Case Report
Platelet Rich Plasma: A Potential Treatment Option in Hyper Pigmentation of Skin
Raghvendra Bohara1, Nilesh Bhamre1, Jeevitaa Kshersagar1, Kishor Tardalkar1, Shashikant
Desai2, Mayuresh Despande3 and M G Joshi1*
1Department of Stem Cells and Regenerative Medicine, D.Y. Patil University, Kolhapur, India
2Stem Plus Cryopreservation, Maharashtra, India
3Department of Surgery, D.Y. Patil University, Kolhapur, India
*Corresponding author: Meghnad G Joshi, Department of Stem Cells and Regenerative Medicine, D.Y. Patil Education Society, Deemed University, D.Y. Patil Vidyanagar, Kasaba Bawda
Published: 20 Mar, 2018
Cite this article as: Bohara R, Bhamre N, Kshersagar
J, Tardalkar K, Desai S, Despande
M, et al. Platelet Rich Plasma: A
Potential Treatment Option in Hyper
Pigmentation of Skin. Clin Surg. 2018;
3: 1938.
Abstract
Melasma is characterized by symmetrical hyper pigmented macules and patches on the sun exposed area of the face, commonly on forehead, cheeks, lips and nose especially in women. It is an acquired pigmentary disorder. Its pathogenesis is not yet fully understood but the common risk factors for melasma include pregnancy, oral contraceptives, genetic factor, Ultraviolet exposure. Most recently data supported that pathogenesis of melasma involves vascular growth factors, Wnt pathway modulator genes and inducible Nitric Oxide Synthase (iNOS) expression and down regulation of H19 genes with a still unresolved pathogenesis. A wide variety of treatments include hydroquinone, tretinoin, kojic acid, and tranexamic acid, azelaic acid, glycolic acid, laser, broad spectrum sunscreen and sun avoidance. Though it is effective treatment, adverse effects have also been identified. In recent times, Platelet Rich Plasma (PRP) is fetching attention in aesthetic medicine. Interestedly, this study aimed to assess the effectiveness of PRP injection intradermally. It demonstrated that PRP injection resulted in the reduced hyper pigmented lesions in melasma with fewer side effects. Hence, PRP could be a promising treatment and hope for new safe and effective treatment options.
Introduction
Hyper pigmentation disorders corresponding intracellular signaling cascades that lead to the
stimulation of melanogenesis it include Ultraviolet B (UVB) hyper pigmentation and melasma
[1]. Use of anti-pigmenting agents developed so far can inhibit melanogenesis partially. However,
topical agents used for hyper pigmented skin area are potent anti-melanogenic agents capable of
suppressing constitutive pigmentation and this may lead to hypo pigmentation in surrounding
areas [2]. From this viewpoint, controlling the activation of melanogenesis would be an appropriate
approach to develop new potent anti-pigmenting agents without the risk of hypo pigmentation.
With this approach, we have been looking for unique growth factors that have the potential to
resolute hyper pigmentation.
Platelet-Rich Plasma (PRP), platelet-rich concentrate, autologous platelet gel or platelet releasate
all refer to one concept which is an autologous concentration of human platelets contained in a small
volume of plasma. It is known for a long time that fibrin clot and platelets have haemostatic and
tissue repairing effect [3,4]. In 1975, an article was published with the concept of platelet gel under
the title of ‘use of platelet-fibrinogen-thrombin mixture as a corneal adhesive. An exciting report
was published in 1979 about the usage of gel foam in suture less nerve anastomosis. After a few
years an animal model showed that platelets and fibrin initiate a process consist of cell migration,
collagen synthesis, fibroplasia and angiogenesis which helps the lesion healing. The real application
of platelet releasate in treating wounds has begun in the mid-1980s after publication of Kingthon in
1986. In 1997 another important reports was published about the maxillofacial surgery and platelet
gels. The usages of platelet gel became more popular in late 1990s, after the publication (1998) of a
paper about the effectiveness of the Platelet-Rich Plasma (PRP) in bone regeneration in the field of
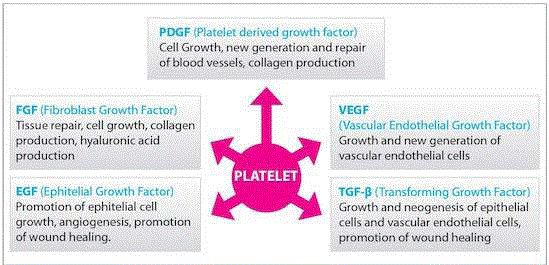
dental care. There are several growth factors in α-granules of platelets, secreted after the activation of
platelets by aggregation initiators. These factors including Platelet-Derived Growth Factor (PDGF),
Transforming Growth Factor (TGF), Vascular Endothelial Growth Factor (VEGF) and Insulin-like
Growth Factor (IGF) regulate cell migration and attachment (Figure 1) [5].
Some studies indicate that platelets have anti-inflammatory and analgesic effects and secrete
antimicrobial peptides, thus have antibiotic effects [6,7]. More than 800 proteins are secreted in this matrix affect on various cell types: osteoblasts chondrocytes fibroblasts, endothelial cells, mesenchymal stem cells from different
origins, myocytes and tendon cells which lead to a wide range of
surgical and clinical procedures and treatments which help the
platelet concentrated products [8]. Nowadays, there are publications
about the use of PRP in chronic wound treatment, soft tissue injuries,
periodontal and oral surgery, maxillofacial surgery, orthopedic and
trauma surgery, spinal surgery, heart bypass surgery, burns, cosmetic
and plastic surgery, gastrointestinal surgeries. We want to summarize
the use of PRP in dermatology especially in skin hyper pigmentation.
Disorders of Hyper Pigmentation
Melasma
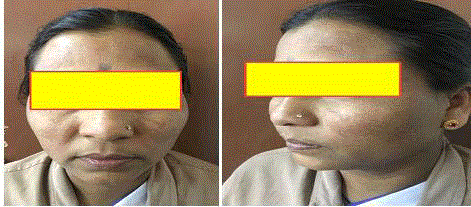
Melasma is a common acquired hypermelanosis that occurs
exclusively on sun-exposed areas, mostly on the face and occasionally
on the neck and forearms (Figure 2). Melasma is a dermatological
disease easily diagnosed by clinical examination, typically chronic
with frequent recurrences with many unknown physiopathological
aspects [9-11].
Epidemiology and etiology
Melasma is more common in women. Men have been reported to
represent 10% of cases. Melasma is more apparent during and after
periods of sun exposure. The exact cause of melasma remains elusive,
but the one most important factor implicated in its etiopathogenesis is
sunlight. Other factors in criminated in the pathogenesis of melasma
include pregnancy and exogenous hormones (i.e. oral contraceptives
and hormone replacement therapy), thyroid dysfunction, cosmetics,
phototoxic and antiseizure drugs [12-15].
Figure 1
Figure 2
Figure 3
Figure 4
Clinical Features and Classification
The number of hyper pigmented patches may range from one
single lesion to multiple patches located usually symmetrically on
the face and occasionally cheeks and both sides of nose. The lesions
have serrated, irregular, and geographic borders. According to
the distribution of lesions, the following three clinical patterns of
melasma are recognized [16-19].
• The centrofacial pattern: This is the most common pattern.
It involves the forehead, cheeks, upper lip, nose and chin.
• The malar pattern: This involves the cheeks and nose.
• The mandibular pattern: This involves the ramus of the
mandible.
Melasma can be classified into four histologic types [20,21].
• Epidermal type: The pigmentation is intensified under
Wood’s light examination. It is the most common type of melasma.
Melanin is increased in all epidermal layers. Only a few scattered
melanophages can be observed in the papillary dermis.
• Dermal type: The pigmentation is not increased under
Wood light examination. Many melanophages are throughout the
entire dermis.
• Mixed type: Under Wood’s light examination, the
pigmentation becomes more apparent only in some areas, whereas in
others there is no change. Melanin is increased in the epidermis, and
there are many dermal melanophages.
• Indeterminate type: Wood’s light examination is of no
benefit in individuals with skin type VI.
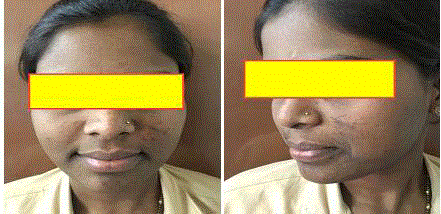
Erythema Dyschromicum Perstans
Erythema Dyschromicum Perstans (EDP) or ashy dermatosis is an idiopathic, acquired, and chronic skin disorder characterized by hyper pigmented patches on the trunk, face, and extremities.
Epidemiology and etiology most adult patients with EPD are of
Hispanic origin but white adult patients have also been reported [22].
As far as children are concerned, there are more reports with white
children than with Hispanic children. There appears to beno sexual
predilection for EDP. The cause of EDP remains unknown. There is
considerable evidence that an immunologic mechanism is involved
in the pathogenesis of the disease. A variety of possible causative
agents have been reported including ingestion of ammonium nitrate,
orally administered X-ray contrast media, exposure to chlorothalonil,
chronic hepatitis C infection, and exposure to environmental
contaminants [23].
Clinical features
EDP is characterized by oval or round-shaped blue-gray patches
over the face, trunk, and extremities. Early lesions may have a
raised, erythematous border that disappears later, and usually it is
not evident upon initial consultation. The lesions are occasionally
pruritic, and their diameter may vary from a few millimeters to
several centimeters. The trunk and proximal extremities are more
usually affected, followed by the face and neck [24]. There seems to be
no predilection for exposed or unexposed areas (Figure 3).
Figure 5
Histopathology
The histologic features of EDP are relatively non specific.
The inflammatory border of the lesions may show vacuolization
of the basal cell layer and occasional colloid bodies. There is also
mononuclear infiltrate of the mid and upper dermis and pigment
incontinence with many melanophages. In the inactive lesions, the
incontinence of pigment prevails, whereas the vacuolization of the
basal cell layer and the cellular infiltrate show different degrees of
intensity [25].
Treatment and prognosis
Sun protection is of pivotal importance in the treatment of
melasma. Without strict adherence, any treatment regimen will fail.
Recurrence during the summer is very common in patients with
melasma. The treatment of melasma includes topical formulations,
chemical peels, lasers, and light sources. Combinations of treatments
are often used to maximize results in difficult cases [26]. Topical
treatment with combination of hydroquinone, tretinoin, and a
steroid appears to be the most effective initial therapy. Glycolic acid
peels may be a useful adjunct to topical treatment, especially after a
patient’s pre-treatment with hydroquinone for 2 weeks to minimize
the risk for post procedure hyper pigmentation. Fractional laser
therapy is the only laser treatment for melasma and it could be used
as a third-line treatment in severe cases who have not responded
toother treatments and who are willing to accept the risk for post
procedure hyper pigmentation. There is no consistently effective
therapy for EDP. In children, a high rate of spontaneous remission
has been observed. Dapsone, Clofazimine, and narrow-band UVB
phototherapy have been used with good results in small series [27].
Mechanism of Hyper Pigmentation
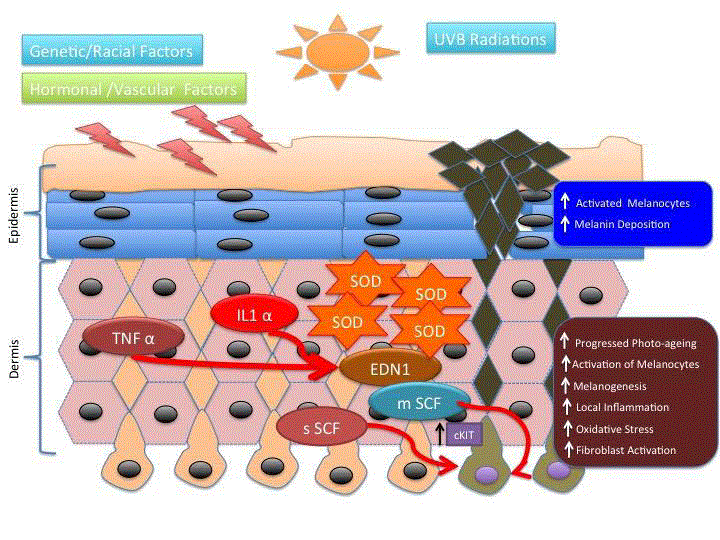
Depletion of the ozone layer results in greater potential exposure to UV-B Radiation (UVR). Single exposure to UVR results in increase in the size of melanocytes followed by an increase in tyrosinase activity. Repeated exposures to UVR lead to an increase in the number melanosomes transferred to keratinocytes, as well as an increase in the number of active melanocytes [28]. In melasma, the secretion of soluble SCF (sSCF) by dermal fibroblasts is up-regulated in the lesional dermis, probably due at least in part to the photo aging process. This leads to the penetration of sSCF into the epidermis through the basement membrane and then to the activation of epidermal melanocytes via the SCF signaling cascade, resulting in the stimulation of epidermal pigmentation. Based on the evidence that EDN1 and SCF are only the intrinsic melanogenic cytokines in hyper pigmentary disorders, it is strongly anticipated that substances able to interrupt the EDN1- or SCF-specific intracellular signaling pathways but do not directly inhibit tyrosinase activity, would be effective new anti-pigmenting agents. They would have the advantage that they do not affect normally pigmented skin where the intracellular signaling cascade is not activated, but would have distinct inhibitory effects on hyper pigmented areas of the skin with UVB-melanosis, solar lentigo and melasma (Figure 4). For such anti-pigmenting agents, there seems to be a low risk of eliciting hypo pigmentation because they have no direct inhibitory effect on tyrosinase activity and because there is no activated intracellular signaling cascade in normally pigmented skin [29,30].
PRP Treatment
Platelet-Rich Plasma (PRP) treatment is performed via the autologous injection of high concentration of platelets in a small volume of plasma. Platelet-rich plasma was obtained by spin method, followed by the collection of 10 ml of autologous whole-blood into tubes containing trisodium citrate as anticoagulant [31]. The collected blood was first centrifuged at 150 g to 200 g for 10 minutes at room temperature to separate the red blood cells at the bottom of the tube, the buffy coat (containing the white blood cells) in the middle and the plasma above (soft spin). Then, the upper plasma was pipetted above the buffy coat to undergo another centrifugation at 1500 g to 2000 g for 15 minutes (hard spin) to obtain a platelet pellet in the bottom of the tube (with a platelet count 4-4.5 times higher than that of baseline), and a Platelet-Poor Plasma (PPP) in the upper part [32]. The PPP is partly removed and partly used to resuspend the platelets to finally produce 2 ml of PRP. Platelet-rich plasma was activated by adding 10% calcium chloride 0.1 ml per 0.9 ml plasma. Local anesthetic cream (eutectic mixture of lidocaine and prilocaine) was applied to the face for approximately 45 min to 60 min before the procedure. After sterilization of the face with alcohol, 0.1 to 0.3 ml PRP was injected intradermally into the atrophic scars using insulin syringe with a total of 1 ml PRP in each side of the face (Figure 5). Gentle massaging was performed after the procedure, followed by topical antibiotic for 3 days after treatment, but application of sunscreen was not required.
Discussion
Because of the prevalence of acne scarring and the strong negative
emotions it causes in affected patients, dermatologists are frequently
presented with the challenge of evaluating and providing treatment
recommendations to patients with acne scars. The efficacy and the
safety of PRP in the treatment of atrophic acne scars and hyper
pigmentation is a today’s cardinal treatment. To the best of our
knowledge, we evaluated the efficacy and safety of PRP intradermal
injection alone in the treatment of atrophic acne scars. Platelet-rich
plasma is an autologous preparation of platelets in concentrated
plasma. It has recently attracted much attention in various medical
fields, including orthopaedic, plastic, and dental surgeries and
dermatology for its wound-healing ability. Platelets release various
cytokines and growth factors that promote angiogenesis, tissue
remodeling, and wound healing. Platelet-rich plasma works by the
degranulation of granules in platelets, which contain the synthesized
and prepackaged growth factors. Many growth factors have short
half-lives, therefore greatest effectiveness may result if they are
activated at or just before injection. Fibroblasts accumulate at the site
of injection and start to lay down collagen.
Activated platelets release several growth factors, cytokines, and
chemokines, including vascular endothelial growth factor, plateletderived
growth factor, epidermal growth factor, fibroblast growth
factor, transforming growth factor-b, insulin-like growth factor,
IL-8, macrophage inflammatory protein-1a, and platelet factor- 4.9.
Platelet-rich plasma separation involves centrifugation of the wholeblood
by single-spin or double-spin method. The single-spin method
separates the whole blood into 3 basic components: red blood cells
(bottom of the tube), PRP (middle of the tube), and PPP (top of the
tube). PRP with a single spin would not produce a true PRP. Instead,
it would produce a mixture of PRP and PPP with low platelet counts.
Regardless of the rate of centrifugation or the time of centrifugation,
a single spin cannot adequately concentrate platelets because the red
blood cells will interfere with the fine separation of the platelets. The
device must use a double centrifugation technique. The first spin
will separate the red blood cells from the plasma, which contains the
platelets and the second spin will finely separate the platelets from
the PPP. The second centrifugation produces a platelet pellet that
can be easily resuspended with maximum platelet concentration and
the least platelet loss in the above PPP. The produced PRP contains
almost no blood-derived cell types other than platelets.
Leukocytes are absent in PRP prepared by this method as they
should be avoided in PRP preparations because of their potential
pro-inflammatory effect. PRP intradermal injections alone in the
treatment of different types of acne scars were applied. Promising
results have been achieved using this PRP method as compared with
traditional methods.
References
- Niwano T, Terazawa S, Nakajima H, Wakabayashi Y, Imokawa G. Astaxanthin and withaferin A block paracrine cytokine interactions between UVB-exposed human keratinocytes and human melanocytes via the attenuation of endothelin-1 secretion and its downstream intracellular signaling. Cytokine. 2015;73(2):184-97.
- Cheng J, Vashi NA. Treatment Strategies for Hyperpigmentation. Dermatoanthropology of Ethnic Skin and Hair. 2017; 417-36
- Frelinger AL, Torres AS, Caiafa A, Morton CA, Berny-Lang MA, Gerrits AJ, et al. Platelet-rich plasma stimulated by pulse electric fields: Platelet activation, procoagulant markers, growth factor release and cell proliferation. Platelets. 2016;27(2):128-35.
- Mehryan P, Zartab H, Rajabi A, Pazhoohi N, Firooz A. Assessment of efficacy of platelet-rich plasma (PRP) on infraorbital dark circles and crow's feet wrinkles. J Cosmet Dermatol. 2014;13(1):72-8.
- Sabrkhany S, Kuijpers MJ, Verheul HM, Griffioen AW, oude Egbrink MG. Platelets: an unexploited data source in biomarker research. Lancet Haematol. 2015;2(12):e512-3.
- Banihashemi M, Nakhaeizadeh S. An introduction to application of platelet rich plasma (PRP) in skin rejuvenation. Reviews in clinical medicine. 2014;1(2):38-43.
- Farghali HA, AbdElKader NA, Khattab MS, AbuBakr HO. Evaluation of subcutaneous infiltration of autologous platelet-rich plasma on skin-wound healing in dogs. Biosci Rep. 2017;37(2).
- Ma S, Xie N, Li W, Yuan B, Shi Y, Wang Y. Immunobiology of mesenchymal stem cells. Cell Death Differ. 2014;21(2):216-25.
- Lee MH, Noh TK, Lee JH, Roh MR, Na JI, Jung EC, et al. Clinicoepidemiological Features of Melasma in Korean Patients at Five University Hospitals: A Cross-sectional Multicenter Study. Korean J Dermatol. 2016;(7):532-7.
- Nicolaidou E, Katsambas AD. Pigmentation disorders: hyperpigmentation and hypopigmentation. Clin Dermatol. 2014;32(1):66-72.
- Eshghi G, khezrian L, Ashari FE. Comparison between Intralesional Triamcinolone and Kligman's Formula in Treatment of Melasma. actaMedicaIranica 2016;54(1):67-71.
- Handel AC, Miot LDB, Miot HA. Cosmetic Approach for Melasma. Daily Routine in Cosmetic Dermatology. 2017;1-14.
- Lee HC, Thng TGS, Goh CL. Oral tranexamic acid (TA) in the treatment of melasma: A retrospective analysis. J Am Acad Dermatol. 2016 Aug;75(2):385-92.
- Tamega A, Miot HA, Moco NP, Silva MG, Marques MEA, Miot LDB. Gene and protein expression of oestrogen-β and progesterone receptors in facial melasma and adjacent healthy skin in women. Int J Cosmet Sci. 2015;37(2):222-8.
- Ali S, Bali S, Sharma RP. Hormonal evaluation in females having melasma. JEMDS. 2015;4(76):13240-7.
- Raju BTVN, Kumar GA, Vardhan KRH. A clinicoepidemiological study of melasma in a tropical area. J. Evolution Med. Dent. Sci. 2016;5(79):5894-7.
- KrupaShankar DS, Somani VK, Kohli M, Sharad J, Ganjoo A, Kandhari S, et al. A Cross-Sectional, MulticentricClinico-Epidemiological Study of Melasma in India. Dermatol Ther (Heidelb). 2014;4(1):71-81.
- Choubey V, Sarkar R, Garg V, Kaushik S, Ghunawat S Sonthalia S. Role of oxidative stress in melasma: a prospective study on serum and blood markers of oxidative stress in melasma patients. Int J Dermatol. 2017;56(9):939-43.
- Ogbechie-Godec, OA, Elbuluk N. Melasma: an Up-to-Date Comprehensive Review. Dermatol Ther (Heidelb). 2017;7(3):305-18.
- Chuah SY, Thng TGS. Diagnosis of Melasma in Brown Skin: Wood’s Lamp, Dermoscopy, and Confocal Microscopy. In: Handog E., Enriquez-Macarayo M. (eds) Melasma and Vitiligo in Brown Skin. Springer. 2017; 41-9
- Lee MC, Chang CS, Huang YL, Chang SL, Chang CH, Lin YF, et al. Treatment of melasma with mixed parameters of 1,064-nm Q-switched Nd:YAG laser toning and an enhanced effect of ultrasonic application of vitamin C: a split-face study. Lasers Med Sci. 2015;30(1):159-63.
- Wolfshohl JA, Geddes ERC, Stout AB, Friedman PM. Improvement of erythema dyschromicumperstans using a combination of the 1,550-nm erbium-doped fractionated laser and topical tacrolimus ointment. Lasers Surg Med. 2017;49(1):60-2.
- Bagherani N, Gianfaldoni S, Smoller B. An Overview on Melasma. Pigmentary Disorders 2015;2:216.
- vanGeel N, Speeckaert R. Acquired Pigmentary Disorders. Rook's Textbook of Dermatology, Ninth Edition. 2016;1-65.
- Tienthavorn T, Poohglin T, Sudtikoonaseth P. Patch testing and Histopathology in Thai patients with hyperpigmentation due to Erythema dyschromicumperstans, Lichen planuspigmentosus, and Pigmented contact dermatitis. Asian Pac J Allergy Immunol. 2014;32(2):185-92.
- Rodrigues M, Pandya AG. Melasma: clinical diagnosis and management options. Australas J Dermatol. 2015;56(3):151-63.
- Fabbrocini G, Cacciapuoti S, Izzo R., Mascolo M, Staibano S, Monfrecola G. Efficacy of Narrowband UVB Phototherapy in Erythema DyschromicumPerstansTreatment:Case Reports. Acta Dermatovenerol Croat. 2015;23(1):63-5.
- Henry-Kirk RA, Plunkett B, Hall M, McGhie T, Allan AC, Wargent JJ, et al. Solar UV light regulates flavonoid metabolism in apple. Plant Cell Environ. 2018;41(3):675-88.
- Dai NT, Chang HI, Wang YW, Fu KY, Huang TC, Huang NC, et al. Restoration of skin pigmentation after deep partial or full-thickness burn injury. Adv Drug Deliv Rev. 2018;123:155-64.
- Ryan J Sullivan, David E Fisher. The Biology of Pigmentation. In: Fisher D, Bastian B, editors. Hematol Oncol Clin North Am. 2014;28(3):437-53.
- Cervelli V, Garcovich S, Bielli A, Cervelli G, Curcio BC, Scioli MG, et al. The Effect of Autologous Activated Platelet Rich Plasma (AA-PRP) Injection on Pattern Hair Loss: Clinical and Histomorphometric Evaluation. BioMed Research International. 2014;2014:9.
- Gentile P, Garcovich S, Bielli A, Scioli MG, Orlandi A, Cervelli V. The Effect of Platelet-Rich Plasma in Hair Regrowth: A Randomized Placebo-Controlled Trial. Stem Cells Transl Med. 2015;4(11):1317-23.