Case Report
Unique Treatment Considerations for Chest Wall Desmoid Tumor Invading the Breast
Miriam S, Jody MK, Miriam D and Maria C*
Department of Surgery, Jacobi Medical Center, USA
*Corresponding author: Maria C, Department of Surgery, Jacobi Medical Center, 1400 Pelham Parkway South Bronx, NY 10461, USA
Published: 20 Mar, 2018
Cite this article as: Miriam S, Jody MK, Miriam D, Maria C.
Unique Treatment Considerations for
Chest Wall Desmoid Tumor Invading
the Breast. Clin Surg. 2018; 3: 1942.
Abstract
Desmoid Tumors (DT), a fibromatous proliferative disease, are defined by the World Health Organization as clonal fibroblastic proliferations that arise in the deep soft tissues and are characterized by infiltrative growth and a tendency toward local recurrence but an inability to metastasize. DT are rare tumors, occurring in three to four cases per one million of the U.S. population. DT of the chest wall represents 8% to 10% of all cases. Although surgery is the primary treatment modality, there remains significant controversy amongst surgeons regarding the management of disease involving adjacent breast given the possibility for significant functional and aesthetic compromise. This is a rare and interesting case of a young woman with a massive chest wall DT displacing the breast necessitating extensive chest wall resection and right partial mastectomy.
Case Presentation
A 21 year old woman presented with right breast pain and a gradually increasing mass over
3 months duration. On examination, a 9 cm to 12 cm right breast mass encompassed the entire
outer quadrant of the right breast, seemingly adherent to the chest wall. Breast ultrasound showed
a complex 8.4 cm by 6.0 cm by 10.3 cm hypoechoic mass with mild increased Doppler flow.
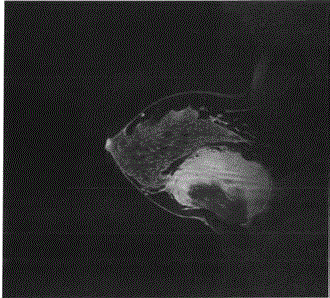
Mammography and breast MRI revealed a chest wall mass anteriorly displacing the breast tissue,
originating from the pectoralis major muscle (Figure l). Core needle biopsy revealed a spindle cell
lesion. Immunohistochemical analysis of the core needle biopsy was negative for markers AE1/AE3,
supporting a diagnosis of fibromatosis.
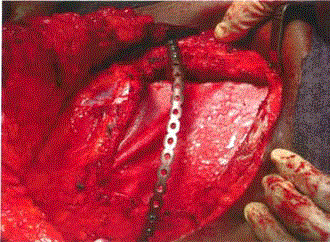
Surgical excision of the DT, including partial right mastectomy, and chest wall resection and
reconstruction, was undertaken with both breast and thoracic surgeon involvement. The mass was
mobilized and noted to originate from the pectoralis major muscle. Resection of the fifth and sixth
ribs was necessitated by adherent tumor. A chest wall reconstruction was performed using a rib
plate and strattice mesh to reconstruct the chest wall contour and prevent lung herniation. Strattice
mesh was fixed to the thoracic cage as an underlay to reapproximate the parietal pleura and protect
the lung from direct contact with the rib plate and subcutaneous tissues. This was secured in place
with sutures placed through drill holes within the ribs and sternum. A plan for delayed muscle
flap reconstruction of the breast was anticipated due to large volume loss in the breast (Figure 2).
Anatomic pathology revealed a 15 cm tumor with WHO classification of desmoid type Fibromatosis
(intermediate, locally aggressive, 2/10 mitosis per high-power field, no necrosis, 2 cm margins).
The patient was discharged after a short hospital stay without complication. On return visit
the patient was found to be pregnant, delaying breast reconstruction and precluding hormonal
treatment typically recommended for DT. At follow up two and a half years post surgery, the patient
remains without recurrence of tumor on PET CT imaging. On exam, the scar is well healed, with
a significant loss of volume in her right breast compared to the left, but with preserved chest wall
contour. The patient was referred for reconstructive breast surgery. A muscle flap is planned to
provide a breast mound.
Discussion
Chest wall tumors have long represented a unique challenge for surgeons. In the past, an
inability to perform successful reconstruction for large thoracic wall defects led to high perioperative
morbidity from infection of the pleural cavity, respiratory failure and paradoxical breathing. Since
improvements in surgical reconstruction, antibiotics prophylaxis and anesthesia, the role of surgical
treatment for primary malignant tumors is now established. DT is treated as a primary malignant
chest wall tumor with regard for adequate margins. For resections involving a defect greater than 5 cm in diameter, thoracic wall solidity must be reconstituted, and can best be achieved with a combination of myocutaneous flaps and mesh
materials that provide solidity, stability, protection of endothoracic
organs and prevent paradoxical breathing [1,2]. A study in patients
undergoing microvascular breast reconstruction suggested that rib
plating adds limited additional morbidity and effectively prevents
chest contour deformity [3].
While surgery has become the therapeutic mainstay for
resectable, localized DT, because of clinical course variability where
many tumors spontaneously regress or grow, varying site behavior
and an absence of a prospective randomized trial to compare different
treatment methods, the exact application of the above mentioned
surgical intervention has recently been extensively debated [4].
Support for aggressive surgical resection for extra-abdominal
DT with safe margins of 2 cm to 3 cm stems from the potential of
fibromatosis to grow locally and invade surrounding structures,
causing symptoms, and high rates of local recurrence [5]. Support
of more conservative treatment stems from a less optimal cosmetic
outcome and risk of loss of function, an absence of reports showing
DT can metastasize or evidence that residual tumor has a negative
impact on five year disease free or overall survival [6].
A review of standard treatments of truncate sarcomas, including
DT shows that age (younger), tumor site (chest wall) and size (>10
cm) are the strongest predictors of tumor recurrence (50% after
5 years), with margin status (RO compared to RI) not considered
predictive. While initial surgical resection for chest wall desmoids
is justifiable given their higher rate of symptomatic progression
and re-recurrence, consideration should be given to non-operative
management of recurrence rather than repeat resection which may
lead to significant morbidity [l]. Our patient met criteria for resection
based on rapid tumor growth, DT chest wall location, tumor size
exceeding 10cm, and significant risk factors for recurrence, including
young age. Studies have shown that optimal chest wall reconstruction
achieves stability and maintains pulmonary functionality [2-4].
Given our patient's necessity for complete excision of chest wall DT
involving a multiple rib resection, we opted to utilize rib plating to
achieve optimal aesthetic and functional outcome.
Figure 1
Figure 1
MRI scan (sagittal view) demonstrating a 10cm by 10 cm retro
mammary complex mass with predominantly enhancing solid components
as well as cystic areas. The mass is inseparable from the chest wall muscle
and anteriorly displaces the right breast.
Figure 2
References
- Wilder F, D'Angelo S, Crago A. Soft tissue tumors of the trunk: Management of local disease in the breast and chest andabdominal walls. J Surg Oncol. 2015;111(5):546-52.
- Incarbone M, Pastorino, U. Surgical Treatment of Chest Wall Tumors. W J Surg. 2001;25(2):218-30.
- Ahdoot MA, Echo A, Otake LR, Son J, Zeidler KR, Saadian I, et al. The matrix rib plating system: Improving aesthetic outcomes in microvascular breast reconstruction. Ann Plast Surg. 2013;70(4):384-8.
- Kasper B, Strobel P, Hohenberger P. Desmoid tumors: Clinical features and treatment options for advanced disease. Oncologist. 2011;16(5):682-93.
- Shen C, Zhou Y, Che G. Management of a female with recurrence of fibromatosis of the chest wall adjacent to the breast: A case report. J Cardiothorac Surg. 2013;8:41.
- Neuman HB, Brogi E, Ebrahim A, Brennan MF, Van Zee KJ. Desmoid tumors (fibromatoses) of the breast: A 25 year experience. Ann Surg Oncol. 2008;15(1):274-80.