessel
was evident in decellularized hUAs, without the presence of any
cellular populations (Figure 5).
Figure 3
Figure 3
Evaluation of capillary tube formation by differentiated ECs. In vitro capillary tube formation by WJ-MSCs and differentiated ECs after 2h (A,D), 4h (B,E)
and 6h (C,F), respectively. Original magnification 10x, scale bars 100 μm. G) Tube length measurement in WJ-MSCs and differentiated ECs. Statistical significant
difference was observed in tube length between WJ-MSCs and differentiated ECs (p<0.001).
Figure 4
Figure 4
Histological analysis of re-endothelialized hUAs. Re-endothelialized hUA with H&E (A) and indirect immunofluorescence against fibronectin in combination
with DAPI stain (B,C). Black arrow in image A indicated the presence of ECs in the vessel’s lumen. Decellularized hUA (negative control group) with H&E (D) and
indirect immunofluorescence against fibronectin in combination with DAPI stain (E, F). Images A,B,D and E, original magnification 10x, scale bars 100 μm. Ιmages
C and F, original magnification 40x, scale bars 50 μm.
Discussion
Development of small diameter vascular grafts (<6 mm) utilizing
the tissue engineering approaches including tissue decellularization
and vessel fabrication, is a rapidly evolving field. These grafts can
be used as substitutes in order to replace damaged vessels such the
coronary artery [11]. However, the majority of these substitutes
lack of a well-organized endothelium. The use of ECs derived from
a vascular biopsy, is a challenging process. Endothelium formation
requires a great number of cells that can be efficiently expanded and
seeded in vitro onto vascular scaffolds. Major achievements have been
performed towards this direction, involving the isolation of ECs from
various sources such as peripheral or cord blood and human umbilical
vein [12,13]. However, many studies have controversial results, while
the proper seeding of ECs is still a demanding process [12,13].
This aim of this study was to establish and produce reendothelialized
small diameter vascular grafts derived from human
umbilical arteries. For this purpose, hUAs were initially decellularized
with protocols that utilizes CHAPS and SDS reagents. Histological
analysis with H & E, showed the successful decellularization of the
hUAs, where no cellular or nuclear materials were evident, while at
the same time the arterial ultrastructure was preserved. In addition,
these results were further confirmed by DNA quantification, where
decellularized hUAs were characterized by a low DNA amount.
Moreover, fibronectin, an abundant structural protein of the
vascular wall, was retained in decellularized hUAs as indicated by the
indirect immunofluorescence results. Similar observations regarding
the preservation of structural proteins in decellularized matrices have
been also reported by other groups [4,5,14].
Further evaluation of the decellularization process, involved
the biochemical quantification of collagen and sGAGs, which are
key elements of the vascular wall. In our study, the hydroxyproline
concentration which corresponds approximately to collagen content
of the hUAs, was increased in decellularized samples. This elevation
in collagen content might be attributed by the loss of cell mass after
the decellularization process, leaving only the tissue scaffold. On the
other hand, sGAG content was decreased after the decellularization
process. It is known, that SDS reagent is an anionic detergent which
distracts the non-covalent interactions in proteins, causing in this
way the denaturation of proteoglycans and sGAGs [14,15].
Next step of this study was the establishment of the EC
differentiation process. WJ-MSCs were submitted to differentiation
into ECs by using the VEGF-A as a key protein inducer. Differentiated
ECs were characterized by elevated expression of CD31, VEGF-A,
VEGFR, FLK-1 and vWF while didn’t express OCT4 and NANOG.
Furthermore, differentiated ECs were characterized by increased
levels of CD31 and CD29 markers as indicated by flow cytometric
analysis and were capable to produce tube like structure that was
stable for at least 6 hours. The ability of MSCs to differentiate into
ECs is very promising. Taking into consideration, that patients with
CVD don’t have suitable vessels for ECs isolation, the differentiation
of MSCs could be a solution. Ideally, MSCs can be isolated from bone
marrow or adipose tissue of those patients, and then submitted into
EC differentiation in order to be used in the re-endothelialization of
the vascular grafts.
Once we have established the decellularization protocol in
hUAs and the proper differentiation of WJ-MSCs into ECs, then we
proceeded in the re-endothelialization of the vessels. Differentiated
ECs were capable to be attached in the decellularized vascular wall
as shown by the histological results. Indirect immunofluorescence
confirmed the presence of ECs into the vessel’s lumen, and further
showed their connection with the fibronectin of decellularized hUAs.
Fibronectin, along with collagens, is a key structural element of the
vascular wall, which provides attachment sites for the ECs through its
interaction with α5β1 and α4β1 [16]. In addition, it has been reported
a possible synergism between integrin interaction, fibronectin and
growth factors receptors. ECs are attached in the CS5 domain of
fibronectin through collaboration of α5β1 and α4β1, which results to
activation of focal adhesion kinase and extracellular regulated kinase
(ErK) [16,17]. Prolonged activation of ErK leads to EC proliferation
and migration, thus contributing in the endothelium formation
[16]. Furthermore, the association between VEGF-R and fibronectin
interaction, contributes even more in promoting biological responses
by ECs, such as vessel homeostasis.
Tissue engineered vascular grafts must be characterized by
a proper endothelium, avoiding neointima formation and graft
occlusion. Until now, the re-endothelialization process of vessels
needs considerable attention. A great number of research groups have
focused on improving the re-endothelialization process by treating
the vascular grafts with chemical compounds [7-9,18]. Polyelectrolyte
multilayer development and immobilization of heparin sulfates in
decellularized vessels have been proposed by modifying the amino
acids of the vascular wall [8,9]. However, such modifications have
negative impact in the vessel functionality. In this way, preventing
the capability of decellularized vessel to be remodeled in vivo reduces
significantly its function, making it susceptible for calcification
development, reduction in patency rate and graft rejection. Especially,
when these vascular grafts are intended to be used in pediatric
patients, the biological properties of the vessel must not be hampered.
Unlike, to these approaches, in our study was shown that
differentiated ECs could successfully be seeded onto decellularized
hUAs without treating them with any chemical compounds.
Figure 5
Figure 5
SEM analysis of the re-endothelialized hUAs. Re-endothelialized hUAs, where the seeded cells can be observed clearly (A,C). Decellularized hUAs,
where only the vessel’s ECM is visible (B, D). No evidence of cellular populations was observed in decellularized hUAs. Images A and B, original magnification
1000x, scale bars 100μm. Images C and D, original magnification 2000x, scale bars 50 μm.
Conclusion
In conclusion, our study focused on the development of a small diameter vascular graft derived from hUAs and its proper endothelialization. The decellularized hUAs could be used as an alternative source for the development of small diameter vascular grafts that could be used as coronary artery substitutes. In addition, these vessels could be used as a model for better understanding the molecular mechanisms of EC differentiation, proliferation and migration in the vascular wall in order to take full advantage of its clinical potential. Future research is needed for the development of biocompatible tissue substitutes derived from decellularized tissues that can potentially be used in clinical and translational medicine.
References
- Mathers CD, Loncar D. Projections of global mortality and burden of disease from 2002 to 2030. PLoS Med. 2006;3(11):e442.
- Klinkert P, Post PN, Breslau PJ, van Bockel JH. Saphenous vein versus PTFE for above-knee femoropopliteal bypass. A review of the literature. Eur J Vasc Endovasc Surg. 2004;27(4):357-62.
- Stephen M, Loewenthal J, Little JM, May J, Sheil AG. Autogenous veins and velour dacron in femoropopliteal arterial bypass. Surgery. 1977;81(3):314-8.
- Gui L, Muto A, Chan SA, Breuer CK, Niklason LE. Development of Decelluarized Human Umbilical Arteries as Small-Diameter Vascular Grafts. Tissue Eng Part A. 2009;15(9):2665-75.
- Mallis P, Gontika I, Poulogiannopoulos T, Zoidakis J, Vlahou A, Michalopoulos E, et al. Evaluation of decellularization in umbilical cord artery. Transplant Proc. 2014;46(9):3232-9.
- Blakemore AH, Voorhees AB Jr. The use of tubes constructed from vinyon "N" cloth in bridging arterial defects-experimental and clinical. Ann Surge. 1954;140(3):324-33.
- Kerdjoudj H, Moby V, Berthelemy N, Gentils M, Boura C, Bordenave L, et al. The ideal small arterial substitute: Role of cell seeding and tissue engineering. Clin Hemorheol Microcirc. 2007;37(1-2):89-98.
- Kerdjoudj H, Berthelemy N, Rinckenbach S, Kearney-Schwartz A, Montagne K, Schaaf P, et al. Small vessel replacement by human umbilical arteries with polyelectrolyte film-treated arteries: in vivo behavior. J Am Coll Cardiol. 2008;52(19):1589-97.
- Dimitrievska S, Cai C, Weyers A, Balestrini JL, Lin T, Sundaram S, et al. Click-coated, heparinized, decellularized vascular grafts. Acta Biomater. 2015;13:177-87.
- Kim DH, Heo SJ, Kang YG, Shin JW, Park SH, Shin JW. Shear stress and circumferential stretch by pulsatile flow direct vascular endothelial lineage commitment of mesenchymal stem cells in engineered blood vessels. J Mater Sci Mater Med. 2016;27(3):60.
- Weinberg CB, Bell E. A blood vessel model constructed from collagen and cultured vascular cells. Science. 1986;231(4736):397-400.
- Mead LE, Prater D, Yoder MC, Ingram DA. Isolation and characterization of endothelial progenitor cells from human blood. Curr Protoc Stem Cell Biol. 2008;Chapter 2:Unit 2C.
- Shin JW, Lee DW, Kim MJ, Song KS, Kim HS, Kim HO. Isolation of endothelial progenitor cells from cord blood and induction of differentiation by ex vivo expansion. Yonsei Med J. 2005;46(2):260-7.
- Luo J, Korossis SA, Wilshaw SP, Jennings LM, Fisher J, Ingham E. Development and characterization of acellular porcine pulmonary valve scaffolds for tissue engineering. Tissue Eng Part A. 2014;20(21-22):2963-74.
- Gilbert TW, Sellaro TL, Badylak SF. Decellularization of tissues and organs. Biomaterials. 2006;27(19):3675-83.
- Wijelath ES, Rahman S, Murray J, Patel Y, Savidge G, Sobel M. Fibronectin promotes VEGF-induced CD34 cell differentiation into endothelial cells. J Vasc Surg. 2004;39(3):655-60.
- Heilshorn SC, DiZio KA, Welsh ER, Tirrell DA. Endothelial cell adhesion to the fibronectin CS5 domain in artificial extracellular matrix proteins. Biomaterials. 2003;24(23):4245-52.
- Hoshi RA, Van Lith R, Jen MC, Allen JB, Lapidos KA, Ameer G. The blood and vascular cell compatibility of heparin-modified ePTFE vascular grafts. Biomaterials. 2013;34(1):30-41.