Research Article
Impact of Postoperative Complications on the Long-Term Outcome of Colorectal Cancer Patients who Underwent Curative Resection
Jen-Hsien Huang1,2, Chun-Chi Lin1,2, Hung-Hsin Lin1,2, Yuan-Tsiu Lan1,2, Shin-Ching Chang1,2, Huann-Sheng Wang1,2, Wei-Shone Chen1,2, Tzu-Chen Lin1,2, Shung-Haur Yang1,2, Jen-Kou Lin1,2 and Jeng-Kai Jiang1,2*
1Department of Colon and Rectal Surgery, Taipei Veterans General Hospital, Taiwan
2Department of Surgery, School of Medicine, National Yang-Ming University, Taiwan
*Corresponding author: Jeng-Kai Jiang, Department of Colon and Rectal Surgery, Taipei Veterans General Hospital, No. 201, Sec. 2., Shih-Pai Rd, Taipei city, Taiwan 11217
Published: 14 Jul, 2018
Cite this article as: Huang J-H, Lin C-C, Lin H-H, Lan Y-T,
Chang S-C, Wang H-S, et al. Impact
of Postoperative Complications on
the Long-Term Outcome of Colorectal
Cancer Patients who Underwent
Curative Resection. Clin Surg. 2018; 3:
2020.
Abstract
Aim: The study evaluated the impact of postoperative complications on long-term outcomes in
patients after curative resection for Colorectal Cancer (CRC).
Methods: Patients undergoing curative resection for CRC from January 1993 to December 2009
were identified from a prospectively maintained database. Postoperative complications were graded
through Clavien-Dindo classification, and the patients were divided into minor (grade 1 and grade
2) and major (grade 3 and grade 4) complication groups. Factors potentially affecting disease-free
survival (DFS) and overall survival (OS) were examined using univariate and multivariate analyses.
Results: A total of 3 666 patients (2 375 men, 64.8%) were included. Complications developed in
823 patients (22.4%), of which 313 (8.5%) were major and 510 (13.9%) were minor complications.
The 5-year OS rates for those with major, minor, and no complications were 69.3%, 79.4%, and
86.1% (P<0.001), respectively, whereas the 5-year DFS rates were 62.9%, 71.1%, and 79.7 %
(P<0.001), respectively. Major complications were negative predictors of both OS (stage II: hazard
ratio [HR]=2.174, 95% confidence interval [CI]: 1.510-3.129, P<0.001; stage III: HR=2.026, 95%
CI: 1.482–2.771, P<0.001) and DFS (stage II: HR=1.499, 95% CI: 1.165-1.928, P=0.002; stage III:
HR=1.515, 95% CI: 1.226-1.872, P<0.001) in stage II and III patients.
Conclusion: Postoperative complications adversely affect the long-term outcomes of CRC patients
after curative resection. The impact of major complications was particularly strong in stage II and
III patients.
Keywords: Colorectal; Postoperative complication; Recurrence; Survival
Introduction
Colorectal Cancer (CRC) is the most frequently diagnosed cancer and the third leading cause of cancer death in Taiwan. Surgical resection remains the mainstay cure for patients with localized disease; however, postoperative morbidity and mortality are considerable. Postoperative complications are associated with a prolonged hospital stay and a higher reoperation rate, as well as a higher hospitalization cost. The postoperative morbidity of patients who underwent colorectal surgery has been well surveyed in previous studies, and ranges from 27.3% to 40.2%; this is high compared with the operative mortality of these patients [1-3]. Prior research has addressed the negative influences of postoperative complications on long-term outcomes in various cancers, including hepatocellular carcinoma, pancreatic cancer, gastric cancer, lung cancer, and CRC [3-7]. One retrospective study demonstrated that postoperative complications adversely affected not only long-term survival but also the disease recurrence rate in patients who underwent curative resection for CRC; however, the severity of the complications was not classified.3 Another retrospective study explored the relation between postoperative morbidity and the outcomes of CRC patients receiving elective resection; the researchers determined that major complications reduced 5-year survival, but had no significant impact on time to recurrence [8]. A third retrospective study examined 12 075 cases from the Veterans Affairs Surgical Quality Improvement Program and Central Cancer Registry database, and demonstrated that the presence of postoperative complications after CRC resection is associated with decreased long-term survival, independent of patient, disease, or treatment factors. Notably, this study recruited male participants in a proportion higher than 90%, and it lacked the data of disease recurrence outcomes [9]. Therefore, the influence of postoperative complications on longterm outcomes following CRC tumor resection deserves additional clarification. This study aimed to elucidate the effect of postoperative complications on disease recurrence and survival in CRC patients after curative resection in a tertiary referral center.
Figure 1
Figure 2
Figure 2
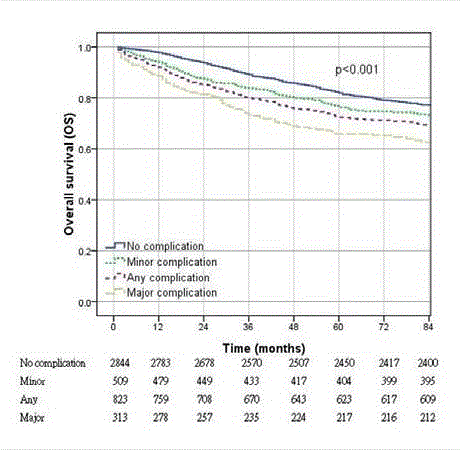
Kaplan-Meier curves comparing overall survival of patients with
different complication severities (P<0.001, log rank test).
Material and Methods
Patients who underwent curative resection for CRC from
January 1993 to December 2009 at Taipei Veterans General Hospital
were reviewed from a prospectively maintained computerized
database. Patient characteristics included age, gender, Charlson-
Age Comorbidity Index (CACI), types of operation and oncological
characteristics comprised of tumor size, lymph node harvest, adjuvant
radiotherapy or chemotherapy were collected as baseline variables.
The preoperative staging included colonoscopy, serum tumor marker
(CEA, CA-199) measurements, chest X-ray or chest Computed
Tomography (CT), abdominal CT, and magnetic resonance imaging
of the pelvis for patients with rectal cancer. Patients diagnosed
with CRC associated with inflammatory bowel diseases or familiar
adenomatous polyposis, who were at stage 0 or IV, or who had had
palliative resections or local excision without radical resection were
excluded. The tumor site was classified as being in the right or left of
the colon (either proximal or distal to the splenic flexure) and rectum.
The preoperative stage was documented according to the American
Joint Committee on Cancer (AJCC Version 6.0) staging system.
Complications that occurred within 1 month of the operation were
recorded as postoperative complications. If there were more than
one complication occurred in the same patient, the complication
that caused the worst severity was shown in Table 2. Complication
severity was categorized according to the Clavien-Dindo classification
system; the patients were divided into the major complication group,
which comprised grade 3 or 4 complications (requiring surgical,
radiological, or endoscopic intervention or intensive care) or minor
complication group, which comprised grade 1 or 2 complications
(requiring conservative or medical treatment only).
In stage III disease cases, postoperative chemotherapy was
administrated. For stage II patients with poor prognostic histological
features (i.e., poor differentiation, perineural invasion, lymph
vascular invasion, <12 harvested lymph nodes) or who presented with
tumor perforation, bleeding, or obstruction, 5-fluorouracil-based
adjuvant chemotherapy was recommended. In cases of rectal cancer,
patients with radiological evidence of T3 or T4 lesions, or lymph
node invasion, underwent preoperative chemo radiation therapy and
radical resection 6weeks to 8 weeks later routinely since 2000. Before
2000, the patients with locally advanced rectal cancer would have
neo adjuvant therapy according to the individual conditions and the
opinions from their Attending Physicians in charge.
All patients were followed up at an outpatient department every
3 months for the first 2 years, every 6 months for the third and fourth
years, and annually thereafter. Follow-up examinations included a
thorough physical examination, serum tumor marker (CEA, CA-
199) measurements, a chest X-ray, and abdominal ultrasonography.
Abdominal, pelvis, or chest CT was scheduled annually, or performed
whenever recurrence was suspected. Operative mortality was defined
as death that occurred within 30 days of the primary operation. In the
analysis of survival and recurrence, patients who died within 30 days
of the operation were excluded.
Primary outcome measures comprised Overall Survival (OS) and
disease-free survival (DFS). A patient was considered disease free if
no evidence of clinical, endoscopic, or radiological recurrence was
noted during follow-up.
Figure 3
Figure 3
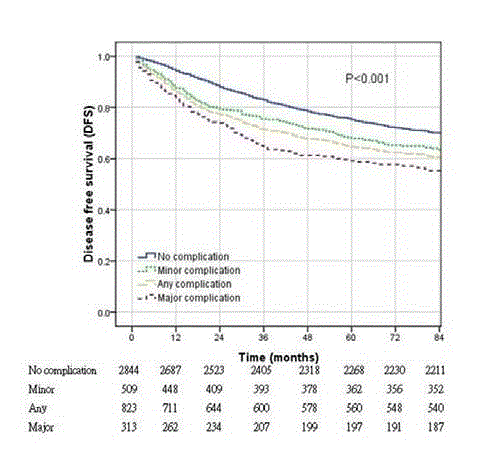
Kaplan-Meier curves comparing disease-free survival of patients
with different complication severities (P<0.001, log rank test).
Data Analysis
Statistical analyses were carried out using SPSS Version 16.0. Categorical variables were compared using a chi-square test, whereas continuous variables were compared using the Mann-Whitney U test. OS and DFS were analyzed using the Kaplan-Meier method. Variables with P<0.05 in univariate analyses were entered into the multivariate analysis, for which Cox proportional models were used to identify independent predictors of survival.
Table 1
Results
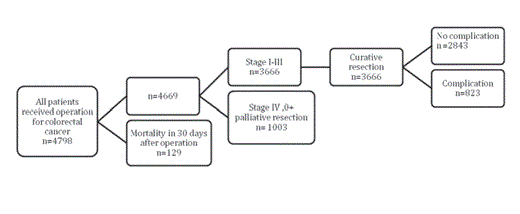
Between January 1993 and December 2009, 4798 patients
diagnosed with CRC underwent surgical resection at our institute.
The perioperative mortality rate was 2.6%; additionally, our study
excluded patients who died within 30 days of the primary operation
(n=129). Patients with stage 0 (n=210) and stage IV (n=913) cancer,
and those who received palliative resection (n=895) were also
excluded from the analysis. Therefore, a total of 3 666 patients who
underwent curative resection were included in the study (Figure 1).
A total of 823 patients developed complications, with an overall
postoperative morbidity rate of 22.4%. Clinic pathological data of
the patients are listed in Table 1. Notably, there were more male
patients, rectal cancers, and higher Charlson Comorbidity Index and
emergency operations in the complication group; additionally, less
laparoscopy-assisted surgery and more simultaneous stoma creation
were performed in the complication group. There were no significant
differences between the two groups in tumor size, number of lymph
nodes harvested, AJCC stage, tumor differentiation, or the presence
of micro invasive carcinoma.
Table 2 summarizes the details of the complications. In the
complication group, 313 patients (38.0%) had major and 510
patients (62.0%) had minor complications. Wound infection was the
most common surgical complication (n=280, 7.64%), followed by
anastomosis leakage (n=130, 3.55%). Postoperative ileus occurred in
82 patients (2.24%).
The mean follow-up duration was 55.2, 64.0, and 58.7 months
in complication-free, minor complication and major complication
groups, respectively (Table 1). There were 88 patients in the
complication group and 362 patients in non-complication group who
had lost of follow-up within two years, and the rates of loss of followup
are 11% and 12.7% successively. There were 29 patients (9.3%) of
the group with major complication, 59 patients (11.6%) of the group
with minor complication and 362 patients (12.7%) of the group
without complication lost to follow-up within 2 years after surgery
(P=0.12). Of the 3 666 patients, 425 (11.6%) experienced recurrence;
the mean time to recurrence was 22.2 months. Recurrence occurred
in 54 patients in the major complication group, in 76 patients in
the minor complication group and in 295 patients in the noncomplication
group (17.3% vs. 14.9% vs. 10.4%, P<0.001). Though
more recurrence were observed in the complication group, the time
to recurrence did not differ between the three groups (25.0 ± 20.1
months vs. 21.9 ± 22.3 months vs. 21.8 ± 17.8 months, P=0.501).
Table 3 shows the univariate and multivariate analyses for
5-year OS in patients with or without complications. According to
the results of univariate analysis, the confounders including gender,
age, Charlson Comorbidity Index, tumor location, emergency
operation, simultaneous stoma creation, laparoscopy surgery,
adjuvant treatment, AJCC stage, tumor size, lymph nodes harvest
and micro invasive carcinoma were adjusted in multivariate analysis
for estimating overall survival. A significantly worse 5-year OS
was observed in the complication group, and OS was particularly
low among patients with major complications (no complication
vs. minor complication vs. major complication =86.1% vs. 79.4%
vs. 69.3%, P<0.001, the numbers of event are 394, 105, and 96
successively) (Figure 2). Similarly, the multivariate analysis revealed
that the hazard ratio (HR) increases in the major complication group
(HR=1.70, P=0.001). The other factors associated with an adverse
5-year OS included the age ≧ 68, Charlson Comorbidity Index ≧
7, simultaneous stoma creation, open surgery, without adjuvant
therapy, AJCC stage III cancer, the presence of micro invasive
carcinoma, tumor size ≧5cm and lymph nodes harvest <18.
The univariate and multivariate analyses for 5-year DFS in
patients with or without complications are summarized in Table
4. The confounders including Charlson Comorbidity Index, tumor
location, emergency operation, simultaneous stoma formation,
laparoscopy surgery, adjuvant treatment, AJCC stage, and tumor
size and micro invasive carcinoma were entered into multivariate
analysis according to the results in univariate analysis. A significantly
worse 5-year DFS was observed in the complication group, and
DFS was particularly low among patients with major complications
(no complication vs. minor complication vs. major complication
=79.7% vs. 71.1% vs. 62.9%, P<0.001, and the number of events are
576, 147, 116 successively) (Figure 3). The multivariate analysis
revealed that the HR increases as the severity of the complications
increases (HR=1.25, 1.63 in minor and major complication groups,
respectively). The other factors associated with an adverse 5-year DFS
included the Charlson Comorbidity Index ≧ 7, simultaneous stoma
creation, an emergency operation, open surgery, AJCC stage II or III
cancer, and the presence of micro invasive carcinoma.
Subsequently, we examined the influence of complication
severity on long-term outcomes at different clinical stages. Minor
complications posed no significant influence on 5-year OS or DFS
in patients at any stage; conversely, major complications significantly
reduced both OS and DFS in stage II and stage III patients (Table 5).
However, the impact was not seen in stage I patients.
Table 2
Table 3
Table 3
Univariate (Cox Proportional Hazard Regression) and Multivariate (Cox
Proportional Hazard Regression) Analysis of 5-Year Overall Survival (OS) in
Patients with Minor complication, Major complication and Without Complications.
Table 4
Table 4
Univariate (Cox Proportional Hazard Regression) and Multivariate
(Cox Proportional Hazard Regression) Analysis of 5-Year Disease-Free Survival
(DFS) in Patients with Minor complication, Major complication and Without
Complications.
Table 5
Table 5
Influence of Major Complications on 5-Year Overall Survival and Disease-Free Survival in Different Stages.
Discussion
This study consisted of the largest patient cohort from a single
institute. The overall complication rate was 22.4%, which is lower
than the results that have been reported in prior research and
reflects the higher quality of care provided through our institute as
a tertiary referral center [2,3,8,9]. Our data reveal that postoperative
complications adversely affected the long-term outcomes of CRC
patients who underwent curative resection, with the HR being
approximately 1.5 for both OS and DFS, which is similar to that in a
previous study [9]. Moreover, we demonstrated that adverse outcomes
were related to the severity of the complications and the cancer stage
of the patients; thus, stage II and III patients who experienced a major
complication had the poorest OS and DFS outcomes.
Several studies have demonstrated significantly less favorable
long-term outcomes in patients with postoperative complications
after surgical resection for CRC with liver metastases [10-13].
However, studies investigating the impacts of complications after
curative resection for stage I, II, and III CRC patients are sparse. Law
et al. [3] demonstrated that postoperative complications significantly
negatively affect the OS and overall recurrence rate in stage I to
III CRC patients after curative resection. The study enrolled 1 657
patients from one hospital between 1996 and 2004; however, the
complications were not stratified by severity. Odermatt et al. [8]
reported significant effects of major complications on OS, but the
same negative influence was not observed in the multivariate analysis
for DFS; moreover, the total number of patients and the number of
patients with complications were limited. Artinyan et al. [9] analyzed
a large number of cases from a system-wide database of veterans
in the United States and determined that complications, especially
infectious complications, were related to poor OS. However, the
database comprised male patients in a proportion greater than 90%
and lacked detailed information on the complications; furthermore,
the study did not explore disease recurrence. Conversely, our study
comprises a large patient population with thorough information on
postoperative complications after curative CRC surgery, and is the
first to discuss the relationship between complication severity and
long-term outcomes in patients at different clinical stages.
In our study, postoperative major complications had no
significant impact on the long-term survival of stage I CRC patients.
The OS and DFS did not differ between those with and those without
major complications in stage I CRC patients (OS: 86.6% vs. 83%,
P=0.22; DFS: 83% vs. 86.6%, P=0.28), which may be due to the less
or limited invasive nature of early-stage cancers. Moreover, the
recurrence rate in these patients was low, limiting the influence of
any complications on long-term outcomes. However, adverse effects
of major complications were evident in both stage II and III patients.
Similar results were observed by Khoury et al. [14], who reported lower
5-year OS and DFS rates in patients with complications that required
early reoperation, which were classified as major complications in
our study; additionally, Nachiappan et al. [15] reported anastomosis
leakage that could be managed with conservative treatment was not
an independent factor for poor long-term outcomes as well.
Other independent factors associated with poor DFS include
old age, higher Charlson Comorbidity Index, simultaneous fecal
diversion, open surgery, emergency operation, and micro invasive
carcinoma. The complication group in this study consisted primarily
of patients who were older, male, had higher Charlson Comorbidity
Index score, rectal cancer and micro invasive carcinoma. All these
factors had been reported to be associated with poor prognosis
[16-19]. Additionally, more patients in the complication group
underwent emergency surgery, which was also viewed as an
independent predictor for adverse DFS in the study by Hogan et
al. [20] The result that higher Charlson Comorbidity Index being
significantly associated with poorer disease recurrence outcome after
tumor resection were observed in various cancer including renal
cell carcinoma, pancreatic cancer, breast cancer and hepatocellular
carcinoma [21-25]. The adverse effects of increased Comorbidity
burden and Charlson Comorbidity Index on the survival of patients
with colon cancer were also stated in previous studies [26-28].
Ouellette JR et al. [16] reviewed 279 patients and divided the patients
into two groups using Charlson Comorbidity Index score 7 as a cutoff
based on the median of their dataset. They showed significant cancerrelated
survival advantage for Charlson Comorbidity Index score of 7
or less. 16 similarly, according to a review by Marventano Stefano, et
al. [29], increased Charlson Comorbidity Index scores had an inferior
cancer-specific survival and those with Charlson Comorbidity Index
scores ≧ 2 had about 1.6-fold increased risks of death [29]. The
higher Charlson Comorbidity Index scores was found related to the
postoperative complication in patients underwent colorectal cancer
resection, and the poor outcome of patients with higher Charlson
Comorbidity Index scores may attribute to this [30-32].
There were also more patients undergoing open surgery in the
complication group. In a previous study, Wind et al. [19] detected
significantly fewer circulating tumor cells during laparoscopic
surgery; by contrast, Rahbari et al. [20] presented a meta-analysis
that concluded that the detection of circulating tumor cells in the
peripheral blood indicated poor prognosis in patients with primary
CRC [33,34]. We argue that open surgery probably induced more
tumor cells into circulation, which may be responsible for further
metastasis. However, the impact of open surgery on long-term
outcomes can be confounded by other factors, such as complicated
lesions (e.g., large tumor size or locally advanced cancer) and an
emergency requiring open surgery.
Previous studies investigating the influence of postoperative
complications on the outcomes of patients with CRC, metastatic
liver tumors, lung tumors, and gastric tumors have indicated that
complications increase the risk of tumor recurrence [3-9,12,13].
Proposed mechanisms for this increase include a period of immune
suppression, possibly caused by a systemic inflammatory response,
following postoperative complications. Particularly after major
complications, the possible reoperation and critically ill status
can induce the systemic inflammatory response, which was likely
correlated with a temporarily immune compromised system [35,36].
Prior research has also suggested that the presentation of cytokines
and the subsequent down regulation of antigen-presenting cells
play a key role in metastasis [37,38]. Other studies have revealed
that bacterial antigen-mediated processes increase cell adhesion and
metastasis [39,40]. Consequently, the immune suppression status
induced through complications might not only break the resistance
to spreading tumor cells but also promote tumor recurrence. These
findings can facilitate explaining why minor complications do not
affect patient outcomes. Specifically, the systemic inflammatory
response following minor complications may be limited and not
reach the threshold that affects outcomes.
Another important issue raised by Krarup et al. [41] is the
complete omission of adjuvant therapy, which is another potential
reason for poor prognosis in the complication group. However,
in our study, the percentage of patients who underwent adjuvant
therapy did not differ between the three groups significantly (major
complication vs. minor complication vs. no complication =32.9% vs.
35.5% vs. 37.2%, P=0.284).
There are limitations in this study. First, the starting date of
adjuvant therapy was not specified in the database; therefore, the
effect of delayed adjuvant chemotherapy could not be assessed. It is
quite common that adjuvant therapy is delayed by the occurrence of
complications, which may be attributed to the unfavorable outcomes
in the patients with complication as well [41]. Second, this is a
retrospective study, and the complications recorded were obtained by
the chart review. Some minor complications, such as wound infection
or urinary tract infection, may be omitted in the discharge diagnosis.
Therefore, the impact of minor complication could be difficult to
evaluate.
Postoperative complications after curative resection for CRC
had adverse effects on not only long-term OS but also DFS. The
poor outcomes were related to the severity of the complications and
the cancer stage of the patients, with stage II and III patients who
experienced major complications after surgery having the poorest OS
and DFS outcomes.
References
- Bokey EL, Chapuis PH, Fung C, Hughes WJ, Koorey SG, Brewer D, et al. Postoperative morbidity and mortality following resection of the colon and rectum for cancer. Dis Colon Rectum. 1995;38(5):480-6.
- Alves A, Panis Y, Mathieu P, Mantion G, Kwiatkowski F, Slim K. Postoperative mortality and morbidity in French patients undergoing colorectal surgery: results of a prospective multicenter study. Arch Surg 2005;140(3):278-83.
- Law WL, Choi HK, Lee YM, Ho JW. The impact of postoperative complications on long-term outcomes following curative resection for colorectal cancer. Ann Surg Oncol. 2007;14(9):2559-66.
- Chok KS, Ng KK, Poon RT, Lo CM, Fan ST. Impact of postoperative complications on long-term outcome of curative resection for hepatocellular carcinoma. Br J Surg. 2009;96(1):81-7.
- Kamphues C, Bova R, Schricke D, Hippler-Benscheidt M, Klauschen F, Stenzinger A, et al. Postoperative complications deteriorate long-term outcome in pancreatic cancer patients. Ann Surg Oncol. 2012;19(3):856-63.
- Rueth NM, Parsons HM, Habermann EB, Groth SS, Virnig BA, Tuttle TM, et al. The long-term impact of surgical complications after resection of stage I nonsmall cell lung cancer: a population-based survival analysis. Ann Surg. 2011;254(2):368-74.
- Tokunaga M, Tanizawa Y, Bando E, Kawamura T, Terashima M. Poor survival rate in patients with postoperative intra-abdominal infectious complications following curative gastrectomy for gastric cancer. Ann Surg Oncol. 2013;20(5):1575-83.
- Odermatt M, Miskovic D, Flashman K, Khan J, Senapati A, O'Leary D, et al. Major postoperative complications following elective resection for colorectal cancer decrease long-term survival but not the time to recurrence. Colorectal Dis. 2015;17(2):141-9.
- Artinyan A, Orcutt ST, Anaya DA, Richardson P, Chen GJ, Berger DH. Infectious postoperative complications decrease long-term survival in patients undergoing curative surgery for colorectal cancer: a study of 12,075 patients. Ann Surg. 2015;261(3):497-505.
- Tanaka K, Kumamoto T, Nojiri K, Matsuyama R, Takeda K, Endo I. Impact of Postoperative Morbidity on Long-Term Survival After Resection for Colorectal Liver Metastases. Ann Surg Oncol. 2016;23(5):929-37.
- Schiesser M, Chen JW, Maddern GJ, Padbury RT. Perioperative morbidity affects long-term survival in patients following liver resection for colorectal metastases. J Gastrointest Surg. 2008;12(6):1054-60.
- Laurent C, Sa Cunha A, Couderc P, Rullier E, Saric J. Influence of postoperative morbidity on long-term survival following liver resection for colorectal metastases. Br J Surg. 2003;90(9):1131-6.
- Farid SG1, Aldouri A, Morris-Stiff G, Khan AZ, Toogood GJ, Lodge JP, et al. Correlation between postoperative infective complications and long-term outcomes after hepatic resection for colorectal liver metastasis. Ann Surg. 2010;251(1):91-100.
- Khoury W1, Lavery IC, Kiran RP. Impact of early reoperation after resection for colorectal cancer on long-term oncological outcomes. Colorectal Dis. 2012;14(3):e117-23.
- Nachiappan S, Askari A, Malietzis G, Giacometti M, White I, Jenkins JT, et al. The impact of anastomotic leak and its treatment on cancer recurrence and survival following elective colorectal cancer resection. World J Surg. 2015;39(4):1052-8.
- Ouellette JR, Small DG, Termuhlen PM. Evaluation of Charlson-Age Comorbidity Index as predictor of morbidity and mortality in patients with colorectal carcinoma. J Gastrointest Surg. 2004;8(8):1061-7.
- Chan JA, Meyerhardt JA, Niedzwiecki D, Hollis D, Saltz LB, Mayer RJ, et al. Association of family history with cancer recurrence and survival among patients with stage III colon cancer. JAMA. 2008;299(21):2515-23.
- Yun HR, Lee LJ, Park JH, Cho YK, Cho YB, Lee WY, et al. Local recurrence after curative resection in patients with colon and rectal cancers. Int J Colorectal Dis. 2008;23(11):1081-7.
- Nikberg M, Chabok A, Letocha H, Kindler C, Glimelius B, Smedh K. Lymphovascular and perineural invasion in stage II rectal cancer: a report from the Swedish colorectal cancer registry. Acta Oncol. 2016;55(12):1418-24.
- Hogan J, Samaha G, Burke J, Chang KH, Condon E, Waldron D, et al. Emergency presenting colon cancer is an independent predictor of adverse disease-free survival. Int Surg. 2015;100(1):77-86.
- Reinstatler L, Klaassen Z, Madi R, Terris MK, Moses KA. Comorbidity Is a Competing Factor for Disease Recurrence Postnephrectomy. South Med J. 2017;110(5):369-74.
- Asano T, Yamada S, Fujii T, Yabusaki N, Nakayama G, Sugimoto H, et al. The Charlson age comorbidity index predicts prognosis in patients with resected pancreatic cancer. Int J Surg. 2017;39:169-175.
- Frees S, Kamal MM, Knoechlein L, Bell R, Ziesel C, Neisius A, et al. Differences in Overall and Cancer-specific Survival of Patients Presenting With Chromophobe Versus Clear Cell Renal Cell Carcinoma: A Propensity Score Matched Analysis. Urology 2016;98:81-7.
- Chang HT, Shi HY, Wang BW, Yeh SJ. Breast Cancer Incidence and Predictors of Surgical Outcome: a Nationwide Longitudinal Study in Taiwan. Clin Oncol (R Coll Radiol). 2017;29(6):362-9.
- Chiu CC, Wang JJ, Chen YS, Chen JJ, Tsai TC, Lai CC, et al. Trends and predictors of outcomes after surgery for hepatocellular carcinoma: A nationwide population-based study in Taiwan. Eur J Surg Oncol. 2015;41(9):1170-8.
- Hines RB, Chatla C, Bumpers HL, John W. Waterbor, Gerald McGwin, Jr, Ellen Funkhouser, et al. Predictive capacity of three comorbidity indices in estimating mortality after surgery for colon cancer. J Clin Oncol. 2009;27(26):4339-45.
- Hines RB, Shanmugam C, Waterbor JW, McGwin G Jr, Funkhouser E, Coffey CS, et al. Effect of comorbidity and body mass index on the survival of African-American and Caucasian patients with colon cancer. Cancer. 2009;115(24):5798-806.
- Iversen LH. Aspects of survival from colorectal cancer in Denmark. Dan Med J. 2012;59(4):B4428.
- Marventano S, Grosso G, Mistretta A, Bogusz-Czerniewicz M, Ferranti R, Nolfo F, et al. Evaluation of four comorbidity indices and Charlson comorbidity index adjustment for colorectal cancer patients. Int J Colorectal Dis. 2014;29(9):1159-69.
- Tian Y, Xu B, Yu G, Li Y, Liu H. Age-adjusted charlson comorbidity index score as predictor of prolonged postoperative ileus in patients with colorectal cancer who underwent surgical resection. Oncotarget 2017;8(13):20794-801.
- Tan KK, Koh FH, Tan YY, Liu JZ, Sim R. Long-term outcome following surgery for colorectal cancers in octogenarians: a single institution's experience of 204 patients. J Gastrointest Surg. 2012;16(5):1029-36.
- Asa Z, Greenberg R, Ghinea R, Inbar R, Wasserberg N, Avital S. Grading of complications and risk factor evaluation in laparoscopic colorectal surgery. Surg Endosc. 2013;27(10):3748-53.
- Wind J, Tuynman JB, Tibbe AG, Swennenhuis JF, Richel DJ, van Berge Henegouwen MI, et al. Circulating tumour cells during laparoscopic and open surgery for primary colonic cancer in portal and peripheral blood. Eur J Surg Oncol. 2009;35(9):942-50.
- Rahbari NN, Aigner M, Thorlund K, Mollberg N, Motschall E, Jensen K, et al. Meta-analysis shows that detection of circulating tumor cells indicates poor prognosis in patients with colorectal cancer. Gastroenterology. 2010;138(5):1714-26.
- Ogura H, Tanaka H, Koh T, Hashiguchi N, Kuwagata Y, Hosotsubo H, et al. Priming, second-hit priming, and apoptosis in leukocytes from trauma patients. J Trauma. 1999;46(5):774-81.
- Brochner AC, Toft P. Pathophysiology of the systemic inflammatory response after major accidental trauma. Scand J Trauma Resusc Emerg Med. 2009;17:43.
- Elaraj DM, Weinreich DM, Varghese S, Puhlmann M, Hewitt SM, Carroll NM, et al. The role of interleukin 1 in growth and metastasis of human cancer xenografts. Clin Cancer Res. 2006;12(4):1088-96.
- Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454(7203):436-44.
- Doan HQ, Bowen KA, Jackson LA, Evers BM. Toll-like receptor 4 activation increases Akt phosphorylation in colon cancer cells. Anticancer Res. 2009;29(7):2473-8.
- Hsu RY, Chan CH, Spicer JD, Rousseau MC, Giannias B, Rousseau S, et al. LPS-induced TLR4 signaling in human colorectal cancer cells increases beta1 integrin-mediated cell adhesion and liver metastasis. Cancer Res. 2011;71(5):1989-98.
- Krarup PM, Nordholm-Carstensen A, Jorgensen LN, Harling H. Anastomotic leak increases distant recurrence and long-term mortality after curative resection for colonic cancer: a nationwide cohort study. Ann Surg. 2014;259(5):930-8.