Research Article
Recurrent Hiatal Hernia: Evolving Definitions and Clinical Implications
James M Tatum* and John C Lipham
Department of Surgery, Keck School of Medicine of the University of Southern California, USA
*Corresponding author: James M Tatum, Department of Surgery, Keck School of Medicine of the University of Southern California, 1450 San Pablo Street, Healthcare Consultation Center 4 Suite 6200, Los Angeles, CA 90033, USA
Published: 24 Jan, 2018
Cite this article as: Tatum JM, Lipham JC. Recurrent Hiatal
Hernia: Evolving Definitions and Clinical
Implications. Clin Surg. 2018; 3: 1884.
Abstract
Recurrence of hiatal hernia after surgical repair remains of the most challenging problems in general
thoracic surgery. Multiple adjuncts to crural closure have been suggested and implemented in the
past three decades. Measuring and comparing rates of recurrence between methods of repair is
made challenging by inconsistent definitions and follow-up periods between studies.
Keywords: Hiatal hernia; Hiatal hernia repair; Videoesophagography
Key Points
• Adjusted freedom from recurrence is often lacking in reported studies.
• Standard size criteria for recurrence grading are needed.
• Consensus on appropriate modality for diagnosis of small recurrence needs to be achieved.
Introduction
The laparoscopic repair of a hiatal hernia was first described in a 1992 in case report [1]. Recurrence of hiatal hernia following laparoscopic repair were reported soon thereafter [2]. Recurrence of hiatal herniation after surgical repairremains one of the challenges of thoracic surgery, and plagued the specialty even when repairs were performed through open surgery. A large recurrence is easily diagnosed by any of several diagnostic modalities, but there remain inconsistencies on how we define the lower limits of recurrence and by which modalitythe diagnosis is most appropriately made. Varying follow-up times reported in studies compound the difficulty in understanding or comparing recurrence rates among methods of repair. The progression (expansion) of recurrent hiatal hernia over time in the setting of a failed repair is virtually unexamined in the literature. It is not clear if the natural history of the recurrent hiatal hernia parallels that of an untreated hiatal hernia or follows a more benign course. Consistent criteriafor diagnosis is essential for the study of the disease and in planning appropriate surveillance and treatment of patients not only during study design, but in clinical practice. The aim of this article is to review how select publications describe recurrence rates of hiatal hernia--examining how as a surgical community we are measuring and definingthe lower limits of recurrence.
Methods
Non-systematic review of the surgical literature.PubMed, SCOPUS and Embase were searched for all articles mentioning “hiatal hernia”, “paraesophageal hernia” or “recurrent hiatal hernia”. Articles were selected based on relevance by authors JMT and JCL to represent a cross-section of major publications since the introduction of laparoscopic hiatal hernia repair to surgical practice. Additional criterial for inclusion were that the article describes: type of hernia (I-IV); modality of pre-operative diagnosis; method of intra-operative measurement; criteria for diagnosis of postoperative recurrence; modality by which recurrence is diagnosed; time of imaging follow-up, proportion of patients in which post-operative radiographic follow-up was obtained; recurrence rates.
Results
Forty-four studies were deemed representative of current clinical practice; the elevenmanuscripts most completely reporting the predetermined criteria, from 1999 through 2017 were included in this analysis.
Table 1
Table 2
Table 3
Definition
Findings are summarized in (Table 1). Recurrent hiatal hernia carried definitions including “any evidence of herniation of the stomach above the diaphragm”, “any gastric tissue in the thorax”, as well as more specific size criteria. Early studies are more likely to subjectively define recurrent hiatal [3,4] later studies largely define recurrent hiatal hernia as >2 cm of gastric tissue above the diaphragmatic hiatus [5,6]. Several studies distinguish between small and not-small hiatal hernia recurrences at a threshold of 4 cm [5,7].
Modality of Assessment
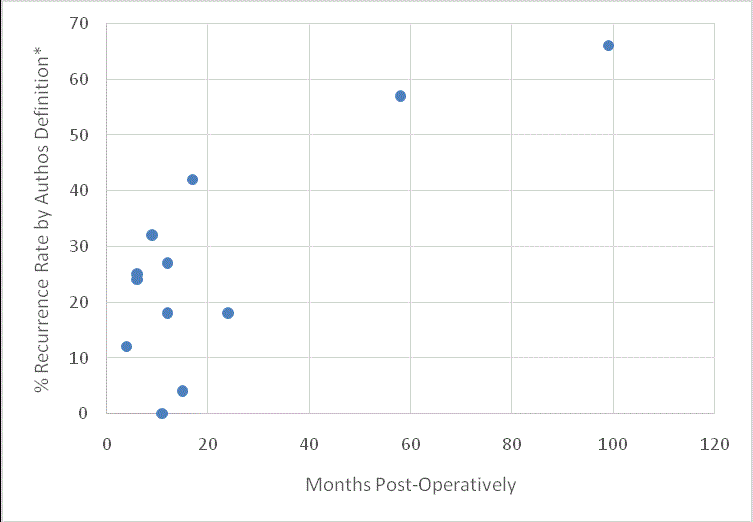
The modality by which recurrence is diagnosed is generally by a contrast Barium Esophagram (BE) (variably referred to as UGI, barium esophagram, or videoesophagram). One early study used CXR as an adjunct diagnostic in patients who failed to receive a BE [8]. The use of EGD to diagnose recurrent hiatal hernia occurs in several of the papers, generally from the USC group, and only as a supplemental means of diagnosis when patients had not receive the intended barium esophagram [9-11]. Recurrence rates as observed in the studies we examined are shown in (Figure 1).
Follow-up Time
Length of follow-up time to diagnosis varies widely in this sample of studies and is not always clearly defined or completely reported. Follow-up times during which recurrence is diagnosed range from 4 months to beyond 8 years [4,6]. Median and means are variable reported, based on sample size and distribution of follow up, from 3 months to 99 months [3,6,12]. Recurrence rates vary over the course of the studies (as to adjuncts to repair and changes in technique), however they range from 66% to 0% [4,13].
Progression over Time
Two of the studies that we reviewed in detail discuss progression over time. One study identified 13 patients with recurrent hiatal hernia at 6 months, at subsequent long-term follow up none of these 13 patients had a progression hiatal hernia size [5]. Neither 6 month nor long-term follow-up hernia sizes are reported for these 13 patients [5]. A second study reported that 9 patients that had ≥ 1 additional videoesophagrams performed a year or more after a recurrent hernia was initially detected, and of these, 7 had no change in the size of the hernia at a median follow-up of 12 months after recurrence (maximum 36 months). In 2 patients of the 9 patients, the hernia size increased at subsequent follow-up, in one patient going from 2 cm to 4 cm in 12 months, in the second from 2 cm to 3 cm after 24 months [9].
Discussion
Definition of recurrence
Recurrent hiatal hernia has proven difficult to study. This is in in
part due to a lack of consensus definitions, the relative infrequency of
the operation and the relatively arduousness of follow-up studies for
patients. It has been suggested that recurrence be defined as >2 cm of
gastric tissue above the hiatus. This suggestion was not made based on 2 cm being a threshold predicting progressive expansion of the
recurrent hiatal hernia, but rather on the point at which authors felt
the size of hernia correlated with a worsening of symptoms in their
series, which achieved 88% follow up on BE at 1-year of 101 patients,
88 of whom were followed for one year post-operatively, of whom
only 58% received a repeat barium esophagram at one year with a
28% recurrence rate, this in a series where bio-prosthetic buttressing
was used [14].
While an argument can be made that recurrence should be
based on at what size a recurrent hernia becomes troublesome, or at
what size a clinical can expect further progression, until that data is
reported, this seems a difficult argument to make. We advocate going
forward that not only innovative methods of repair including: relaxing
incisions; use of mesh or topical agents; repair with prosthetic devices-
-but also traditional methods of repair with primary cruroplasty
and fundoplication be accompanied by protocolized follow-up with
the diagnosis of recurrence based on consistent guidelines with the
appropriate and consistent modality. Our suggested post-operative
monitoring protocol is detailed in (Table 2).
Diagnosing a recurrence
Multiple modalities exist to assess the hiatus. Each has potential
downfalls. Small hiatal hernia can be transient, and their presence
is depended on the relative position of the diaphragm during the
respiratory cycle and competing intrathoracic and intrabdominal
pressures [15]. Barium esophagram is regarded as the most reliable
due the dynamic nature of a video esophogram, this benefit is at least
in part attenuated if only static plates are obtained during an UGI
series. Video allows for the GEJ to be observed during each phase
of the respiratory cycle and we believe allows the most clinically
meaningful diagnosis. We have found both HRM and EGD to be
unreliable in the diagnosis of small hiatal hernia due to the dynamic
nature of sliding or transient hernia and the confounding of positive
pressure gastric distension during endoscopy [16,17]. There seems
to be little debate that BE is the appropriate means of diagnosis and
measurement of recurrent hiatal herniation both initially and while
screening or diagnosing recurrence. Screening for a moderate or large
hiatal hernia can reasonable be accomplished by a variety of imaging
modality and they allow a much higher sensitivity and specify as the
hernia increases in size. A summary of our suggested appropriate uses
of imaging modalities is summarized in (Table 3).
Timing and natural history
McMurray et al. performed a meta-analysis in 2007 and included
only studies which quoted recurrence rate of BE in their analysis [18].
They however did not clearly specify either what was considered a
recurrence, or did they consider variable follow up time for the
studies included in their analysis. They argued that studies needed to
be done with better adherence to a scheduled 1-year follow up barium
esophagram. We agree that the need for this study persists, and until
it has been completed there is little literature available allowing the
delineation of the natural history of recurrent hiatal hernia at or
beyond one year besides than mentioned above from the University
of Washington and University of Southern California groups.
Unadjusted (those not considering time from surgery) recurrence
rates should not be reported. Future studies reporting recurrence
rates should carefully detail statistics describing the follow-up data
(mean, SD or Median, IQR and Range) as well as an accounting and
comparison of patients lost to follow-up and those included, as well
as a percent completeness of both clinical and radiographic followup.
Recurrence rates should be reported according to a standard
definition (Table 3) and these recurrences rates (or freedom from
recurrence) should only be reported in patients who have had the
appropriate radiographic study particularly among those diagnosed
with a small recurrence. Ideally each patient would have multiple
studies and progressive time points allowing for a more detailed
Kaplan-Meier survival analysis, in the fashion reported by Reynolds
et al. [11] At the least, further studies that do not achieve follow up
within a narrow margin of variability (i.e. 12 months +/- ≤ 2months)
should report adjusted results accounting for patients lost to followup
with a survival analysis. Stating a single value as a recurrence
rate as measured in patients with inconsistent lengths of follow-up
from a sample with low follow-up rates is deceiving. Understanding
the natural history of a small recurrent hernia requires that patients
receive serial examinations with attention to progression. This is a
critical area in immediate need of further study.
Figure 1
Figure 1
Recurrence of Hiatal Hernia at Follow-Up Time.
*Mean or Median follow up time as quoted. Middle value imputed when
ranges reported.
References
- Congreve DP. Brief Clinical Report; Laparoscopic Paraesophageal Hernia Repair. J Laparoendo Surg. 1991;2:45-48.
- Trus TL, Bax T, Richardson WS, Branum GD, Mauren SJ, Swanstom LL, et al. Complications of laparoscopic paraesophageal hernia repair. J Gastrointest Surg. 1991;1(3):221-7.
- Wu JS, Dunnegan DL, Soper NJ. Clinical and radiologic assessment of laparoscopic paraesophageal hernia repair. Surg Endosc. 1999;13(5):497-502.
- Hashemi M, Peters JH, DeMeester TR, Huprich JE, Quek M, Hagen JA, et al. Laparoscopic repair of large type III hiatal hernia: objective follow up reveals high recurrence rate. J Am Coll Surg. 2000;190(5):553-60.
- Oelschlager BK, Petersen RP, Brunt LM, Soper NJ, Sheppard BC, Mitsumori L, et al. Laparoscopic paraesophageal herniarepair: defining long-term clinical and anatomic outcomes. J Gastrointest Surg. 2012;16(3):453-9.
- Lidor AO, Steele KE, Stem M, Fleming RM, Schweitzer MA, Marohn MR. Long-term quality of life and risk factors for recurrence after laparoscopic repair of paraesophageal hernia. JAMA Surg. 2015;150(5):424-431.
- Diaz S, Brunt LM, Klingensmith ME, Frisella PM, Soper NJ. Laparoscopic paraesophageal hernia repair, a challenging operation: medium-term outcome of 116 patients. J Gastrointest Surg. 2003;7(1):59-66.
- Andujar JJ, Papasavas PK, Birdas T, Robke J, Raftopoulos Y, Gagné DJ, et al. Laparoscopic repair of large paraesophageal hernia is associated with a low incidence of recurrence and reoperation. Surg Endosc. 2004;18(3):444-7.
- Zehetner J, Demeester SR, Ayazi S, Kilday P, Augustin F, Hagen JA, et al. Laparoscopic versus open repair of paraesophageal hernia: the second decade. J Am Coll Surg. 2011;212(5):813-20.
- Alicuben ET, Worrell SG, DeMeester SR. Impact of crural relaxing incisions, Collis gastroplasty, and non-cross-linked human dermal mesh crural reinforcement on early hiatal hernia recurrence rates. J Am Coll Surg. 2014;219(5):988-92.
- Reynolds JL, Zehetner J, Bildzukewicz N, Katkhouda N, Lipham JC. A Durable Laparoscopic Technique for the Repair of Large Paraesophageal Hernias. Am Surg. 2016;82(10):911-5.
- Dallemagne B, Kohnen L, Perretta S, Weerts J, Markiewicz S, Jehaes C. Laparoscopic repair of paraesophageal hernia. Long-term follow-up reveals good clinical outcome despite high radiological recurrence rate. Ann Surg. 2011;253(2):291-6.
- Buckley FP, Bell RCW, Freeman K, Doggett S, Heidrick R. Favorable results from a prospective evaluation of 200 patients with large hiatal hernias undergoing LINX magnetic sphincter augmentation. SurgEndosc. 2017:1-7.
- Lidor AO, Kawaji Q, Stem M, Fleming RM, Schweitzer MA, Steele KE, et al. Defining recurrence after paraesophageal hernia repair: correlating symptoms and radiographic findings.Surgery. 2013;154(2):171-8.
- Ayazi S, DeMeester SR, Hsieh CC, Zehetner J, Sharma G, Grant KS, et al. Thoraco-abdominal pressure gradients during the phases of respiration contribute to gastroesophageal reflux disease. Dig Dis Sci. 2011;56(6):1718-22.
- Tatum JM, Samakar K, Bowdish ME, Mack WJ, Bildzukewicz N, Lipham JL. Videoesophagography vs. Endoscopy for Prediction of Intraoperative Hiatal Hernia Size. Am Surg. 2017.
- Tatum JM, Alicuben E, Bildzukewicz N, Sandhu K, Samakar K, Houghton C, et al. Intertest Reliability of Hiatal Hernia Diagnosed by High Resolution Manometry, Video Esophagography or Endoscopy and Association with Gastroesophageal Reflux Disease. Unpublished abstract. 2017.
- Rathore MA, Bhatti MI, Andrabi SI, McMurray AH. Laparoscopic repair of paraesophageal hernia requires cautious enthusiasm. Int J Surg. 2008;6(5):404-8.
- Swanstrom LL, Jobe BA, Kinzie LR, Horvath KD. Esophageal motility and outcomes following laparoscopic paraesophageal hernia repair and fundoplication. Am J Surg. 1999;177(5):359-63.
- Oelschlager BK, Pellegrini CA, Hunter J, Soper N, Brunt M, Sheppard B, et al. Biologic prosthesis reduces recurrence after laparoscopic paraesophageal hernia repair: a multicenter, prospective, randomized trial. Ann Surg. 2006;244(4):481-90.