Case Report
Tracking Down the Cause of Necrotizing Fasciitis in a Patient with Negative Cultures
Bas P Vierhout1, Evert van Zanten2, Guido Wisselink2, Mirjam AMD Kooistra-Smid2,3 and Alewijn Ott2*
1Department of Surgery, Wilhelmina Ziekenhuis, Assen, Netherlands
2Department of Medical Microbiology, Certe, Groningen, Netherlands
3Department of Medical Microbiology, University of Groningen, University Medical Center Groningen, Groningen, Netherlands
*Corresponding author: Alewijn Ott, Department of Medical Microbiology, Certe, Groningen, Netherlands
Published: 12 Jul, 2018
Cite this article as: Vierhout BP, van Zanten E, Wisselink G,
Kooistra-Smid MAMD, Ott A. Tracking
Down the Cause of Necrotizing Fasciitis
in a Patient with Negative Cultures. Clin
Surg. 2018; 3: 2011.
Abstract
In a patient suspected of lower abdominal necrotizing fasciitis cultures remained negative, possibly because broad spectrum antibiotics had already been given before tissue for culture was obtained. 16S-23S rDNANext Generation Sequencing showed that 99.7% of bacterial DNA was of Streptococcus pyogenes. 16S-23S rDNA can replace culture for identification of bacteria, also in polymicrobial infection.
Background
Necrotizing Fasciitis (NF) is a severe disease of sudden onset that spreads rapidly [1]. Symptoms
include red skin, severe pain and fever. Risk factors include poor immune function and a break
in the skin, for instance a wound. But also healthy persons may suffer from NF, without a visible
entrance through the skin, mostly in the perineal region or the limbs.
The disease is feared because of its high mortality and subsequent morbidity. The infection
spreads rapidly over the fascia of the muscles and open surgical drainage is the only treatment
option: Infected tissue must be removed without delay, in combination with intravenous antibiotics.
Most often, the causative agent is a Streptococcus from Lancefield group A (S. pyogenes). If S.
pyogenes is involved antibiotic treatment with a high dose of penicillin suffices. But combinations
of antibiotics with a broader spectrum are required in infections in the perineal area, because these
may be polymicrobial.
Case Presentation
A 68-year-old female patient complained of severe pain and localized redness since a few hours,
spreading over the lower abdomen. Four days before, she had some localized pain on the right
side of the abdomen, which gradually shifted to the left. Mobilizing aggravated the pain and made
her nauseous, and as a result, she ate and drank less. She had no abdominal surgery or relevant
infectious medical history in the past. Only bad teeth had been removed 6 years earlier, to replace
them by prosthesis.
At the emergency room she had a fever of 39.2°C, with a pulse of 107 per minute and a tension of
111/63 mmHg. The abdominal redness was moderately delimited and there was no swelling (Figure
1). Some bowel sounds were heard and the redness felt firmer than the surrounding tissue.
Laboratory tests showed a leukocytosis of 44,000*106 per liter, with a left upper shift. A
hyponatremia was accompanied by elevated urea and creatinine level of 23.3 mmol/l and 272
μmol/l, respectively. C-reactive protein was 639 mg/l. An emergency CT-scan did not show intraabdominal
disorder: no abnormal air or fluid collection in the abdominal cavity and normal
intra abdominal organs. But the subcutaneous fat of the anterior abdominal wall showed signs of
inflammation without gas.
The patient was started on piperacillin with tazobactam in combination with clindamycin and
surgical exploration was performed on suspicion of NF. During the operation the fascia seemed
intact, but the subcutaneous tissue, adjacent to the fascia had a discolored, greyish, avital aspect.
Biopsies for cultures and histology of the tissue were taken and the wound was left open.
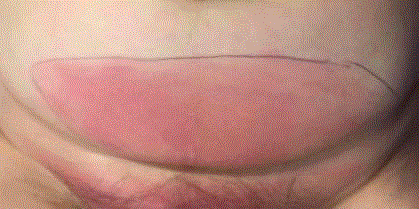
Figure 1
Figure 1
Redness of the lower abdomen of the patient before surgery. The
line around the redness was drawn in the emergency department in order to
indicate any increase.
Microbiology
Blood cultures taken on admission and culture of the tissue taken during surgery unfortunately remained negative. No bacteria were seen in the Gram stain of the tissue. Because of these negative findings and uncertainty about the best choice of antibiotics to continue the treatment we additionally examined the tissue with 16S-23S rDNANext Generation Sequencing [2]. This method is based on a PCR amplification of the 16S-23S rRNA region followed by amplicon sequencing on the MiSeq (Illumina, Inc., San Diego, CA). The resulting reads are denovo assembled into contigs. Species identification is based on an alignment of the contig sequences with the sequences deposited in the reference databases. Th is resulted in identification of Streptococcus pyogenes only, with a Relative Abundance (RA) of 99.7%. The RA indicates the relative percentage of reads that represents the same species in a sample.
Discussion
The best remedy for NF is early recognition, followed by surgery
and antibiotic treatment focused upon the causative agent(s).
Unfortunately, in some cases, these agents cannot be identified, as
was the case in this patient. Most likely this was due to the high dose
of intravenous antibiotic that was already given at the emergency
department. The infection was in an early stage and therefore tissue
circulation was still intact. Streptococcus pyogenes is usually highly
sensitive to the penicillin class antibiotics.
Knowing the causative bacteria enabled us to finish the treatment
with oral amoxicillin after the patient recovered. 16S-23S rDNA
NGS could exclude the alternative diagnosis in this patient of the
polybacterialFournier NF, which requires a broader spectrum of
antibiotics [3]. 16S-23S rDNA NGS can therefore contribute to
rational antibiotic use.
References
- Stevens DL, Bryant AE. Necrotizing Soft-Tissue Infections. N Engl J Med. 2017;377:2253-65.
- Sabat AJ, van Zanten E, Akkerboom V, Wisselink G, van Slochteren K, de Boer RF, et al. Targeted next-generation sequencing of the 16S-23S rRNA region for culture-independent bacterial identification - increased discrimination of closely related species. Sci Rep. 2017;7(1):3434.
- Mallikarjuna MN, Vijayakumar A, Patil VS, Shivswamy BS. Fournier's Gangrene: Current Practices. ISRN Surg. 2012:942437.